Access to all articles, new health classes, discounts in our store, and more!
Some New Fundamentals for the Prevention of Dental Disease, with Special Consideration of Calcification and Decalcification Processes
Read before the New Jersey State Dental Society, Asbury Park, April 19, 1928. Published in Dental Cosmos, February 1929.
* * *
The dentistry of the past and of the present has been chiefly concerned with repair and replacements, and while this has constituted a great blessing to humanity, it is very far short of the ideal service to be rendered by dental science.
Dental practice has not been unmindful of the need for this better service. The fact remains, however, that the etiologies of dental caries and pyorrhea have not been known. Each has been considered in terms of local environment, the programs being directed almost entirely toward the combating of these scourges by local treatment. The researches which will be reported in this communication deal almost entirely with factors which are involved in the systemic aspects of these disturbances. Both dental caries and advanced pyorrhea involve hard tissues, and are decalcifications by two quite different processes. The essential first step in determining the etiology and the prevention of decalcifications will be to know some of the processes by which calcium is utilized in normal tooth and bone formation, and in changes which occur as normal processes. This paper presents some new data and discusses other new data recently presented by the author and others which relate to the fundamental processes of calcium utilization.
As an approach to the problem, some general observations are important, and we can present these most readily as questions. Why does dental caries attack individuals at particular periods, which periods are directly related to stresses, for example, those of childhood and of the early teens, gestation, lactation, febrile disturbances and privation? Why does pyorrhea become conspicuously apparent beyond forty years of age, and why is it particularly severe with certain metabolic disturbances, such as diabetes, or in such an infectious disease as miliary pulmonary tuberculosis? Over against these clinical expressions we are wont to ask the question: Why are certain growth disturbances of childhood markedly present in certain seasons of the year and absent in others, and why does such a disturbance as rickets involve 90 per cent of the children under two years of age in certain locations, whereas it is practically unknown under certain other conditions? Why are the dental structures which have developed at one age of an individual different in susceptibility to dental caries from those of another period? This last is strikingly illustrated by the following case: A young woman presented at sixteen with much enamel and dentin destroyed by caries. This condition developed at twelve to fifteen years of age. But the caries had attacked chiefly the first permanent molars and incisors, the bicuspids being quite immune. It is evident that there occurred in this individual a major disturbance at the time these teeth were in process of calcification, which has greatly contributed to their susceptibility to dental caries. A study of the history reveals that as a small child she suffered with convulsions. This immediately indicates a disturbance of calcium metabolism, since several of the convulsive states, including tetany, are now known to be primarily due to calcium metabolism disturbances.
There are certain outstanding factors in the development of our knowledge regarding the etiology of dental caries and pyorrhea. Perhaps none is more conspicuous than the slowness with which progress has been made. Some of our most carefully observing students of the problems of dental caries and its prevention are agreed that neither prophylaxis nor diet, together or separately, is adequate to check the advance of this disturbance in children in many, if not in most cases. This is also true of the period of very great susceptibility associated with motherhood. The fact that in the entire history of our profession very little progress has been made in determining the etiology of these two conditions strongly suggests that we have overlooked fundamental factors. While this lack of progress has been recognized, the elusive missing factors have not been discernible. The evidence being presented in this paper will relate to the direct influence of irradiation by special rays and by products of irradiation activators not as the sole additional factor, but as very important contributing factors, and it seems quite possible that they are the most important involved.
In our first approach to this problem, we shall consider the disturbances of calcium associated with growth. Very many workers have contributed to knowledge in this field. The bibliography is far too voluminous to include here; it can be found in the various journals dealing with biological chemistry and clinical practice. These have shown in general that certain vitamins or accessory food factors are primarily concerned with function and growth. These are spoken of as vitamins A to E, and are associated with disturbances which are in general as follows: A, antixerophthalmic and growth; B, antineuritic; C, antiscorbutic: D, antirachitic; E, fertility and growth, and development of sex glands; F, antipellagric, is associated with B. It is my belief, based on the available evidence of my own researches and those of others, that a solution of the dental caries problem involves more than the above vitamins; and that other factors are involved which also are related directly and indirectly to radiant energy. The presentation in detail of even the most fundamental of my recent data would require much more space than would be available in this report of a particular phase. These are being put in book form and will include both a review of data already presented in various papers and much that has not been published heretofore.
In 1923, I discovered that the exposure of rabbit serum to ultraviolet irradiation produced changes which were recognized by the two following procedures: When this serum was exposed to radiant energy and also placed beneath a photographic plate, it produced a distinct fogging of the plate, the nature of which was later determined to be due to a process which involved the production of hydrogen dioxide; also, when this serum, so treated, was injected intravenously into a rabbit, it produced a distinct change in the calcium level of the blood.1 An extended study was made of the effect of exposing various substances to irradiation, the results being presented in various papers.2,3,4 A striking illustration of the direct effects of irradiation was shown as a result of the exposure of cod-liver oil for different periods of time. It was revealed that the effect upon the photographic plate was approximately proportional to the amount of irradiation up to a certain point.
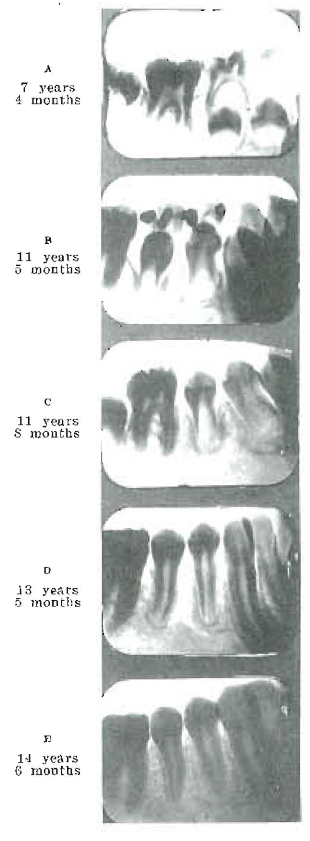
During the past seven years an important phase of these researches has involved studies of the blood with regard to the morphological and chemical characteristics, particular attention being given to calcium and phosphorus in various phases. At this time the chemical studies furnish data from 1600 patients in which the calcium factors had been intensively studied. During this time a very large number of experiments with animals also have been made. Special effort has been made to relate the chemical findings to the clinical observations. Several things are clear from a comparative study of the blood chemical analyses and the clinical conditions, made both before and after treatment, among them the following: (1) That the range of variation that is possible in several calcium and phosphorus factors is quite limited if the individual is to have normal health and normal defense against dental caries; (2) that certain relationships between different factors constituting degrees of balance or of imbalance seem to be more important than total quantities; (3) certain of these imbalances seem quite directly subject to influence by treatment; (4) there is evidence of marked clinical change, with changes in the blood chemical pictures. As an illustration, note the change in Fig. 1.
This boy has the following general history: At eleven years and three months of age he was brought to us with a history of lassitude, poor appetite and not able to attend school regularly. Roentgenograms had been made of his teeth at seven years and four months of age. These showed the developing crowns of the bicuspids as seen in Fig. 1, a. At eleven years and five months, the deciduous molars were still in place and the roots showed much less advancement in calcification than would be normal for his age, and the supporting alveolar bone showed poor calcification. After being placed on treatment to improve his calcium metabolism, there was a rapid and marked change in his general physical condition. It expressed itself in many ways, including a desire to go to school, to play with his playmates, and a marked improvement in appetite and in sleeping.
Within a month’s time after beginning the treatment, the deciduous teeth were shed spontaneously. They began to get loose very soon after the treatment was started. The blood chemical change was particularly noticeable, in that the calcium balance as expressed by the product of serum calcium and serum phosphorus, +40 or 40 (40 is normal), increased from -26 to +33 (a change from 14 to 73). In sixty days he gained four pounds in weight. The blood serum inorganic phosphorus increased from 2.65 to 7.71, and the serum calcium from 9.06 to 9.56, changing the calcium balance as just indicated. During this sixty-day period he grew eleven-sixteenths of an inch. His appetite changed; from shunning fruits and vegetables, he came to like them; and his nervous state changed from being listless and irritable to being very anxious to go to school. His general physical appearance was very greatly improved. In two and a half years he grew in height seven and thirteen-sixteenths inches and increased in weight thirty-six and a half pounds, a net gain of 50 per cent in weight. For most of his life he had been a semi-invalid; he has rapidly changed to a vigorous, athletic boy, keen mentally and strong physically.
Fig. 1–The speeding up of delayed calcification by use of activators. Treatment began at eleven years of age. Increased calcification was associated with marked clinical improvement.
This change was produced by putting into his system a type of activator which very markedly changed his ability to utilize the calcium and phosphorus present in his food, and to metabolize them for utilization and growth. The degree of activation of the cod-liver oil which was given to him in small doses was in excess of what we would use now with increased experience. I do not deem it necessary or best to change an individual so rapidly from a negative to a positive phase as we did this boy. The treatment given him at first was about one and a half grams of activated cod-liver oil with each meal. It was activated by being exposed to direct sunshine for from fifteen to thirty minutes (I now use much shorter exposure, one to five minutes) for each one-sixteenth inch of depth in the shallow tray in which it was exposed. With this, he received with each meal two 5-gr. calcium lactate tablets, besides drinking large quantities of milk. The progressive development of the roots of the teeth is shown in letters A to E in Fig. 1.
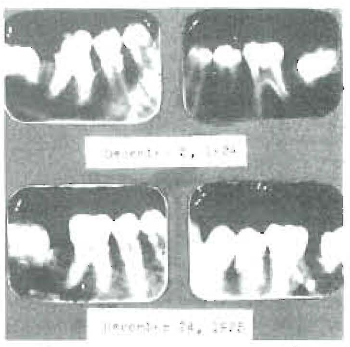
Another of the serious disturbances which tremendously affects child life, both because of its direct distress and serious handicapping of efficiency later, as a result of the sequela, is the rapid caries of childhood. I am seeing frequently very distressing cases where, in spite of every effort of the dentist in charge and of vigorous, prophylactic care at home, the teeth are rapidly disintegrating from caries. A striking illustration is a boy of fourteen, roentgenograms of whose teeth are shown in Fig. 2, presented with seventeen cavities and a very marked transparency of the bicuspid roots. As is typical of such cases, his nervous condition was so disturbed that it was with very great difficulty that operations could be made. This is in part because the disturbed calcium metabolism greatly increases the sensitivity of the nervous system. Roentgenograms of his teeth one year later are shown in the same figure and reveal a very marked change in the degree of calcification, both of the tooth roots and the supporting alveolar bone.
Fig. 2–Severe dental caries with seventeen open cavities in boy of fourteen years, completely checked by treatment. Note reduction in size of pulp chambers in one year’s time, especially in bicuspids.
During the year which followed placing him on the treatment, he developed only one very small cavity and it is a question whether this had not been overlooked the year before. Incidentally, the hypersensitivity of the nervous system is very greatly relieved by treatment which increases the calcium utilization. This is strikingly illustrated by a recent case sent from some distance because the boy of fourteen could scarcely endure having his teeth operated upon. Upon studying him, I found that simply drawing the rough handle of an instrument across his teeth produced such distress as to almost send him into paroxysms. As a result of treatment he so greatly improved that several cavities could be filled at one sitting, and in three months he gained five and three-fourths pounds in weight and one and five-eighths inches in height. A characteristic of these cases is usually abnormally large pulp chambers, due to a lack of progress in calcification by the odontoblasts, a result of which is that even moderate caries at certain positions tends to easily expose the pulp.
Caries in Pregnancy
Another of the distressing conditions which we encounter is that of the rapid caries attendant upon pregnancy and lactation. It is common knowledge among the laity, as well as among numbers of the healing professions, that these constitute periods of very great stress. “A tooth for every child” is the old adage taught by our grandmothers, and one which we have so often seen verified in some families. It is my belief that this condition also should be considered as a symptom of disturbed calcium metabolism.
A striking illustration will be found in the following case of pregnancy at twenty-four years of age, with a history of heart involvement in childhood, associated with acute rheumatism. In Fig. 3 I have presented in detail three blood chemical analyses of her case. The first studies were made in the fourth month of pregnancy, at which time her calcium phosphorus product was 34 (6 less than 40). Unfortunately, she was not able to return for two months, due to illness of her mother, at which time her physical condition had become quite markedly disturbed. At this time her calcium-phosphorus product was 21. Notwithstanding the fact that the demands of her system for calcium were increasing at an accelerating rate beyond the six months, under treatment, which began immediately after the second blood study, she was placed in positive calcium balance, the calcium-phosphorus product being 40.8, an increase of twenty points. Associated with this was a very marked improvement in her physical condition, which she and her attending physician attributed to the special treatment for improving the calcium metabolism. The birth was normal, the mother and child being in excellent condition. The caries, which was quite active before the treatment, ceased after the condition of negative calcium balance was relieved and there was very marked improvement in her general condition and in the heart action. The husband reported with earnestness that it was his belief that the changed conditions saved the life of the child and contributed greatly to the safety of the mother.
Fig. 3–Blood chemical and microscopic changes in a case of pregnancy: (A) Fourth month; (B) sixth month; (C) eighth month. Treatment began at B. Note improvement in serum calcium and inorganic phosphorus.
A number of such cases could be used as illustrations; some have been reported in preceding papers, but the present cases will illustrate this phase of the problem.
If the administration of activators can produce distinct changes in calcium anabolism and catabolism and modify the body defenses both for the maintenance of physical efficiency and prevention of abnormal states, by what processes do these physical betterments occur? If it is simply a matter of radiant energy, why should not the individuals be exposed to this force, and what is the evidence for and against this procedure? And, further, what kind of radiant energy should be used? Or if the same results can be obtained by the administration of cod-liver oil, why bother with radiant energy either on the patient or on the oil, and what are the reasons for and against each procedure? And, further, is there a difference in the different samples of cod-liver oil?
Let us first study the effects of exposure to radiant energy from a liberal source of ultraviolet rays. This immediately raises the question as to what constitutes a liberal source of ultraviolet rays. While there is great need for brevity, it is desirable that we shall understand somewhat of the difference in the radiation from different sources. In order to obtain a basis for judgment in this matter, I have designed and had constructed three spectrographs for different purposes. One is for very accurate determination of amounts of radiant energy in different parts of the spectrum of radiation from various sources (Fig. 4); a second, for determining and recording the amount of radiant energy from the sky, made both very rigid and portable; it can be used either on a tripod at attached to the inside of the door of the automobile, and passes outward and upward when the glass is lowered. This is shown in Fig. 5. It can be shifted to any angle or direction and permits of rapid recording of the relative and actual amounts of radiant energy for different parts of the spectrum in different localities. The third is built so large that two series of fifteen rats can be exposed at once, each pair, one individual above the other, receiving a different part of the spectrum, for determining the effect of different groups of bands. With these instruments, I have, with the aid of the technicians, obtained very important and helpful new data.
Fig. 4–Specially constructed spectrograph for exposing fluids to different parts of the spectrum and for recording absorption bands.
Fig. 5–Special portable spectrograph for recording radiant energy from the sky and its spectral distribution.
In my paper2 read before the American Dental Association in October 1927, I presented data indicating the shift in diffusibility of calcium and of various solutions of calcium and proteins, both by biological liquids, such as milk and serum and organic mixtures in solution or water suspension. These have shown that in general the following conditions have been found to obtain: Exposure of the liquids, including such biological solutions as blood and milk, to radiation consisting in part of ultraviolet, such as produced by a quartz-mercury vapor arc, a carbon arc, a high temperature filament, and direct sunlight, increases to a greater or lesser degree the diffusibility of the calcium as determined by a movement of part of the calcium through a colloidal sac. These are of the proper construction to have approximately the same porosity as blood-vessel walls, the osmotic pressure being balanced by a positive pressure inside equal to 150 millimeters Hg., or a negative pressure outside. Further, when raw cod-liver oil is shaken with these substances in a mechanical tumbler for one-half hour, it has the effect of reducing the diffusibility of the calcium of the fluid when the cod-liver oil is removed by centrifuging and separating. When, however, the cod-liver oil is exposed to the above radiations and then shaken with these fluids containing calcium compounds, there tends to be a shift in the diffusibility of the calcium toward an increase instead of the decrease produced by the raw cod-liver oil.
In the study of several calcium factors of blood of individuals in normal and disturbed conditions, it has been disclosed that with the use of colloidal sacs which best approximate tissue conditions, the ratio of diffusible calcium to non diffusible calcium constitutes an equilibrium with nearly equal quantities in each phase, with a slight excess of diffusible over non-diffusible calcium. These studies have shown, however, that variations in the textures of the sacs will cause them to make separations between these two factors at different levels. This will be reported in detail in another communication.
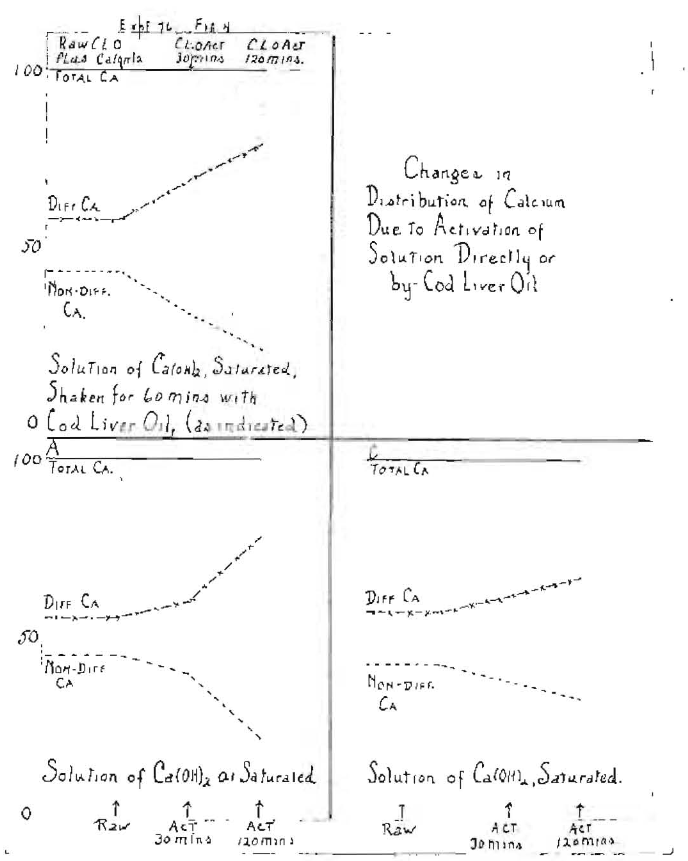
The shift in diffusibility of the calcium in a solution of calcium hydrate, whether it has been shaken with raw cod-liver oil or with cod-liver oil that has been activated, will be seen under these different conditions in Fig. 6. In the first, a saturated solution of calcium hydrate was used. It was shaken for sixty minutes with cod-liver oil, raw or activated, as indicated at the top. The one hundred line shows the total calcium, and the percentages, diffusible and indiffusible, were as follows: 57 to 43 when shaken with raw cod-liver oil; 70 to 30 when shaken with cod-liver oil that had been activated for thirty minutes, and 80 to 20 when shaken with cod-liver oil that had been activated for one hundred and twenty minutes. These were, of course, relatively very long activations as compared with those that we would use on oil that was to be used for medicinal purposes as developed from five years of intensive study on humans and animals.
Fig. 6–Showing shift in the diffusibility of the calcium of calcium hydrate solution, saturated and one-tenth saturated, whether shaken with cod liver oil, raw and activated, or exposed directly, and for different periods of exposure.
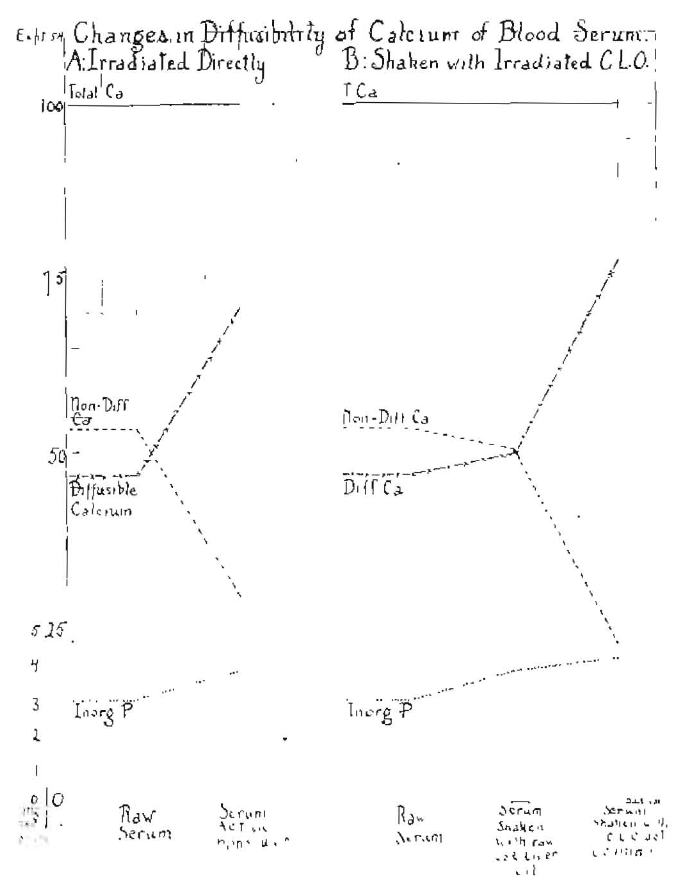
In the second series of Fig. 6, a more dilute solution of calcium hydrate was used, namely, one-tenth saturated. A slightly less marked change was developed. When shaken with raw cod-liver oil, the ratios were as 55 to 45; when shaken with cod-liver oil that had been activated for thirty minutes, it was 60 to 40, and when shaken with cod-liver oil that had been activated one hundred and twenty minutes, it was 78 to 22. The effect of concentration is shown in the lower left of Fig. 6. The effect of the direct activation of the saturated solutions of calcium hydrate gave ratios of diffusible to non-diffusible of 57 to 43; when activated thirty minutes, 62 to 38, and when activated one hundred and twenty minutes, 67 to 33. It is to be noted that we are dealing here with an inorganic form of calcium not associated with an organic substance. When this experiment is made on biological fluids, such as milk and blood, instead of calcium hydrate for variable lengths of time of exposure to irradiation, the results are very similar to what we have shown here. There is also a direct influence on the inorganic phosphorus. This is illustrated in Fig. 7, which demonstrates the effect on the shift and the diffusibility of calcium and inorganic phosphorus in blood serum, whether the serum is irradiated directly or is shaken with cod-liver oil that has been irradiated directly.
Fig. 7–Showing the diffusibility of calcium of blood serum when raw and irradiated directly and shaken with raw or activated cod liver oil.
It is of interest to note that while there is the very marked shift with the activation of the serum from 46.9 per cent to 70.7 per cent, the change is even more marked when the serum is shaken with raw cod-liver oil or with activated cod-liver oil. The change is from 49.8 per cent to 75.2 per cent, the corresponding reductions in the normal diffusibility being from 53.1 per cent to 29.2 per cent, with the serum raw or from direct radiation, 50.2 per cent to 24.7 per cent whether the cod-liver oil is raw or activated. The inorganic phosphorus shifted from 2.8 to 3.7 when the serum was directly irradiated, and from 3.7 to 4.1 whether the serum was shaken with raw cod-liver oil or that which had been activated. We see by these data, first, that there is a marked influence of direct irradiation on the diffusibility of the calcium of the blood, and also that cod-liver oil is able to receive an energy which it can transfer to the blood serum.
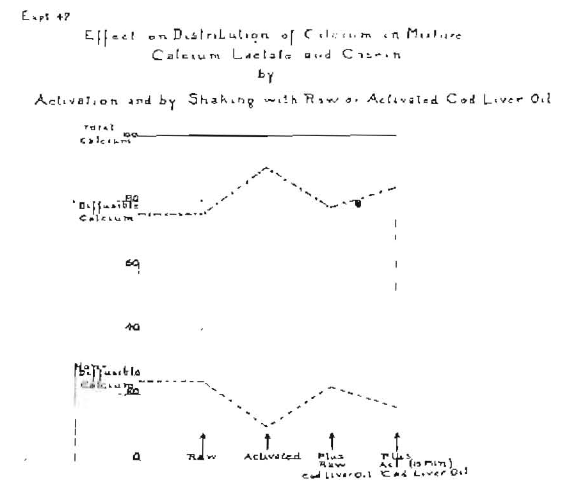
When this experiment was made with milk or with a mixture of casein and calcium lactate, results were obtained similar to those shown with the blood serum. An interesting illustration of the relative influence of raw and of activated cod-liver oil and of activation on the calcium lactate and casein mixture is shown in Fig. 8, in which the level of the diffusible calcium of the mixture is 76.2 per cent and the non-diffusible, 23.8 per cent. When this mixture was activated directly, the diffusible calcium increased to 90 per cent with a corresponding depression of the non-diffusible calcium to 10 per cent. When this mixture was shaken with raw cod-liver oil, the diffusible calcium was at 78 per cent and the non-diffusible was at 22 per cent, and when shaken with cod-liver oil after it had been activated by irradiation for fifteen minutes, the diffusible calcium increased to 84.7 per cent and the non-diffusible calcium decreased to 15.3 per cent. Several hundred experiments have been made on the comparative effect of direct irradiation and by shaking with cod-liver oil and other substances, all of which have shown in general more or less shifting of the diffusibility of the calcium and that several factors are involved, the modification of which directly changes the results. This has included a testing of various parts of the spectrum for relative and actual influence upon the diffusibility of the calcium. Space does not permit of the presentation of these data here.
Fig. 8–Showing a shift in the diffusibility of calcium of a mixture of calcium lactate and casein before treatment, after activation, shaken with raw cod liver oil and shaken with activated cod-liver oil.
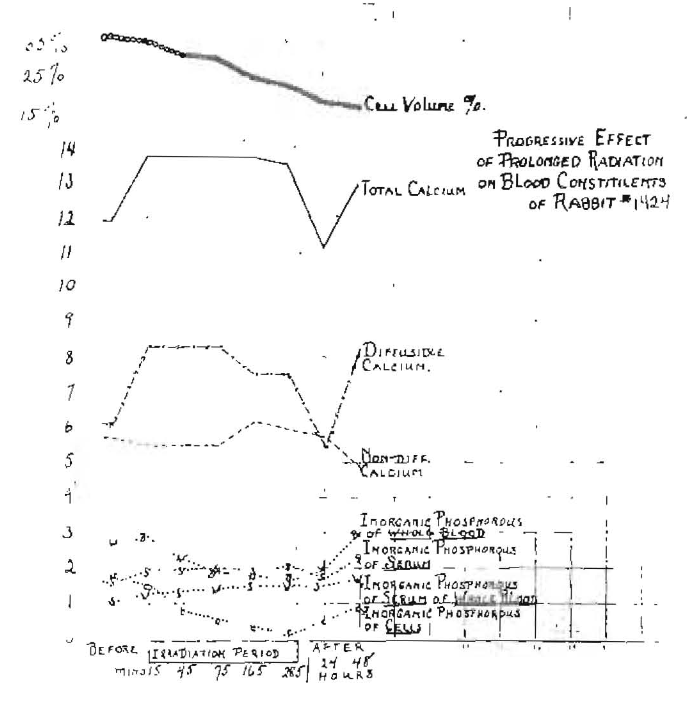
In further illustration of the effect of the full spectrum, we shall observe the changes which have taken place in the blood of a rabbit as shown graphically in Fig. 9. The hair of the rabbit was clipped from its back for an area of about twelve square inches. The rabbit was then exposed to irradiation from a quartz mercury vapor lamp for 285 minutes and small samples of blood were taken at the following periods: 15, 45, 75, 165 and 285 minutes and at twenty-four and forty-eight hours following the exposure. The changes are shown in Fig. 9.
Fig. 9–Blood chemical changes produced by irradiation of a rabbit with mercury-quartz light.
First, it will be noted that the cell volume fell from 35 per cent to 15 per cent of the total blood volume, and that this change was progressive from the start. The rabbit’s total serum calcium at the beginning of the experiment was 11.92. After fifteen minutes of irradiation, the total calcium went up to 13.88 and remained at this level during the time the irradiation was in progress, and then dropped suddenly to 11.2 during the first twenty-four hours. But during the second twenty-four hours it returned to 12.49. By observing the chart, it will be noted that there was relatively small change in the non-diffusible calcium. It first decreased, then increased during the radiation period, and in the post period went progressively down to a point much below its starting level. The change in the diffusible calcium, however, was very marked, increasing over two milligrams as a result of the first fifteen minutes of exposure, and remaining high through the forty-five-minute and seventy-five minute periods, then starting to fall, even though the irradiation was continued, and had been reduced considerably by the end of the period of irradiation. When the irradiation was terminated, there was a sharp fall in the first twenty-four hours in the diffusible calcium, then a rebound in the next twenty-four hours.
Several phases of inorganic phosphorus were changed, including that of the whole blood, of the serum and of the cells. It will be noted particularly that the inorganic phosphorus of the cells was depressed progressively from the very first, there being a slight increase in the serum as a result of the irradiation. The product of serum phosphorus and serum calcium graph was not added here to avoid the crowding of the chart. These figures show an increase for the entire period to the end of the post period of 14.16 to 20.64. The blood morphological changes are not shown but are very marked in such cases. There is at first, with short exposures, an increase in the total leukocytes that practically always is followed by a negative phase or depression which is only slight, with very short exposures, but lasts for a considerable period and a much longer one than the positive phase. With more extended exposures, the extent of the negative phase is increased and may extend over several weeks as a result of a single exposure. There is a direct effect after the depression of polymorphonuclear leukocytes and increase of small lymphocytes. I have previously reported on this phase of the subject and on the effect of blood loss.4
Experiments on the Human
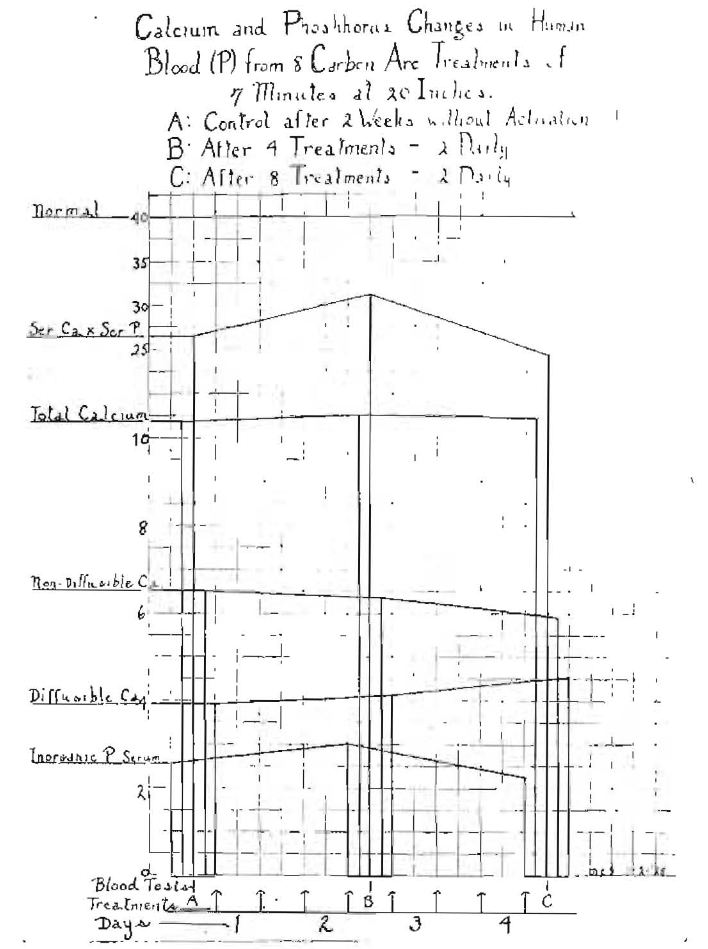
It is particularly important at this juncture, when there is so much interest and misapprehension, largely as the result of lack of information or of misinformation, that data should be presented as to the blood changes which occur in a human being as a result of exposures to radiation from a source that is rich in ultraviolet, such as the quartz-mercury vapor lamp and the carbon are lamp with various carbons. On many occasions, as patients have presented for blood chemical studies, we have found that where they had been receiving treatments from quartz-mercury vapor lamps or carbon arcs, the diffusible calcium was abnormally high, which led me to make tests upon my own system. These are shown in Fig. 10, which shows the results of eight treatments of seven minutes each, taking two daily, morning and evening. A double arc was used, carrying about seven amperes each at 50 volts or at the rate of 350 watts. I have made many experiments on myself and checked them by blood chemical changes, and I am familiar with both my own normal levels and the ease with which I may modify them to advantage or disadvantage. Under ordinary conditions, my efficiency and well-being are greatly enhanced by the taking of the proper quantity of the proper kind of activator. And for these experiments I have stopped the use of activators for a period of two weeks to allow myself to come to my normal level. I previously stated that the diffusible calcium should be slightly higher than the non-diffusible, but at the beginning of this experiment my diffusible calcium was considerably below the non-diffusible, being at 3.9, and the non-diffusible being at 6.5. My inorganic phosphorus was at 2.5, the total calcium at 10.4 and the product of serum calcium and serum phosphorus at 26.5. (Series A, Fig. 10.)
Fig. 10–Blood chemical changes in the author’s blood resulting from treatment with carbon arc light. Note effect of overexposure.
As a result of the first four treatments, two daily (see series B, Fig. 10), my inorganic phosphorus went up to three, the diffusible calcium increased to 4.2, the non-diffusible calcium decreased slightly and the total calcium went up slightly. The Ca x P increased to 32.5. Treatments were continued, and while I experienced a sense of stimulation as a result of the first four treatments, there was a very distinct progressive change in my well-being with a continuation of the treatments. It will be noted that after four more treatments (series C, Fig. 10) the inorganic phosphorus decreased, with the Ca x P at 24.7, resulting in the production of a negative phase of 15.3, and I took the worst cold that I have had in two years as a result of depressing my defenses by overactivation.
Frequently we have patients come to us, or they are sent by physicians, for blood studies to find why their treatments by irradiation are not making them better but are rather making them worse, and we often find evidence suggesting that their condition has been very greatly disturbed to their disadvantage by over irradiation. In a later communication I shall present data indicating the undesirable effects of the extreme ultraviolet part of the spectrum which is furnished abundantly by both of the above artificial sources of ultraviolet rays when used without a filter.
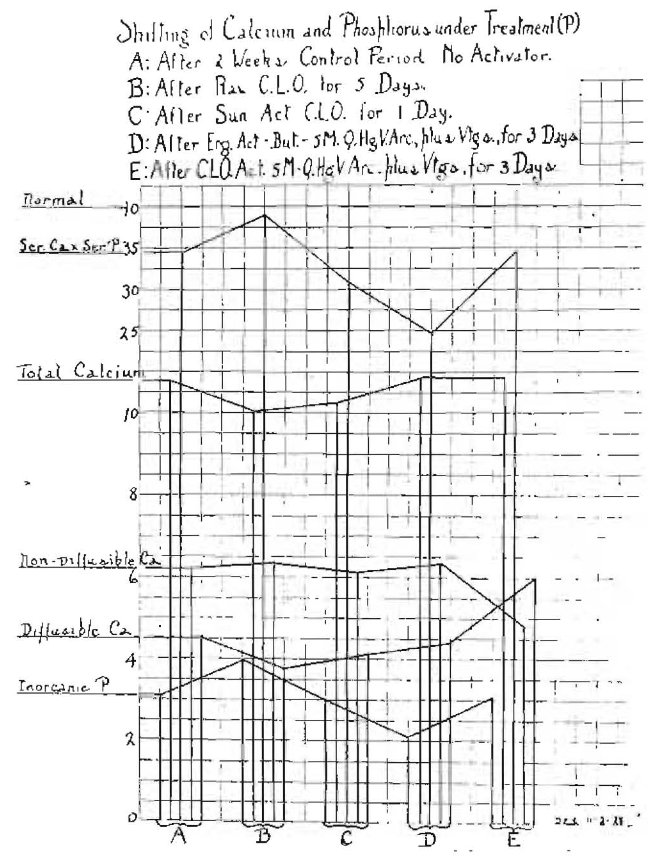
In order that a comparison may be made both between the use of cod-liver oil and of radiant energy, and of cod liver oil, raw and activated, I shall present here in connection with the last experiment another made upon myself. After the two weeks’ control period without activator, the diffusibility of my calcium again went down, this time to 4.5, with my non-diffusible calcium at 6.18, as shown in Fig. 11, A. At the end of the period, the inorganic phosphorus of serum was at 3.1, the total calcium of serum at 10.7 and the serum Ca x P at 33.8. I first took raw cod-liver oil for five days, two capsules with each meal, each containing six-tenths of a gram (0 size). The effect of this is shown in Fig. 11, B. My inorganic phosphorus of serum went up to 3.8; my diffusible calcium dropped to 3.7; my non-diffusible calcium went up slightly to 6.4; my total serum calcium dropped to 10, and my serum Ca x P increased to 38.
Fig. 11–Changes of calcium and phosphorus factors in the author’s blood from the taking of activators by the mouth. Note the ease with which several factors can be changed.
In the next period I took cod-liver oil that had been activated in the sun for fifteen minutes, but only took it for one day, a total of three doses. The effect is shown in Fig. 11, C. The diffusible calcium, which had decreased under the raw cod-liver oil, increased to 4.2. The non-diffusible calcium decreased to 6. The total serum calcium increased to 10.2, the serum inorganic phosphorus decreased to 3, making the serum Ca x P 30.7.
In the next period I took ergosterol that had been activated in butter for five minutes. The activation was done through a filter of Vitaglass, the radiant energy coming from a quartz-mercury arc. This preparation was taken for three days, with the result shown in the graph in Fig. 11, D. Reading from the bottom of the chart up, the inorganic phosphorus went down to 2.2; the diffusible calcium went up to 4.5; the non-diffusible went up to 6.3; the total calcium went up to 10.7, and the serum Ca x P dropped to the low level of 24.5. I could tell by my feelings that something undesirable was going on.
I then changed to a cod-liver oil that had been activated for five minutes through Vitaglass to cut out the shorter ultraviolet radiation to make it more nearly correspond with sun radiation. This was taken for three days. While the blood chemical changes are very striking, the change in my feelings was the most marked part of the data. The blood changes are shown in Fig. 11, E. The inorganic phosphorus went up to 3.2; the diffusible calcium increased to 6 and the non-diffusible decreased to 4.6, reversing and correcting the relationship between these factors. The diffusible should slightly exceed the non-diffusible. The total serum calcium remained about constant at 10.7, but the serum Ca x P increased to 34.5 and was rapidly returning toward the normal of 40. In clinical practice I would not make the change so rapidly.
I have made observations of importance on several occasions with a number of preparations and with radiant energy and treatments, checking my sensations and clinical condition against blood chemical changes, in order that I might understand better the many observations I have had made on animals, which were made with this disadvantage–that we could not obtain information regarding the early clinical changes, because the animals could not report their feelings. We could, however, observe a marked physical depression and in extreme cases even serious physical harm. Another purpose for making this series of studies was to compare the spectrographic changes in the blood. These data are very important and will be included in a separate communication.
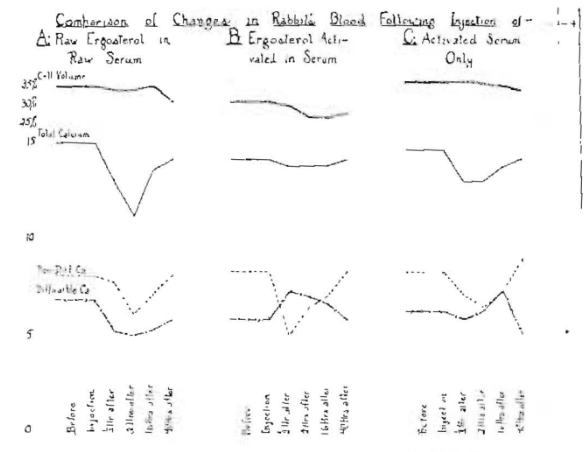
I have similarly made studies of the effect of injecting raw ergosterol added to raw blood serum from the same animal; also ergosterol activated in serum and serum activated alone. The effect is quite different as produced by these different preparations as shown in A, B and C of Fig. 12. This chart shows the changes in cell volume, total calcium, diffusible calcium and non-diffusible calcium, each on the day before the injection, at the time of the injection, and one-half, two, sixteen and forty hours after injection. It will be observed that in series A and C there was a marked fall in the total calcium following the operation and that the fall was very slight in B, where the ergosterol had been activated in the serum, and also that the diffusible calcium went up immediately following the injection of the ergosterol activated in the serum and was still much higher than the normal sixteen hours after, whereas with the raw ergosterol, there was only a marked immediate depression in both the diffusible calcium and non diffusible calcium. An activated serum peak rise is shown at sixteen hours. In other studies we see the peak come between two and sixteen hours, usually about five hours.
Fig. 12–Blood chemical changes in a rabbit from the intravenous injection of each activated serum only (C), 1 cc. raw serum and 2 mg. raw ergosterol (A) and 2 mg. ergosterol in 1 cc. serum activated together (B).
This is very important to note, since there is likely to be some harm done by the administration of ergosterol, raw and activated, by any of several means that might be used. The dosage of ergosterol used here is 2 mg, and in some reports doses larger than this have apparently been given to infants and small children. Since only a small percentage of the ergosterol being activated will be changed by the readily available processes, it may be expected that a wide range of difference will be obtained in clinical results. I accordingly advise against our profession administering this drug until more research data are available.
From these various methods of study, we see how readily calcium utilization can be influenced by each medication, radiation and hypodermic injection. Any and all of these procedures have possibilities for both great good and great harm. It is therefore very important that the dangers of over-treatment should be emphasized. Reduced to its simplest and safest form, it would seem that the administration of small doses, say from ½ gram to 1½ grams, of a mixture of raw cod liver oil and a cod-liver oil that has been activated for five minutes in the sunshine, then placed in capsules and used before chemical change has been allowed to take place, will be the procedure of choice for the members of the dental profession, for use for increasing the defense of growing children against dental caries. This problem is still complicated by the fact that there is still a great difference in different samples of cod-liver oil. In cases of overload, such as pregnancy, this amount should be increased and can to advantage include calcium lactate tablets of five grains each. Blood chemical studies made in this connection will give important information as to the proper amount, and in many cases will establish quite specifically the nature of the treatment that is indicated.
In another communication I have already published data dealing with this problem.
From these and other studies, it seems clearly indicated that dental caries has as its chief etiological factor the presence or absence in the system of activators, which are provided chiefly by foods and exposure to radiant energy. A very important new problem has developed in the matter of determining the relative and actual quantities of activators and vitamins present in various food products, grown or produced under different conditions, such as varying season, whether outdoor or hothouse, and in the case of milk products, the percentage of time the cows are in the stable and in the sunshine. We are finding a very wide variation resulting from these contributing factors, which will be reported in detail in a special text covering this entire series of researches on calcium utilization.
References Cited:
- W. A. Price: “Fundamentals Suggested by Recent Researches for Diagnosis, Prognosis and Treatment of Dental Focal Infections.” Journ. of the A. D. A., June 1925, p. 641.
- Idem: “Calcium Metabolism Studies on the Nature and Role of the Activators. Researches on Fundamentals for the Prevention of Dental Disease.”
- Idem: Dental Infections–Oral and Systemic. Vol. I., 1923.
- Idem: “Calcium, Its Activation, Utilization and Metabolism.” Journ. A. D. A., April 1928, p. 729.
- Idem: “Calcium Metabolism Studies. (A) The Raising of Serum Calcium by Topical Applications of Raw and Activated Cod Liver Oil. (B) Disturbances Associated with the Active Dental Caries of Childhood and Pregnancy.” Amer. Journ. Dis. of Children, January 1927, p. 78.