Access to all articles, new health classes, discounts in our store, and more!
Histochemistry of the Gingiva VI: Distribution and Localization of Phosphorylase
Published in The Journal of Periodontology, Vol. 32, pp. 338-342, October 1961.
* * *
Among the group of homopolysaccharides, glycogen represents the most important highly polymerized carbohydrate in animal tissue. Its synthesis from glucose-1-phosphate (Cori Ester) is accomplished by the action of two different enzymes, namely phosphorylase and a branching enzyme,4 i.e. the amylo 1,4 → 1,6-transglycosidase7 (Q enzyme6). The polysaccharide formed from glucose-1-phosphate by the enzyme phosphorylase alone resembles amylose in that the glucose residues are arranged in a linear fashion like the amylose chain, their sole linkage being alpha -1,4. Amylo 1,4 → 1,6-transglycosidase is the enzyme responsible for the formation of the branching chains of the glycogen molecule or of amylopectin in such a manner that the alpha 1,6 linkage is formed from an alpha -1,4 link.
This paper deals with the localization and concentration of phosphorylase in human gingivae. The distribution of the enzyme in the gingival tissue could also be regarded as indirect evidence for the distribution of glycogen in the gingival mucosa.
Materials and Methods
Twelve gingival specimens were obtained from patients undergoing blood glucose-tolerance tests. Block anesthesia was used in order to maintain the normal tissue metabolism and a partial gingivectomy of the labial mucosa in the upper right quadrant was performed. The age of the patients varied from 16 to 50 years; sex and race were not taken into consideration.
Experimental Procedure: Immediately upon removal, specimens were cut into blocks of 2 or 3 mm. in thickness, frozen on dry ice and mounted on the cryostat set at -20°C. Sections were then cut 6 to 10 micra thick and mounted on clean slides. After drying, sections were immersed in the incubating mixture of Takeuchi and Kuriaki.9 This consisted of 50 mg. of glucose-1-phosphate, 10 mg. of muscle adenylic acid, 15 ml. distilled water, M/5 acetate buffer at pH 4.8, and 15 units of insulin. Furthermore, about 5 mg. of water-soluble glycogen was added to the mixture to act as a “primer.” The solution was preheated to 37° C for 30 minutes. Sections were then incubated for 10, 20, 40 and 50 minutes. After incubation the sections were rinsed in a large volume of dilute Lugol’s iodine solution (10 ml. Lugol’s solution, U.S.P. in 190 ml. distilled water) (I2:K1:H2O; 1:2:400) for five minutes. Finally, the sections were mounted with glychrogel alone or glychrogel containing a few drops of concentrated Lugol’s solution. The sites of phosphorylase activity appeared blue black.
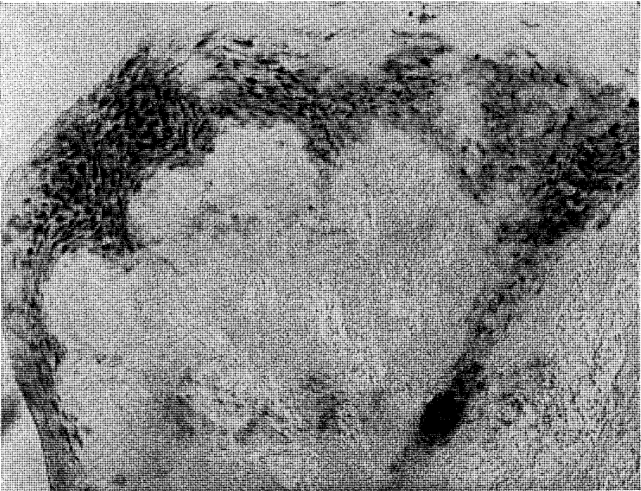
By means of this histochemical method two different staining reactions may be seen in the gingival mucosa. One, stained dark blue by iodine, represents the new polysaccharide synthesized from glucose molecules by phosphorylase. The other, stained light brown, demonstrates the pre-existing glycogen in the tissue (Figure 1). This difference in color thus helped to distinguish between the newly formed polysaccharide and the native glycogen present in the gingiva.
Fig. 1. Irregular localization of phosphorylase in dark black, while the native glycogen appears in dark grey. Note a small reactive parakeratotic pearl at the bottom of the picture.
Control Procedure: Three different controls were utilized. In one, the sections were immersed in the incubating solution without the substrate, glucose-1-phosphate. The second control consisted of placing sections directly into dilute Lugol’s solution (5 ml. Lugol’s solution, U.S.P.; 95 ml. distilled water) for the demonstration of native glycogen. For the third control, rat tongues were employed since striated muscle contains large amounts of phosphorylase.
Results
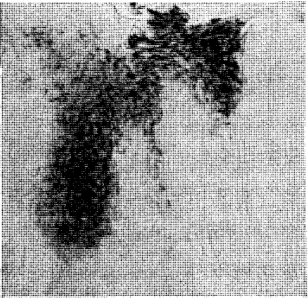
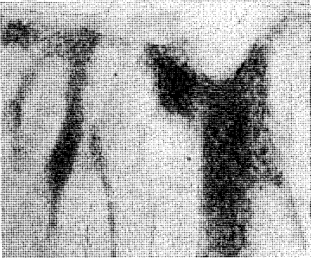
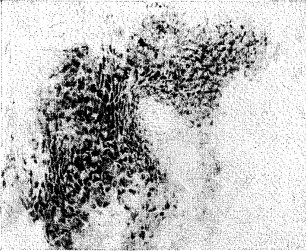
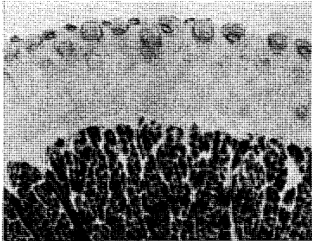
The distribution and localization of phosphorylase in the gingiva appears in selective points of the epithelial layer (Figure 2). Some rete pegs show a definite enzymatic reactivity while others, even though immediately adjacent, are completely devoid of the enzyme. In all cases, phosphorylase was distributed in the stratum spinosum and often in the stratum granulosum. However, none was demonstrated in the stratum germinativum. The keratotic layer did not react toward the enzyme (Figure 3). The crevicular epithelium was often strongly reactive (Figure 4) whereas the epithelium of the attached gingiva was, in most instances, unreactive. In cases in which the degree of inflammation was rather severe, the enzymatic activity appeared after 10 minutes of incubation. Conversely, in tissues with a mild inflammatory reaction the intensity of the enzymatic reactivity was moderate. Interestingly, the enzyme was always present where some interstitial edema or acanthotic phenomena occurred. In fact, sections with structurally normal stratified squamous epithelium did not store any amount of phosphorylase. The iodine-treated sections of the control substantiate these observations in that no native glycogen could be detected. The underlying connective tissue, the inflammatory cells and the blood vessels (arterioles) were all negative. From a cytologic point of view, only the cytoplasm was reactive with the nuclei remaining unstained.
Fig. 2. Selective deposition of the enzyme in the prickle cell layer and the parakeratotic horny layer. Note the absence of phosphorylase in the adjacent rete pegs, in the stratum basale and the keratotic layer.
Fig. 3. Another aspect of the enzyme deposition. Note the large unreactive zone at the top of the picture where hyperkeratosis is present.
Fig. 4. Crevicular epithelium. Distribution of phosphorylase in the cells of the stratum spinosum.
Altogether different was the localization of phosphorylase in the tongue of rats (Figure 5). While the epithelium and the connective tissue appeared to be completely devoid of enzyme, the muscular fibers were strikingly reactive thus demonstrating the highest concentration of phosphorylase.
Fig. 5. Rat tongue. Intense enzyme reaction in the muscle bundles while the epithelium and the underlying connective tissue are devoid of phosphorylase.
Discussion
The distribution of phosphorylase can be considered as indirect evidence of histochemically demonstrable carbohydrates in tissues which normally synthesize polysaccharides of the amylose or glycogen type. In the gingival mucosa, the enzyme distribution appears selectively involving certain cells of the stratum spinosum and granulosum. Sometimes the parakeratotic layer shows positive reactivity. However, the localization of the enzyme is not uniform. Some rete pegs are highly reactive while others are entirely devoid of phosphorylase.
The findings do not demonstrate the presence of the enzyme in the endothelium of capillaries as claimed by Braun-Falco1 in his studies on the skin. Also, no evidence is derived in this study of positive reactivity in the epithelium of the tongue of rats as reported by Takeuchi, Higashi, and Eatanuki10 in their original paper. To substantiate the latter findings, further experiments demonstrated that tongue sections stained by the PAS method failed to show the presence of glycogen in the epithelium. However, it might be possible, as asserted by Carbonell,2 that the histochemical technique utilized may not be sensitive enough to demonstrate all the glycogen present in the tissue.
During the past few years various investigators have histochemically studied the problem of glycogen deposition in normal and pathologic gingivae. The different opinions expressed by several authors have been recently summarized by Carranza and Cabrini.3
One of the constant findings is the reverse relationship between the rate of keratinization and glycogen distribution in the cells. Hyperkeratinized areas are most often deprived of glycogen. In the skin, Montagna, Chase, and Lobitz, W. C.8 believe that, under physiologic conditions, glycogen is found where the process of keratinization is impaired and that no glycogen occurs in cells undergoing rapid keratinization and high mitotic activity. In human gingivae, Weiss, Weinmann, and Meyer11 have also demonstrated that glycogen appears most frequently in inflamed, unkeratinized epithelia while hyperkeratinized gingivae contain little or no polysaccharide. It has been often reported that glycogen is formed in squamous epithelium affected by spongiosis or inflammatory edema. As regards the gingival mucosa, Dewar5 linked the presence of glycogen with the rate of inflammation.
Although it seems established that the breakdown of glycogen provides energy for cell activity, its functional role upon stratified squamous epithelia remains a question still open to debate and by no means defined.
The Takeuchi-Kuriaki technique for the demonstration of tissue phosphorylase is substantive and reproducible. Since physiologically occurring glycogen in living tissues is a branching polysaccharide and it is stained red-brown by the iodine method, it appears that the newly-formed polysaccharide in the gingiva is one of a relatively low molecular size because of its blue-black color. Since no special inhibitor (HgCl2) was used for the 1,4 → 1,6 transglycosidase, the branching enzyme responsible for the complete synthesis of glycogen, it is believed that the dark color in both the gingival specimens and rat tongue represents a polysaccharide of the amylose type (amylopectin or slightly branched glycogen).
Summary
Phosphorylase, an enzyme essential for the synthesis of glycogen from glucose-1-phosphate, has been demonstrated in human gingivae. The enzyme was revealed utilizing the iodine method of Takeuchi and Kuriaki. Phosphorylase appeared localized at sites known to contain glycogen normally. The cell cytoplasm reacted positively while nuclei were always devoid of enzyme activity. The gingival mucosa showed some variability. However, phosphorylase was generally localized in the stratum spinosum and often in the parakeratotic horny layer. The stratum basale was always negative. Furthermore, the underlying connective tissue structures never gave rise to a positive reactivity.
The tongue mucosa of rats, utilized as controls, demonstrated a striking reactivity in the muscular fibers while the epithelial layers, in contrast to the findings in human gingivae, were entirely devoid of phosphorylase.
References Cited:
- Braun-Falco, O. “Zur histopographie der phosphorylase bei basaliom und psoriasis.” Arch. Klin. u. Exptl. Dermatol. 204:175, 1957.
- Carbonell, L. M. “Phosphorylase and the conductive system of the heart.” Jour. Histochem. and Cytochem. 3:419, 1955.
- Carranza, F. A., Jr. and Cabrini, R. L. “Histochemical reactions of periodontal tissues: A review of the literature.” Jour. Amer. Dent. Assoc. 60:464, 1960.
- Cori, G. T. and Cori, C. F. “Crystalline muscle phosphorylase. IV. Formation of glycogen.” Jour. Biol. Chem. 151:57, 1943.
- Dewar, M. R. “Observations on the composition and metabolism of normal and inflamed gingiva.” Jour. Periodont. 26:29, 1955.
- Haworth, W. N., Peat, S. and Bourne, E. J. “Synthesis of amylopectin.” Nature 154:236, 1944.
- Larner, J. “The action of branching enzymes on outer chains of glycogen.” Jour. Biol. Chem. 202: 491, 1953.
- Montagna, W., Chase, H. B. and Lobitz, W. C. “Histology and cytochemistry of human skin. II. The distribution of glycogen in the epidermis, hair follicles, sebaceous glands and eccrine sweat glands.” Anat. Rec. 114:231, 1952.
- Takeuchi, T. and Kuriaki, H. “Histochemical detection of phosphorylase in normal tissues.” Jour. Histochem. and Cytochem. 3:153, 1955.
- Takeuchi, T., Higashi, K. and Eatanuki, S. “Distribution of amylophosphorylase in various tissues of human and mammalian organs.” Jour. Histochem. and Cytochem. 3:485, 1955.
- Weiss, M. D., Weinmann, J. P. and Meyer, J. “Degree of keratinization and glycogen content in the uninflamed and inflamed gingiva and alveolar mucosa.” Jour. Periodont. 30:208, 1959.