Access to all articles, new health classes, discounts in our store, and more!
Limestone — The Foremost of Natural Fertilizers
Published in Pit & Quarry, May 1947
* * *
Liming the soil has long been an agricultural art. It has now become a modern scientific agricultural practice. Putting ground limestone on the soil may well be viewed as a case of an old practice that has now come to a newer scientific understanding. It is only recently that scientific facts have given us comprehension of the soil and plant relationships that underlie the services which the limestone renders for the crop.
Limestone has always been associated with the production of leguminous plants. They in turn have always been considered good feed for young and growing livestock. We are just coming around to realize that the calcium supplied by limestone is a fertilizer, as the ancients probably also considered it. Calcium is, in fact, the foremost one of all the nutrient elements for animals and man that can be put into the soil to help it grow bigger crops, giving us better nutrition through them.
We use limestone to supply calcium, rather than to remove soil acidity. Acid, or sour soils, have long been known to be low in productivity. Seemingly like spoiled food about which we say, “It is poor food because it has soured,” so we have come to consider “soured” soils as poor soils. This reasoning has appeared sound because we found that limestone, which reduces the soil acidity, also makes better crops. As a consequence of the observation of these two simultaneous results, namely, reduced soil acidity and better crops, from applying limestone, our soils have been widely tested for their degree of acidity in the belief that the acidity was the cause of the soil’s low productivity. Limestone has been the ammunition in the war on acid soils.
During our belligerency toward soil acidity, a very important fact escaped us. It was the recently recognized one, namely, that while the carbonate part of the limestone is removing the acidity, the calcium going with the carbonate is serving to nourish the crops. Delicate electrical instruments and refined chemical tests helped us to detect and measure the acidity, or the activity of the hydrogen ions that is the cause of it. Since hydrogen is the most active of all elements, and since there are more hydrogen than any other ions in the universe, we might well expect this to be the first one of which the activity would come under such accurate measurement. This attention which was focused so sharply on soil acidity resulted in disregard of the declining and deficient soil fertility.
Unfortunately, neither calcium nor any other plant nutrients have as yet submitted themselves to any such minute evaluations. As a result of these facts, the hydrogen ion or soil acidity has held prominence. This prominence resulted because hydrogen could be easily detected and conveniently measured, and not because of knowledge of its activities in the soils or in the plants. Its presence in the soil did not call attention to the absence there of the calcium, the magnesium, the potassium, and the other plant nutrients, all of which are more deficient as hydrogen is more abundantly present.
Calcium goes with other fertilizers. Increasing the amount of calcium applied for these three series of tomato plants (left to right) suggests trouble from extra lime when only little phosphate was used (lower row) but no injury and increasing benefit as more (middle row) and still more phosphate (upper row) accompanied the calcium. Photo by Missouri Agr. Exp. Station.
It was necessary, therefore, to use the physiology of the legume plants and to study their growth and chemical composition when they were produced on soils of which the acidity was nearly neutralized by limestone, or calcium carbonate, in contrast to these properties of those grown on soils likewise given the plant assay method that revealed the forms of calcium sulphate and calcium chloride, neither of which reduces the soil acidity. It was this plant assay method that revealed the fact that soil acidity is in reality a deficiency in soil fertility. It is a soil situation wherein the hydrogen of no nutrient service to the plants has come into combination with the clay to take the place of several nutrients that are there in productive soils. This method demonstrated also that legumes will grow on a soil that is acid provided it supplies amply the fertility or stock of plant nutrients needed.
Unwittingly we have carried calcium as a fertilizer along when we were applying its carbonate as the remedy for soil acidity. The same has also happened for our unrecognized benefits in “correcting the acidity of commercial fertilizers.” More fortunately, however, the dolomitic, rather than the pure calcium limestone, has been the “neutralizer for fertilizer acidity.” Consequently, both calcium and magnesium have been coming along with the nitrogen, phosphorus and potassium (N, P, K) in fertilizers, though probably more because dolomite has higher “neutralizing value” than because of the planned desire to add these two extra nutrients for which no additional fertilizer charges are made. This is a case where calcium has been going ahead of–while going along with–the other fertilizers, even though it was not regarded as such in the designing of commercial fertilizers.
The fact that calcium has been the major component of a fertilizer, like superphosphate for example, has reacted to the pronounced benefit of lateritic or red soils, so common in the South. These soils have little absorbing or exchanging capacity. Consequently they do not absorb or retain much hydrogen or acidity. There has been no “war on soil acidity” in the South. There is, however, a serious need for calcium as a fertilizing help in better animal and human nutrition coming from the soils and as a help for those plants which synthesize the proteins and other chemical complexes of higher food values. While tropical soils do not invite the use of limestone in a war on their acidity, they do demand limestone to supply calcium as a fertilizer in its own right.
Calcium plays an important role in the early life of alfalfa when its percentage emergence and early growth are improved on the soil (Plot 12) that was given limestone some years ago. Photo by Missouri Agr. Exp. Station.
Ash analyses of plants leave confusion about the services of calcium. Unfortunately for the services which mineral nutrients coming from the soil render in the plants, the chemical analyses ignite them to destroy their organic combinations. Such procedures measure mainly the percentage of the ash constituents in the dry plant tissue. This gives no concept of the many organic compounds of plant construction into which the calcium, for example, may have been combined, or in the syntheses of which this nutrient plays an important part. It leaves mainly the idea that the plant hauls either a large or a small load of calcium and other minerals from the soil.
Ash analysis is no measure of the functions performed. by the chemical elements. It fails to point out in what period of plant growth the calcium and other minerals are taken from the soil in major or minor amounts. It has not given calcium its decided importance in the very early life of many plants. It has not emphasized the help of calcium in moving other nutrients into the root of the plants, which Professor A. C. True of the University of Pennsylvania has called a “synergistic effect.” It has given undue importance to potassium, for example, as a fertilizer for potatoes because of emphasis mainly on the potato tubers. It has left unemphasized the facts, (a) that the tops of the potato plants carry more calcium than does a legume crop like red clover, and (b) that lime may be necessary as a fertilizer for potatoes more often than commonly surmised. It is such incomplete chemical analyses, with their attention to the ash constituents only rather than to the more complete organic compounds, that have given the confusion about potato scab in relation to soil acidity, instead of its connection with deficient nutrients, even with respect to calcium.
Ash analyses may measure the mineral elements of soil origin, but such measurements fail to mean much when the more efficient function by the plant in synthesizing important compounds is not necessarily dependent on a larger amount of one single plant nutrient taken from the soil. Unfortunately, such chemical measures do not inform us as to what constitutes the properly balanced supply of all nutrients at any time, nor how that balance must be modified for highest plant efficiencies in different periods of growth. It is such ash analyses that have failed to classify calcium as a fertilizer of even greater deficiency on many soils than are phosphorus and potassium. Yet both of these latter two are so common in and emphasized by the mixed goods of commercial soil treatments, the calcium contents of which get no mention on the label or in the sale.
Soil acidity must be removed by fertilizing with calcium, plus other nutrient elements. That soil acidity should have come under condemnation by the recent scientific attacks on it seems strange, now that we look at the pattern of life on the globe and find it most concentrated, not on the neutral and alkaline soils, but rather on the acid soils bordering those that are calcareous. Agriculture has reached its high food output by the help of grazing animals. Grasses as feed for grazing animals are most common and most nutritious on soil developed under moderate annual rainfalls. Grasses grow under a distribution of rainfall which leaves a droughty period of the growing season that spells destruction of the trees but not permanent extinction of the grasses. Such rainfall distributions develop acid soils but do not leach the soils of their adsorbed supply of calcium and other bases. Such rainfalls gave us the prairies that are acid soils but that are also stocked with plenty of calcium, as was the virgin soil of the corn belt. Man and his companion animals for his support have multiplied most rapidly and concentrated themselves most highly, not on soils that are neutral, but rather on those that are acid in their reaction. Yet we have recently gone forward under the belief that soil acidity is highly detrimental and should be completely neutralized. Scarcely have we cherished the thought that soil acidity might even be beneficial, as the locations of higher concentrations of populations and their better agricultural support suggest.
Fertilizers carrying considerable calcium, like superphosphate (above), do not injure the young plants even at high rates of application. But fertilizers without calcium, like ammonium sulphate (below) and others, are injurious at lower rates. Photo by J. Biuhneli, Ohio Agr. Exp. Station.
When soils are a temporary rest stop of rocks and minerals on their way to the sea and to solution under increasing rainfalls encouraging their faster travel in that direction, we must credit carbonic acid as the natural agency that is mainly responsible for the rock and mineral decomposition. When carbonic acid breaks down the rock, another kind of acid, namely, a soil acid must of necessity be the result. Nature has long been using acid reagents to decompose rocks and minerals of which the end products are nourishment for vegetation. But it has only recently been recognized that the soil acids or clay acids in our productive soils are the agencies that are weathering rocks and minerals to release their nutrient contents to the absorbing clay and thereby for exchange to the growing crops. Soil acidity is an integral part of the chemical mechanisms within the soil by which the mineral reserves are broken down to fertilize the crops.
Surely, then, we must recognize in these facts the need for the soil to be acid. At the same time we must recognize the necessity for that soil to contain mineral reserves, among which limestone is the foremost. This must be there to provide calcium for plant nourishment while this stone is being decomposed on contact with the soil acid. In such a concept of the function of soil acidity, its partial removal is a means of fertilizing the soil and crop with available calcium through the service of this acidity in breaking it out of the limestone rock. Viewed in this light, the application of limestone to the soil is a case of applying calcium as a mineral fertilizer for its mobilization from inert rock form to active form on the clay by means of the acidity of the soil. Surely, then, we would not remove from the soil this mobilizing agent and thereby lose its valuable services.
Can we use too much limestone on the soil? Now that the soil acidity is considered a means of decomposing the soil minerals to make their fertilizing contents available to plants, it is evident that a productive soil is one which contains not only hydrogen or acidity, but one which is also supplied with the elements that are nutrients or fertility. Just as our application of limestone is a means of letting the soil acidity mobilize the stone’s contents of calcium, so many other minerals with fertility contents may be applied to the soil and made available by the soil acidity. Rock phosphate is an example. Other minerals may come into use later as fertilizers under this same category. It was from a mixture of such rocks and minerals that soils were originally developed to form the clay residues and to load these with the many nutrients that give our productive soils.
When the plant roots are exchanging their hydrogen or acidity to the clay of the soil for its fertility contents, we may well visualize the fact that they might remove these so completely that the soil becomes infertile and highly acid. Then if we should put limestone on such an acid soil until all of its acidity is removed, would this make it a highly productive soil because it was now neutral, and because that neutrality was obtained by loading it with nothing but calcium, of which the associated carbonate in the limestone had neutralized the acid after the calcium had displaced it from the clay? If a soil is neutral but completely saturated with only one element–even if it be a plant nutrient ever so extensively taken by plants and animals as food–is that soil going to be any more productive than when completely saturated with the non-nutrient hydrogen that makes it very acid? Alkaline soils are not necessarily productive merely because they are not acid. They are often deficient in some elements of fertility, just as acid soils are deficient in some. Thus a very acid soil made completely neutral by liming as the only treatment becomes a hazardous condition in that the acidity, or hydrogen, on coming originally into the soil, displaced too completely many other nutrient elements as well as the calcium. Very acid soils, then, need other fertilizers besides the calcium we provide by applying the limestone.
Calcium has been the foremost deficiency in fertility on extensive soil areas. That limestone has been the most effective fertilizer is indicated by its use to the total amounts of more than ten million tons in the State of Missouri, for example, in less than ten years. The amounts of all other fertilizers used has not been one-fifth that of the limestone during their maximum use. But now that the calcium shortage in the soil has been remedied to strengthen that one weak line, the next item representing the next weakness will need to be strengthened.
On soils limed now for some years, the potassium deficiencies are being indicated more commonly by crop symptoms. Corn stalks are not so strong and may break over. The ears of the corn are less solid or more chaffy where heavy liming is used to grow legumes that are turned under as green manure. Heavy liming and consequent reduction in the degree of soil acidity are bringing on deficiencies of manganese. This deficiency in the crop may possibly reach greater importance as liming is more generously used, and when we recall that perosis of chicks is caused by deficiencies of manganese in their ration. Manganese, commonly considered among the minor nutrients, takes on major importance if its deficiency in the soil can reflect troubles in the animals by way of the crops they consume.
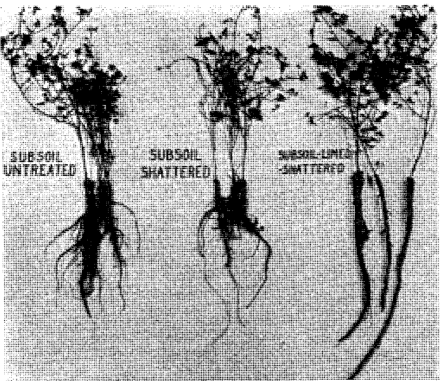
Limestone plowed under helps build up the soil by building down. Limestone put down deeper into the shattered clay subsoil encouraged sweet clover roots to go down into the moist subsoil rather than spread themselves through the shallow, too often dry, surface soil.
Boron and iron, too, are demobilized when the acidity of the soil is much reduced. Alfalfa, root crops, some vegetables and other crops require more boron than may be found available in a soil that has been given heavy dosages of limestone. Whether the excessive carbonate or the excessive calcium is responsible for the trouble has not yet been learned in all these cases. Nevertheless, the trouble is prevented by caution against believing that if a little limestone is good, much more of it will be better. Calcium used alone is not able to offset the shortage of other nutrient elements. Plant nutrition demands calcium in generous amounts for its direct services, and also for many benefits it exercises in mobilizing other nutrients. Nevertheless, soil acidity is also required. It, too, has much mobilizing power for nutrients toward which excessive calcium and neutrality of the soil exercise quite the opposite or a demobilizing effect.
Calcium must lead the parade for other fertilizers. Calcium plays a prominent role in–and is always associated with–protein production by plants. Amino acids, the building blocks of proteins, are not synthesized by animals and humans. They are the synthetic product of plants and micro-organisms but most commonly by those kinds of these producers for which liberal supplies of lime are requisite. Animals and man are always in the struggle for proteins. On the contrary they obtain carbohydrates more easily. On the calcareous soils of the prairies, rather than on the lime-deficient soils cleared of their forests, do we find production of animals less difficult. Animals are grown on the former and fattened on the crops from the latter. By the same token do we have poor reproduction and growth of animals on soils that, through crop removal and neglect, have been depleted of their calcium and the other fertility elements that are associated with it in fertile soils. Lime is first on the list of needs for soils that are to be kept high in animal production.
It was on those soils containing acid properties in the surface horizon, but calcium in their deeper layers, that the bison originally supported himself. It is on these same soils that production of beef and mutton rose to the magnitudes of which we boast. It is on the soils of greater deficiencies in calcium and other fertility constituents where the hog of mainly fat output is the king. Humans, too, may be fitted into the soil picture with more tooth troubles on calcium-deficient soils, as the draftees for the Navy demonstrated. Calcium, which constitutes forty percent of limestone, makes this rock an important fertilizer for the soil in service to man as well as to plants and animals.
Limestone is no longer merely a soil conditioner in terms of the control of the degree of soil acidity. It is the provider of the foremost element for fertilizing effects. It is the mobilizer of other nutrients. It is the catalyzer in the process of synthesizing proteins. It has been delivering these services ever since it was a part of the art of agriculture, dating back some 40 centuries. But, unfortunately, only recently have we realized that calcium is only one of the essential nutrients needed by plants and that many others must come along with it for most efficient crop production. Nevertheless, when animal and human nutrition are taken as the goals of crop production, calcium heads the list of the nutrient elements and stands out as the one demanded in largest amounts from the soil. In terms of human foods for their quality dependent on the soil, limestone is the contributor of calcium and is the synergistic agent for other nutrients; which services make it the foremost of the fertilizers.