Access to all articles, new health classes, discounts in our store, and more!
Quality of Food Crops According to Soil Fertility
Published in The Technology Review, Vol. LII, No. 8, June 1950
* * *
The Quality of Our Products Is at Least as Important as Quantity
Generally speaking, it has come to be a well-established fact that forage feed crops of higher nutritional quality demand that the soil delivered into them a larger variety and a greater quantity of inorganic nutrient elements. Reciprocally, we know that there is also a greater exclusion of silica, for example, and also of some of the other elements which are not yet considered essential for plants and animals. Seemingly, it is on the partial basis of these generally known facts that a higher concentration of almost any one inorganic nutrient element, in the dry matter of it as forage, has come to be erroneously considered as proof of better quality of feed. Therefore, high ash contents have held undue prominence in some minds, through the belief in them as proof of quality. High contents of so-called minerals in the forage have been taken all too commonly as indicative of high quality in terms of animal nutrition.
Ash analyses are suggestive by detection of absence, rather than of presence, of essential inorganic elements. Reasoning by means of the positive (that is, “more minerals mean more quality”) is not as reliable a mental procedure in estimation of feed quality as is the reasoning by means of the negative (that is, “absence of certain inorganic elements means less quality”). This latter reasoning holds to the belief that deficiency and absence are much more reliable indications of low quality, rather than the conviction that the presence of certain quantities of inorganic essentials is an indication of high quality. If the forage fails to contain cobalt, for example, there is no inorganic cobalt delivered to the animal. Nor is any delivered to the microbes in its intestinal tract by the synthetic activities of which this inorganic element may become the important newly discovered vitamin B-12. An analysis of ash for the inorganic elements can be taken as index for quality, mainly in the negative aspect, by indicating the absence of quality. Just how much of each element must be present to guarantee high quality is still an unknown fact.
The presence of an inorganic element in increasing amounts, or its positive aspect in the ash, is of little specific value in proving increasing or higher quality. Its presence merely extends the possibility that it may have served in the plant in those synthetic activities that contribute to higher quality of feed. On the other hand, an inorganic element (such as silica, alumina, barium, and other elements coming from the soil) may have merely been “hitchhiking” into the forage–sometimes in large amounts, without appearing to be essential for any plant functions or the synthesis of any compounds representing food quality. It may have been accumulating because of the shortage of some other essential by which it would have been transformed, or diluted through more plant growth or increase of carbohydrates and plant bulk. An element present in the ash is no proof of physiological performance in the plant’s life processes. For physiological service there must come into consideration the integration of the activities of the element in question with those of all the other elements as synthesizers of essential food compounds. Even if they should be correlated with the fertility levels in the soil, such complex activities surely can neither be measured nor specified by an examination that is no more critical than inorganic analyses for the ash contents alone.
One can demonstrate readily that the concentration of a single element (such as potassium) in the forage depends upon the degree to which the clay of the soil is saturated by that element, or the amount of the element on the colloidal clay complex in the soil. This fails, however, to be evidence for higher nutritional value of such forage as feed when the potassium element is present in excess of the animal’s nutritional needs in all vegetation. Both animals and humans excrete a large share of potassium ingested in the food. Then, too, its function in plant physiology is unknown because of its failure to be present in any known organic food compounds; and our ignorance of its functions in our body is serious. Within the plant this element is highly water soluble and mobile. In fact, it is leached out of a crop by rain and put back into the soil toward crop maturity even before harvest. Delivery of larger concentrations of potassium in the forage can be shown to be correlated with the higher contents of potassium in the soil, but this fails to establish a higher nutritional value because of the greater concentration in the feed of this “potash.”
Nevertheless, the soil is the source of all the inorganic essentials for growth of all life. It is also the source of elements that may not be essential but merely “accompany” others going into the plant. The plant is the means by which elements in the soil serve the nutritional needs of the plant itself. By synthesizing organic compounds, the plant also is the means of making the elements serve the nutritional requirements of animals, and finally for man. In speaking of deriving essential inorganic elements from the soil, then, we are dealing with something more than essentiality for plant growth.
The indirect services of the soil are no less significant than its direct services. Because a change in pasture flora occurs with declining soil fertility, the indirect effect of soil fertility on feed quality often goes unrecognized. Whether the effects and influences of inorganic elements from the soil are direct or indirect, in determining the quality of feed, is not the question. If the supply of fertility elements is too low for the soil microbes, and a diminished crop of lowered quality occurs, such results (commonly attributed to the microbe) do not negate the truth of the statement that the quality of the crop above the soil depends on the fertility of that soil. Occasionally it happens that the declining fertility of the soil causes a weed crop, like broom sedge, to take over the pasture where formerly some forage of higher quality, such as bluegrass, prevailed. One might say that the poor forage quality was the result of the introduction of the seed of this weed, or that the broom sedge “took the pasture.” But here again we have a forage of higher quality starved out and one of poorer quality surviving to give much bulk because the lower fertility of the soil prohibited the survival of the better one.
Are we not merely failing to carry the thinking one step further toward the fundamental cause when we say that the change in its flora is the cause of the poorer pasture? Can’t we recognize the soil as the creative support of any crop and say that the lowered fertility of the soil starved out the protein-producing grass but permitted the wood-making sedge to flourish? Isn’t it time to realize that changes in the nutrients, which the soil feeds to the plants, are the cause of the changes in the flora? Changing the flora artificially by our search for, and introduction of, new species–completely disregarding the soil fertility in relation to their qualities as feeds–has already too long been a national research pastime to bring in more crops of greater bulk but of less nutritional values.
Then, often it is said that there are differences in the degree of maturity of the crop when it is harvested, and hence there are differences in the quality of the crop as feed. When we have measured the growing season so habitually by days, it is difficult to conceive of differences in quality due to differences in physiological age, rather than in age according to the calendar. Because it requires a simple mental effort no greater than merely counting the days, the calendar is far more commonly the criterion of maturity than is the physiology of the plant and the different synthetic performances within it. But these factors, rather than the calendar, determine the differences in nutritional values in accordance with the differences in the fertility of the soil. In soils of poorly balanced fertility, plants mature early, and do not require a certain number of calendar days to attain the small amount of food constituents such little fertility permits. When planted in soil of unbalanced fertility, such as occurs with excess of nitrogen, the plant may fail to complete its growth cycle and maturity may be delayed.
An increase in plant mass is commonly accepted as proof of increase in its quality as feed. If we grant that an improvement in feed quality results from those factors of the soil associated with the increase of protein concentration, then we are in error in believing that increased quantity of crop always means increased quality. The very reverse may be true. Concentration of protein and total tonnage of bulk are not causally connected. They may vary either directly or inversely. Less bulk may mean more quality. More bulk may mean less quality. Protein production and its accumulation in the plant are not the direct result of photosynthesis, or of the process which amasses the vegetative quantity. Sugar production and starch accumulation are. Therefore, more quantity of crop may only mean more sugar and more starch, at least in total, if not in concentration. More crops such as these may well indicate additional fuel or energy foods of less-balanced nutritional value. These foods would require that more protein be added from other sources to supplement the animal’s ration. Such has been the case with increased yields of hybrid corn. But with plenty of fertility and with that in balance, it is possible to have both a larger tonnage of bulk and also a higher quality as feed within it.
Soil fertility makes the difference between vegetative bulk with lower or higher nutritional values. Protein production by the plant still requires sugar production by photosynthesis as a precursor. But in terms of soil fertility it also requires the delivery of a great deal more than is demanded for rapidly multiplying the bulk of sugar, starch, cellulose, and other forms of carbohydrates. Protein production within the crop requires many essential elements and processes in addition to the advanced photosynthetic operation of sugar production. The conversion of sugar into protein is a biosynthetic process, occurring at night as well as in day, in which much of the sugar is burned and combined with nitrogen, phosphorus, and sulfur. Of necessity, such a process brings about a reduction of the carbohydrate bulk through respiration. Consequently, the proteins, which are so prominent in giving high quality to feeds as nutrition for higher animals, are built up slowly.
Photo credit: H. Armstrong Roberts
Photo credit: William M. Kittase
Accordingly, then, we can have a crop producing bulk, either rapidly–as a carbonaceous product–or much less rapidly–as a proteinaceous one. This was demonstrated for the former with the absence, and for the latter with the presence, of inoculation of the soybean crop as the only difference in the experiment under the same accurate controls of the plant nutrition by means of the colloidal clay technique. The carbonaceous soybeans without nodulation, and behaving physiologically as a nonlegume, yielded an increase in bulk, or in hay yield, of approximately 50 per cent over that of a similar crop with nodulation from the same seed, behaving physiologically as a legume and raised on the same soil. At the same time, the crop of lesser bulk was delivering 32 per cent more total protein. A higher concentration of the ash elements or fertility (but not necessarily more total) was likewise found in the smaller crop; a requisite for its service in protein synthesis.
Our criterion of crop quality must consider the physiology of the plant which, in turn, depends on the fertility of the soil. The vegetative bulk delivered, and the pedigrees of the seeds initially planted, are by no means guarantees of their performance in synthesizing the complex organic compounds of higher nutritional values. First place may well be given to the proteins and their essential amino acid constituents as a criterion of quality. Such a criterion is one much more exacting than any we have formerly used. As measured by ashing in sulfuric acid, total nitrogen multiplied by any simple mathematical factor to which a majority of the analysts agree, can no longer be considered a guarantee for quality in our forage feeds. Certainly amino nitrogen by the Van Slyke method is a more significant criterion than is total nitrogen. Still better is a more complete amino acid assay, either chemical, spectrographic, or microbiological. The best and final test of plant nutrition is by means of the higher animals themselves. Quality is no longer measurable by simple ash or inorganic analyses. Nor can much credence be placed in any determination of quality that operates on such a belief.
Many of the fertility elements are known to be essential in plant and microbial processes. But by what chemical or other reactions they serve is still unknown. Such cases as calcium essential for protein synthesis by legumes; calcium for nitrogen fixation by microbes in vitro; phosphorus for fermentation, and energy transformations; magnesium for photosynthesis; copper and iron for our respiration; and numerous other cases of essentiality in organo-synthetic services by the inorganic elements, tell us that they are not materials in construction of the final products. They serve as tools during the construction or synthesis of those organic elaborations we call food compounds. Does a chemical examination of such finished compounds reveal all the tools that were required on the construction job, and then were apparently removed after its completion? Little do we appreciate the extent to which soil fertility serves only as a tool. The relation of soil fertility to the quality of the final construction will not be understood, much less appreciated, until all the processes of construction, as well as the final products, are carefully analyzed and are completely understood.
Food creations by plants are dependent on complicated reactions between the soil colloidal complex and the colloids of the root. Viewed broadly, more food quality in forage plants demands more complicated synthetic services by the plants. These services demand more nutrients from the soil and a more specific balance in the array of them in the plant’s ration.
The total amounts of nutrients mobilized into a crop from a particular soil depend on: (1) the amount of crop root surface and clay surface in contact; (2) the respective ionic activities at their interfaces; and (3) the time periods of their contacts. The amount of root surface varies with different crops. The clay surface varies with the texture of the soil. The activities of the soil at its interfacial boundary with the root depend on: (1) the kind, as well as the amount, of clay; (2) the degree of its saturation with nutrient elements; (3) the suite or balanced combination of these adsorbed ions; and (4) the suite of these delivered to the clay from the reserve minerals and organic matter. The activities of the root at its interfacial boundary with the clay depend on: (1) the nature of the root membrane or cell wall as it is modified by the soil contact; (2) this membrane as it is modified by the internal root cell physiology; and (3) the plant’s physiological behaviors modifying the root cells’ contents and their activities.
Nutrient ion movement from the soil to the inside of the cells of the plant root may well be viewed as an ionic activity or movement: first, from the colloidal clay to the colloidal membrane around the cell, or cell wall, and, second, from this membrane to the colloidal (or solution) contents within the cell. As the ions moving into the root are taken out of action, through their elaboration or synthesis into un-ionized organic compounds, the movement of other ions will continue or be hastened in the same direction. If the cell contents are a colloid of high adsorptive and speedy synthetic capacities, like those of the legumes which synthesize many proteins, then much fertility will go into the plant as a guarantee of higher quality in the resulting forage. This will be possible if the root cell contents are compounds of decidedly proteinaceous make-up.
However, if the root contains nothing more complex than a sugar solution or other carbohydrates, with little or no colloidal adsorption-exchange capacities, then little mass movement of ions into the root from the clay of the soil could be expected. This movement into the root would also be less expectable when the clay colloid has a high adsorption energy and high exchange capacity saturated largely with hydrogen. In fact, in that event, movement in the reverse direction–namely, from the root to the clay–may be expected. This is probable since the colloidal root cell membrane might suffer its own breakdown.
Such behaviors were demonstrated for the nutrient ions of nitrogen,1 of phosphorous,2 and of potassium3 when soybeans were grown with the final crop’s (roots, tops, and all) total contents of these nutrient element amounting to less than that of the planted seed. However, if the cell contents are elaborating the entering nutrients into colloidal complexes, and into organic compounds by means of which their ionic activities are lowered (a different cell membrane will also be elaborated), then more nutrient ions will move in from the outer or higher activity areas of this cell wall-clay region. All this encourages still more elaboration and more synthetic performances within the root cells for growth. More fertility from the soil and more synthetic elaborations within the root are the basis for more quality as nutritional values in the resulting forage crops. Less fertility in the soil may move inorganic fertility elements from plant to soil and grow a crop of forage with less of nitrogen, or of phosphorus, or of potassium than in the seed that was planted. Such a hay crop (that couldn’t be a seed crop) would certainly be poor feed when it contained less of each of the above three elements than was in the seed from which it was grown.
Laws of ionic behavior by adsorbed elements are different from those for ions in gas mixtures and in dilute solutions. In contrast to the behavior of ions of ionic mixed gases, the activity of ions entering the root is not alone a function of their mass. Instead, the suites of different ions, with their respective amounts on the clay, influence the amount of activity of each separate ion. In other words, the activity of each ion is influenced by “the company it keeps.” This interplay of ions on the clay to influence their respective activities (which activities are independent of amounts adsorbed and exchangeable) is a most important factor (yet unappreciated) in controlling the physiology of the plant. This is particularly significant when, up to this moment, we have measured the total amount of each exchangeable nutrient in the soil, and have been content to believe that those combinations are the amounts “available” and are moved into the plant in the measured relative proportions. This interionic effect may be the reason for the pronounced movement into the plant of potassium, as compared to other ions, to encourage photosynthetic storing of its carbonaceousness; or of calcium and its host of cohorts for the successful conversion of the carbohydrates into protein of much less crop bulk. Other physiological differences in the plant, provoked by other differences in ionic activities on the clay, because of variation in the components of the suite, rather than in the amounts of the different ions exchangeable on the clay, can readily be envisaged.
Photo credit: Harold M. Lambert
If the suite of ions on the clay, rather than their respective amounts, comes in to control their activities there, then no great stretch of the imagination is required to visualize–inside the root–a similar “interplay” of ions adsorbed on the colloidal cell contents for consequent variations in the elaborations as proteins and other complexes synthesized there. If there is less concentration of protein within the root, there is a lessened degree to which some of the ions on the clay move into the root. There also would be a varied capacity for total ions within the plant. A leguminous root, inoculated and fixing nitrogen to give it a higher concentration of protein within itself, takes in a larger percentage of the potassium exchangeable on the colloidal clay than when the root is not inoculated and when the soybean plant is behaving physiologically as a nonlegume.4 All this variation in root behavior in relation to soil fertility points out that there are fundamental chemical laws in control of plant physiology, and through these laws there is a control of the nutritional value of the products created.
With a higher concentration of the inorganic elements in balance within the forage, there goes a higher concentration of protein, and, in addition, a smaller total yield of forage. This may occur irrespective of the beliefs to the contrary, namely, that more growth as bulk means better quality as feed. In the experiments reported above, the very reverse of this old belief was found to be the case. There was also more total protein per acre, in spite of less total bulk per acre. We now know that the higher concentration of both protein and of inorganic elements (as evidence for higher quality of feed) can go with the lesser yields of crop bulk.
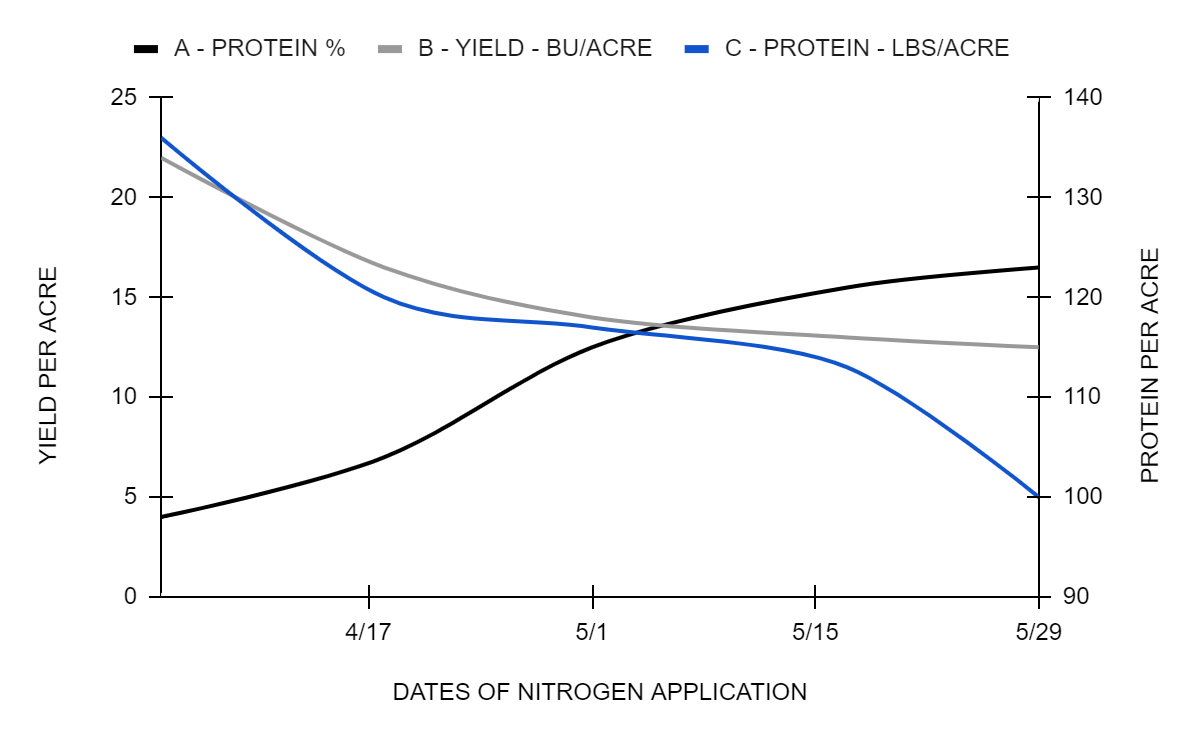
The manner in which the date of application of nitrogen as fertilizer affects yield of wheat is strikingly shown in these curves. Greatest improvement in protein concentration (Curve A) resulted when nitrogen was applied late in the plant’s growth. Nitrogen applied early in the plant’s physiological maturity produced the largest increases in yield (Curve B), and in total protein per acre (Curve C).
Differences in fertility may produce differences in physiological maturity and thereby in nutritional quality. Some other aspects of plant physiology in relation to feed quality deserve attention–namely, the period in the plant’s physiological maturity when there is greatest variation in the suite of ions entering from the soil. Synthetic output, that is, more of carbohydrates or proteins, would be expected to be different, depending on the amount and variety of nutrients taken into the plant. Nitrogen, for example, which is available more generously early, and less plentiful later in the wheat plant’s development, encourages much vegetative growth with a starchy grain of low protein concentration, but produces relatively high bulk yields and possibly even a large amount of protein per acre. That reverse situation (with little nitrogen early but a larger amount of nitrogen available later in the growth cycle) may result in less yields per acre but a higher concentration of protein in the grain.5 It may mean a lower total protein production per acre. If differences in concentration of protein in the grain are considered responsible for these differentiations, “soft” wheat and “hard” wheat are a matter of the time of delivery of a generous amount of nitrogen in relation to other elements. The available nitrogen depends on the conversion of this nutrient from organic to nitrate form by the microbial metabolism of the soil.
The concentration of vitamins in plants deserves some wider consideration in its relation to the fertility of the soil. It raises the question of whether or not the higher concentration of vitamins is a mark of higher nutritional quality. Observations of vitamin C in spinach which had been grown on colloidal clay, under careful control of fertility, suggested a higher concentration of this vitamin when any of the three variables–calcium, nitrogen, and potassium–was in deficiency in relation to the others, as judged by crop appearances and vegetative yields.6 The suggestion has been made that the concentration of this vitamin or catalyst increases in the spinach when any one of these three tested nutrients is so low as to make the “whipping up,” or speedier catalyzing, of its activities necessary. Such a suggestion presents a situation similar to the demonstration of a poorly fed team of horses drawing too heavy a load and being spurred on by the whip. Whether high concentration of a particular vitamin in the forage is a mark of high quality is open to question, particularly if its high concentration is evidence that some nutrient element, and the synthetic processes it supports within the plant, are in deficiency.
Production of food of high nutritional value is a noble objective for soil scientists. Quality of forage feed in relation to soil fertility is a newer concept that is still baffling as an agronomic criterion for wise soil management. For already too long a time have more bulk and higher percentage concentrations of one or all of the inorganic elements been the criteria which we considered sufficient for measuring the values or services given by the soil fertility. But when food production, as the main objective of soil use, forces itself on us more strongly, when the necessity to purchase “protein-supplements” even for fattening a mature animal is so widely recognized, and when the shortages, for example, in the complete array of essential amino acids delivered by plants are more generally appreciated, then the soil fertility, in its more inclusive range of trace as well as major elements, will be considered for its protein-producing power or for all the other qualities of the feeds and foods it gives us. Traditional concepts always cling, of course; and newer ones come to replace them only through studious efforts to comprehend the more fundamental facts. Eventually, we believe, the quality of the feed and food crops will be causally connected with the well balanced and high nutrient levels, or the fertility, in the soil.
References Cited:
- Horner, Glenn M., “Relation of the Degree of Base Saturation of a Colloidal Clay by Calcium to the Growth, Nodulation, and Composition of Soybeans,” Missouri Agricultural Experiment Station Research Bulletin, 232 (1935).
- Hutchings, Theron B., “Relation of Phosphorus to Growth, Nodulation and Composition of Soybeans,” Missouri Agricultural Experiment Station Research Bulletin, 243 (1936).
- Ferguson, Carl E. and Albrecht, William A., “Nitrogen Fixation and Soil Fertility Exhaustion by Soybeans under Different Levels of Potassium,” Missouri Agricultural Experiment Station Research Bulletin, 330 (1941).
- Hampton, H. E. and Albrecht, William A., “Nodulation Modifies Nutrient Intake from Colloidal Clay by Soybeans,” Proc. Soil Sc. Society of America, 8:234-237 (1944).
- Albrecht, William A., “Wheat Is ‘Hard’ or ‘Soft’ According to the Pattern of Soil Nitrogen,” Philfarmer, Second Quarter 1948. Published by Phillips Petroleum Company, Bartlesville, Okla.
- Wittwer, S. H., Schroeder, R. A., and Albrecht, William A., “Vegetable Crops in Relation to Soil Fertility, Vitamin C and Nitrogen Fertilizers,” Soil Science, 58:329-336 (1945).