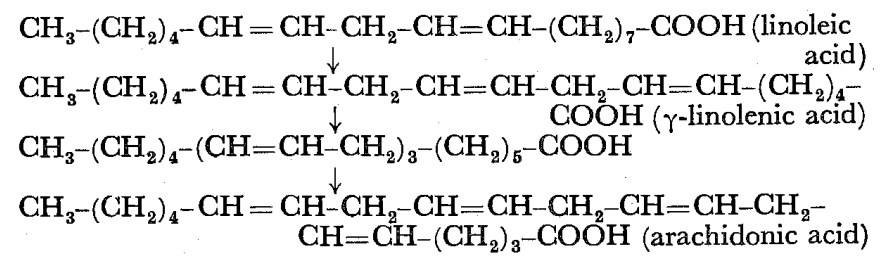Access to all articles, new health classes, discounts in our store, and more!
Essential Fatty Acids in Nutrition and Their Relation to Other Vitamins
Published in Lectures on the Scientific Basis of Medicine, Vol. 6, pp. 260-276, 1956-57.
* * *
Nature of Essential Fatty Acids
The need for fat in the diet of animals was not demonstrated until Evans and Burr (1928) showed that a diet of purified casein and sucrose, supplemented with salts and yeast and cod-liver oil, produced subnormal growth in rats. Various workers have now studied the syndrome of deficiency of essential fatty acids (EFA) in at least seven species of animal including monkeys, and there are strong reasons for believing that they are needed by man.
The most important EFA in the diet is linoleic acid or cis-9:10, cis-12:13-octadecadienoic acid; this has one more double bond than has oleic acid and two more than stearic acid, all of which have 18 carbon atoms. Linoleic acid is converted in the body to γ-linolenic acid by the insertion of a third double bond but this acid does not occur in appreciable amounts in foodstuffs. Then two carbons are added to γ-linolenic acid and a fourth double bond is inserted giving arachidonic acid which is all-cis-5:6, 8:9, 11:12, 14:15-eicosatetraenoic acid. These steps which have been elucidated by Mead and Howton (1957) are as follows:
It seems that vitamin B6 (pyridoxin) is required for the conversion of linoleic to arachidonic acid (Witten and Holman, 1952).
There are certain chemical points that are important. Obviously a large number of isomers of linoleic acid can exist, either positional isomers in which the double bonds are shifted along the molecule to different positions or geometrical isomers in which the hydrogens on one or both double bonds are trans (giving cis-trans, trans-cis or trans-trans isomers–hereafter referred to collectively as trans). Unfortunately the trans isomers are more stable than the natural cis-cis isomer. It will be noticed that the double bonds are methylene-interrupted and not in the more stable conjugated position (-CH=CH-CH=CH-), and that in each of the four compounds shown there is no double bond until the sixth carbon from the methyl end is reached; if a double bond occurs in this part (as in the ordinary form of linolenic acid) the compound has no EFA-activity and this is important since many highly unsaturated (or polyethenoid) fatty acids present in fish oils have a double bond in the 3:4 position counting from the methyl end.
The presence of double bonds makes EFA easily oxidized if oxygen is present, and traces of metals catalyse this oxidation with production of hydroperoxides which are themselves strong oxidizing agents. Reduction also occurs fairly easily under appropriate conditions with production of stearic acid; nickel is used for catalysing this hydrogenation in the manufacture of margarine, and if hydrogenation is not complete trans and positional isomers of linoleic acid are formed. So unstable are the EFA in presence of oxygen that they would not exist for long in the food or body were it not for the presence of antioxidants, the most important of which are the tocopherols (vitamins E). These are distributed in plant sources roughly parallel with the distribution of linoleic acid. They hinder destruction of EFA in the food, in the gut and in the body. There appears to be some synthesis of EFA in the gut by bacteria and in the body; when we speak of arachidonic acid and its precursors as being ‘essential fatty acids’ we mean that one or other or combinations must be supplied in the diet since they cannot be synthesized in the body in adequate amount. Unfortunately bacteria also destroy EFA, and this occurs in the gut of ruminants such as cows and sheep, where bacteria convert cis-cis-linoleic acid from ingested plant fodder into trans isomers which are then stored in the body-fat and excreted in milk.
Sources of EFA
The richest foodstuffs are the vegetable seed oils, more than half of the fatty acids of some of them consisting of linoleic acid. For instance, the following are the approximate percentages of linoleic acid in the fatty acids of certain typical seed oils: walnut, 73; safflower, 73; sunflower, 60; corn, 55; soybean, 50; cotton, 45; arachis, 26; wheat, 25; olive, 15; coconut, 2. Most of these oils are also rich in tocopherols, and it is possible that the content of different tocopherols affects the ability of oils to lower serum cholesterol in man. The tocopherols in corn oil consist of about 89 per cent of the γ- and 11 per cent of 𝛼-tocopherol. The former is the more powerful antioxidant and therefore the better protects linoleic acid in the oil from oxidation in the air or in the gut; but it is itself hardly absorbed from the gut and therefore, unlike the 𝛼-form, is not effective in the body. The combination of a large proportion of the very active unabsorbable form with a small proportion of the less active easily absorbed form is probably excellent. Unfortunately we eat very little unprocessed vegetable seed oils in this country. Refined olive oil is not a good source of EFA, and the large quantities of vegetable seed oils that are imported are used for the manufacture of margarine. Margarines vary considerably in their content of EFA: it seems that in general French margarine is rich, British margarine less rich and American margarine poor. Part of the vegetable oil may be totally hydrogenated and part not hydrogenated at all, in which case the resulting margarine will be rich in EFA. Alternatively, all the vegetable seed oil may be partially hydrogenated, in which case most of the cis-cis-linoleic acid may be lost since the nickel catalyst will isomerize it before hydrogenation occurs. But British margarine is probably richer in EFA than is butter since, as already indicated, almost all the linoleic acid in this is trans isomers.
The presence of these isomers may be important. The requirement of EFA in the diet depends upon various factors, one of the most important of which is the proportion of EFA to other fatty acids. Saturated fat, such as stearic acid, increases the requirement and there is some evidence that isomers of linoleic acid compete with the ordinary form in the body and so tend to produce deficiency on marginal diets. This is a matter of great importance about which we have very little information. I mentioned it after we had done some work upon the presence of isomers in foodstuffs (Sinclair, 1956a), but we stopped our work on this when the elegant and extensive work of J. B. Brown appeared. He has shown (Mabrouk and Brown, 1956; Sreenivasan and Brown, 1956) that typical margarines may contain about 40 per cent of trans acids and that about half the linoleic acid may be conjugated (and therefore not effective as EFA).
Wheat-germ oil is rich in linoleic acid and in tocopherols, and more of the former than one might expect is found in white flour since the starch adsorbs linoleic acid. However, as I have recently pointed out elsewhere (Sinclair, 1957c, d), it is known from the work of various authors that most of the tocopherols is destroyed in flour by the bleaching ‘improvers’ such as chlorine dioxide, and there is some evidence that this destroys and chlorinates some of the linoleic acid though whether this is significant is not at present known. We have to consider both the composition of the fat in foodstuffs and the amount of that fat ingested, and unfortunately we have not adequate analyses at present: the fat of potatoes is about 50 per cent linoleic acid but there is so little fat present that this is not important.
We are even more ignorant about the contribution of animal fats, and their composition is changing in some instances. Some fish oils are very rich in EFA; but some marine oils, such as cod-liver oil, are extremely deficient and contain polyethenoid acids that are antagonistic: cod-liver oil is therefore toxic for EFA-deficient rats or mice. Eggs from hens free-ranging and therefore ingesting linoleic acid may contain considerable quantities of linoleic and arachidonic acids; but if the hens are kept in batteries and fed on poultry meal deficient in linoleic acid, the eggs will also be deficient. Lard used to be rich even in arachidonic acid when pigs ranging freely were fed on scraps and their fat was sold by a butcher; modern pigs, kept in Danish houses, are fed on processed meal and a premium is given for ‘hard’ fat (which means saturated), and then the fat is processed so that it will have a long shelf-life in a grocer’s shop. Little is quantitatively known about the effect of cooking on the EFA in foods, but frying undoubtedly causes losses. Therefore as we process our foods more, we tend to decrease the content of EFA and increase that of their isomers and of saturated fat. Further, a diet excessively high in carbohydrate will give rise in the body to relatively saturated fat which will also increase the requirement of EFA in the diet. Conversely, on very low calorie diets the body will burn EFA ingested and will mobilize the relatively saturated fat from the body stores so that EFA-deficiency will tend to occur. And there are other factors that affect the requirement: it is increased in hypothyroidism and particularly in diabetes mellitus; in the latter case the explanation may be that EFA are swept into the general pool of fat that is rapidly being metabolized.
These dietary considerations have not been adequately considered by those who have concluded that the relative EFA content of our diets has not decreased over the years (Hollingsworth, Vaughan and Warnock, 1956; McCann and Trulson, 1957; Yudkin, 1957). My own studies have led to the opposite conclusion and will be presented elsewhere: I believe that during the past forty years there has been an increasing relative deficiency of EFA in our diets, although there have been fluctuations in certain years. The epidemiology of deficiency of EFA in different countries of the world will be mentioned presently.
Function of Essential Fatty Acids in Lower Animals
The main functions appear to be structural and with certain qualifications all the functions at present known can be explained in these terms. EFA appear to be needed for the formation of cell membranes (including the myelin sheath of nerves), for mitochondrial membranes and the cristae mitochondriales, and for the formation of connective tissue. In cell membranes, EFA esterify cholesterol and form part of phospholipids, and then in combination with protein form lipoprotein. It appears that EFA are needed for proper metabolism of cholesterol and of phospholipids, and the need for essential fatty acids for these purposes may be regarded as part of their structural function. Rather different, however, is the possible need for the esterification of vitamin D which will be mentioned later.
There is as yet no direct evidence that EFA are needed for mitochondrial membranes, but unsaturated fats occur in them. Uncoupling of oxidative phosphorylation is found in deficiency of EFA (Klein and Johnson, 1954) and this might be caused by the defective structure of mitochondria. Ramalingaswami and I found (1953) that there was defective structure of cartilage and surrounding mesenchymal ground substance in the ear of the rat deficient in EFA, and there seems to be a general defect in connective tissue throughout the body. The possible relationship of this to disorders of the skin and of bone I have discussed elsewhere (1957b, e). The greatly increased permeability of the skin to water (Sinclair, 1952; Basnayake and Sinclair, 1954, 1955) is explicable in terms of the structural defect, and the greatly increased fragility and permeability of capillaries found in deficiency of EFA (Kramár and Levine, 1953) probably occurs through a defect in the connective tissue that supports the endothelial cells of the capillaries. Proteinuria and haematuria are sometimes found in such animals and appear to be caused by similar defects in the glomeruli of kidney.
Function of Essential Fatty Acids in Man
Hansen and his colleagues (1957) have studied extensively deficiency of EFA in children, particularly in relation to infantile eczema. They have found that in certain cases of this condition the iodine value of serum lipids is decreased as compared with normal infants and that treatment with fats rich in EFA cures some cases of eczema. A good deal of supporting and also conflicting evidence has been produced in various quarters, and I have recently reviewed it elsewhere (1957b). The balance of evidence is in favour of EFA causing at least some cases of eczema in infants.
Several years ago we considered that some cases of follicular hyperkeratosis in man might be caused by deficiency of EFA and we therefore carefully studied the morphology of the lesion of the skin produced by this deficiency in rats (Ramalingaswami and Sinclair, 1953). Our preliminary studies of certain cases of this condition and of Darier’s disease and of pityriasis rubra pilaris have provided some support for this suggestion, though extensive clinical trials require to be done.
The possible relation of EFA to certain diseases in man, which I have suggested speculatively elsewhere (Sinclair, 1956a), can be most conveniently considered in the following sections.
Atheroma
Apart from the liver, there are seven tissues of the body in which cholesterol may be deposited and all of which have in common the anatomical peculiarity of not being supplied directly with capillaries or drained by lymphatics. These ‘avascular’ tissues are: epithelium, endothelium, cornea (both epithelium and endothelium), lens, tendon, cartilage, the granulosa cells of the ovary and the cells of the seminiferous tubules in the testis. In all these tissues deposits of cholesterol may occur, for instance, in diabetes mellitus or idiopathic hypercholesterolaemia. These tissues (or their appendages such as nails and hair) together with teeth comprise the ‘avascular’ tissues of the body. In lower animals such as the rat it is not easy to study the deposition of cholesterol and phospholipid in the intima of the aorta; but in the belief that the process might be similar in the epidermis, we have studied this. Both the intima and the epidermis are known to be able to synthesize cholesterol and to esterify it. We found (Basnayake and Sinclair, 1955) that the rat fed a diet totally deficient in fat (and therefore containing no EFA) deposited large amounts of cholesterol and phospholipid in its epidermis and that these were abnormal in composition in that the iodine number of the fatty acids was abnormally low. The probable explanation for the deposition is that under normal conditions these compounds are formed with EFA and are then easily combined with protein to form lipoprotein and metabolized in the body. If more saturated fatty acids are used such as the fat that can be synthesized in the body, cholesterol esters of quite different physical properties are formed: they have a higher melting point and are less soluble in organic solvents. Similarly, cholesterol esterified with the ordinary form of linoleic acid is more soluble and probably more easily metabolized in the body than that formed with conjugated linoleic acid. By analogy with this deposition of cholesterol and phospholipid in the epidermis, it seemed possible that the accumulation in other tissues might occur through the same mechanism (atheroma, arcus senilis, cataract, xanthoma tendinosum). This local synthesis and accumulation within the cells of the intima, however, would not account for the patchy distribution of atheroma, though it would account for the earliest lesion being found within the cells and not extracellularly, as might be expected if the cholesterol diffused from serum. As mentioned above, there is increased permeability of capillaries in deficiency of EFA, and this might allow diffusion from plasma to occur predominantly in areas where blood pressure is raised or where there is local trauma (as for instance near branches from the aorta).
The greatly increased requirement of EFA in diabetes mellitus has already been mentioned and the deposition of cholesterol in this condition is perhaps therefore not surprising. It is tempting to explain the three other main complications of diabetes mellitus on a similar basis. Lower animals deficient in EFA sometimes develop nephropathy for a reason that has already been mentioned and the defect in the connective tissue supporting the endothelial cells of venules might be responsible for the aneurysmal dilatations that occur in the retina in diabetic retinitis. The nervous system is very rich in EFA but under normal conditions these are avidly retained when other tissues become depleted. In long-standing deficiency, however, or in very severe deficiency in lower animals, depletion of the nervous system can occur and diabetic neuropathy might be caused in this way.
One interesting fact about EFA is that the requirement for male animals is much greater than that of females during the reproductive period; the ratio is about seven. In some way not understood this seems to be related to the sex hormones. There is also a possible relation of EFA with corticosteroids, as will be mentioned presently, and it may be that steroid hormones become esterified with EFA as do cholesterol and possibly vitamin D. It seems that oestrogens, unlike androgens, increase the excretion in the bile of cholesterol and its breakdown products such as cholic acid. Herein may lie the origin of gallstones which are prevalent in EFA-deficient lower animals. Gallstones occur in the more highly civilized countries, in women rather than men during the reproductive period, and in women who are obese and therefore more likely to be deficient in EFA. In rats deficient in EFA there is desquamation of the epithelium of the gall bladder and thereby a nidus is provided for the formation of a stone. The same may be true of man, and therefore there might be an increasing tendency for stones in women up to the menopause, since oestrogens may increase the concentration of cholesterol in the gall bladder and therefore the tendency for stone to form on the desquamated epithelium.
In a very different category are stones of the urinary bladder which are prevalent in undernourished boys in certain Eastern countries (Passmore, 1953). Here we may have EFA-deficiency causing desquamation of the epithelium of the bladder and so providing a nidus; the prevalence in males might arise from the increased EFA-requirement of males.
Coronary Thrombosis
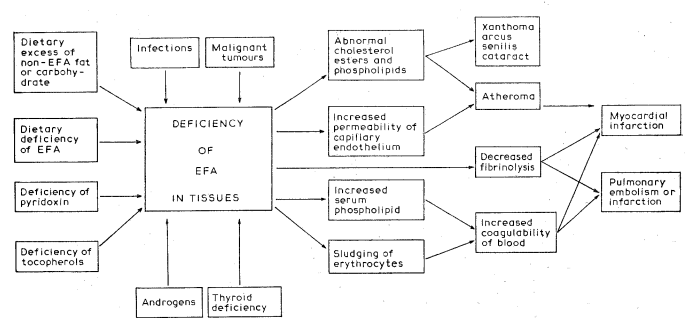
I have attempted elsewhere (Sinclair, 1956a, 1957a) to put forward a unified hypothesis for coronary thrombosis on the basis of EFA-deficiency. Since then, this has been produced experimentally in rats (Hartroft and Thomas, 1957). I have supposed as have others that atheroma and increased coagulability of the blood are required for myocardial infarction (Figure I). Atheroma we have already considered; and it is interesting, as I shall show in detail elsewhere, that deaths from myocardial infarction are increasing at about the same rate as are those ascribed to pulmonary embolism or infarction. This apparent increase in coagulability of the blood of the population might be related to increasing relative deficiency of EFA by two factors. First, Robinson and Poole (1956) have shown that phosphatidyl ethanolamine appears to be increased in plasma after a fatty meal and that it increases the coagulability of blood. This type of phospholipid–the cephalin type–characteristically contains more highly unsaturated fatty acids than the lecithin type. When Basnayake and I found (1955) that phospholipid accumulated in the epidermis in EFA-deficiency and subsequently that the iodine number of the fatty acid in it was abnormally low, we considered that the accumulation was caused by failure to metabolize the abnormal compound. Phospholipid is increased in plasma in myocardial infarction, and the origin of the increase might also be through failure to metabolize adequately an abnormal phospholipid that nevertheless has undiminished coagulating power. EFA are also required for fibrinolysis.
Fig. 1. Diagram of aetiological factors in myocardial infarction.
The second fact of interest is that several years ago Ramalingaswami and I noticed when measuring the blood-pressure of rats deficient in EFA or pyridoxin that, when the circulation in the limb was slowed by a cuff and the capillaries in the paw were observed, there was marked sludging of the erythrocytes in either deficiency but not in the controls. This is similar to the phenomenon observed by Knisely (1951) in man, and might be expected to favour coagulation of blood.
Various authors, particularly Kinseil et al. (1957), Ahrens et al. (1957), Malmros (Malmros and Wigand, 1957), and Bronte-Stewart (1957), have shown that essential fatty acids or highly unsaturated fatty acids lower serum cholesterol in man. For reasons indicated above I am more interested in the fundamental process or processes concerned with deposition of cholesterol and phospholipid in ‘avascular’ tissues than with the level of serum cholesterol: the rat deficient in EFA accumulates cholesterol and phospholipid in its epidermis although the serum cholesterol is abnormally low. Many persons develop coronary thrombosis with normal serum cholesterol: in the series published by James et al. (1957) there was no difference between those who had had a myocardial infarction and the controls. Conversely many persons are walking the streets with serum cholesterol levels fantastically elevated. The work of Kinsell and others lends strong support to the thesis that EFA or polyethenoid fatty acids are required for the normal metabolism of cholesterol. Our own work on the deposition of cholesterol suggests that this is a function of EFA in particular rather than of polyethenoid acids in general, though this is not proved: EFA will remove the accumulated cholesterol and phospholipid from an EFA-deficient rat or mouse, but an attempt to do so with cod-liver oil (rich in polyethenoid acids but deficient in EFA) caused death of the animals.
Keys on the other hand maintained that all fats were equal in their cholesterol-raising effects on plasma, though his more recent experiences with corn oil are in accord with those obtained earlier by other workers and cannot be reconciled with his own earlier results (see Kinsell and Sinclair, 1957). He has maintained that deaths from myocardial infarction in different countries run parallel with total fat in the dietary (1953), but only six countries were selected and some of the obvious misfits such as France were not included. Yerushalmy and Hilleboe (1957) have correctly discounted his views. I have used official figures to correlate deaths from myocardial infarction with assessed relative deficiency of EFA in dietaries, and there is a reasonable correlation as will be presented elsewhere.
Idiopathic Hypercalcaemia of Infants
This condition was first described by Lightwood in 1952 and since then many cases of the severe and mild types have been reported. The infants, almost invariably fed on dried cow’s milk with added vitamin D, develop athrepsia, anorexia, vomiting, polydipsia, hypertension with a systolic murmur over the heart, patchy osteosclerosis, mental retardation and nephrocalcinosis; in serum, calcium, urea, cholesterol (mainly esterified), and vitamin A are increased. Bonham Carter (Bonham Carter et al., 1955) suggested that the systolic murmur was associated with hypertension and atheroma which accompanies this condition in some of its phases. Some cases follow infection.
It seems to be generally agreed that the condition is caused by increased sensitivity to vitamin D, but this cannot explain all the features. Elsewhere (Sinclair, 1956b, c) I have put forward a possible explanation supported by some experimental evidence on lower animals. Cow’s milk is known to be very deficient in ordinary linoleic acid as compared with human milk, but it is rich in calcium. When it is dried the small amount of EFA in it is unstable in presence of air probably because metal ions, unhydrated, catalyse the oxidation of polyethenoid acids with formation first of hydroperoxides and later of aldehydes which cause it to smell and taste rancid. An infant fed on National Dried Milk may therefore be on a diet rich in calcium and vitamin D but low in EFA. The deficiency of EFA could cause the rise in serum cholesterol (mainly esterified) and the alleged atheroma on lines already discussed. Vitamin D has an esterifiable 3-hydroxyl group chemically very similar to that in cholesterol. By analogy, vitamin D might therefore normally be esterified in the body with EFA and its concentration, like that of esterified cholesterol, might be increased if it were esterified with unusual fatty acids in presence of a relative deficiency of EFA; but it might still exert its calcium-retaining effect in the body. In this way deficiency of EFA could cause hypersensitivity to vitamin D: corn oil, a rich source of ordinary linoleic acid, should decrease the potency of vitamin D whereas cow’s milk, a very poor source of ordinary linoleic acid, should increase the potency, and this is in fact the case (Lewis, 1935). The failure to grow, the polydipsia and the mental retardation might all be directly attributable to deficiency of EFA. Infection increases the requirement of EFA and so would tend to precipitate deficiency. An attempt might be made as follows to explain the increase in plasma vitamin A. This exists in plasma mainly as alcohol and is stored in liver mainly as ester, but no information seems available about the fatty acids with which vitamin A is normally esterified. If these were EFA it is possible that in deficiency storage would be hindered and the concentration of the alcohol in plasma increased. To test this hypothesis of increased sensitivity to vitamin D in deficiency of EFA, I administered a non-toxic daily dose of the vitamin to EFA-deficient and control rats. The former rapidly lost weight and died with uraemia caused by nephrocalcinosis, whereas the control animals were unaffected, even when given two times that dose of vitamin D. There is therefore increased sensitivity to vitamin D in presence of deficiency of EFA and this deficiency in children may be enhanced if cod-liver oil is also administered. It is interesting that the disease is very rare in the U.S.A. where either evaporated milks (in which EFA are more stable) or ‘filled’ dried milks are often used, the latter being skim milk to which vegetable seed oil–rich in linoleic acid–is added.
Infants with idiopathic hypercalcaemia often show patchy fragmentation of the internal elastic lamina of smaller arteries with occasional impregnation with calcium salts. As will now be discussed, deficiency of EFA causes faulty connective tissue and this pathological change might be a direct result of such deficiency. It is tempting to suppose that this might be relevant to the apparent increasing incidence of dissecting aneurysm of the aorta and that Monckeberg’s sclerosis might be caused by deficiency of EFA with a relatively high intake of calcium and vitamin D.
Structural Defects as A Basis of Disease
Lower animals chronically deficient in EFA may appear on superficial examination to be fairly normal, yet they react violently to insults or stresses. For instance, application to the back of one paw of a mild dose of ultra-violet light that would produce no obvious lesion in a control rat may cause so violent a reaction in an EFA-deficient rat that there is necrosis of much of the limb. Ultra-violet light destroys EFA, and the permeability of the epidermis to water, already excessive in deficiency of EFA, is further increased by irradiation with UVL. The dermatitis following UVL may be caused by destruction of EFA in the superficial layers of the epidermis with production of a product such as a hydroperoxide that causes a chain reaction of destruction of EFA and produces the erythema and necrosis. The relation of EFA to detergent dermatitis and nickel dermatitis have been discussed elsewhere (Sinclair, 1957b). The violent reactions to insults are similar to the responses found in the so-called collagen diseases of man, or diseases of mesenchymal ground substance. The lower animal deficient in EFA or pyridoxin may get duodenal ulcers, ulcerative colitis, nephrosis with haematuria, easy rupture of capillaries, arthritis, osteoporosis and dental caries. Both duodenal ulcers, now a disease of middle-aged men, and senile osteoporosis are increasing rapidly in this country and dental caries is notoriously a disease of the more highly civilized countries where processed foods are eaten. The altered structure of epithelium in the EFA-deficient animal allows substances to penetrate more easily into the body, and infections are common. This may be relevant to allergic conditions such as asthma in man in which foreign proteins penetrate the mucosa; the more easy passage of viruses through the nasal or intestinal mucosa might be of significance in the occurrence of coryza and the increasing prevalence of poliomyelitis in more highly civilized countries. Defective mitochondrial structure might explain the preliminary evidence I have obtained that rats deficient in EFA are more prone to develop carcinoma of the stomach when fed methylcholanthrene than are controls. If there is an increased sensitivity to administered chemical carcinogens in EFA-deficiency in man, one might expect carcinoma of stomach, bladder and lung (assuming inhalation of a carcinogen) to be more common in males since their requirement of EFA is greater than that of females. This possibility of deficiency of EFA playing a part in the cause of bronchial carcinoma might explain the prevalence of atheroma in this condition and also the nervous system changes which may also accompany carcinoma of the stomach. The nervous system tends to retain EFA even when other tissues have become depleted, but there is a tendency for depletion to occur. As Macmillan and I have recently reported (1957), implantation of a tumour in an EFA-deficient animal precipitates severe deficiency. The cause of carcinomatous neuropathy might therefore be as follows. EFA-deficiency tends to increase sensitivity to a chemical carcinogen and to deplete the nervous system of EFA; if a carcinoma is produced, it will quickly increase the severity of the deficiency and so precipitate lesions in the nervous system, perhaps in peripheral nerves, perhaps in the cerebellum or perhaps in the cortex.
Conclusion
The scientific basis of medicine rests upon the application of the experimental method to man and lower animals, though results obtained upon these may be irrelevant to clinical science. But the observational method and the epidemiological approach can on occasion help. There are many diseases, mainly chronic degenerative diseases, that are found predominantly in the more highly civilized countries, such as coronary thrombosis, duodenal ulcers, dental caries, bronchial asthma and senile osteoporosis. Many such diseases are becoming rapidly more prevalent in these countries, and this is not explicable in terms of the increasing expectation of life; indeed, the expectation of life of a middle-aged man in this country is only four years greater now than it was a century ago despite the tremendous achievements of medical science, and this is because the degenerative diseases are increasing so fast. It is tempting to seek a unified hypothesis for this phenomenon, and twenty years ago I became interested in deficiency of essential fatty acids as a possible cause. The evidence fits together, though the gaps in it are obvious and very wide. One would prefer to try to fill those gaps before speculating unduly, for the scientific basis of medicine must rest upon verifiable facts; but facilities are needed that I do not possess and I therefore commend to others the study of deficiency of essential fatty acids in man as a problem of great and accelerating urgency as our foods become increasingly processed.
References Cited:
- Ahrens, E. H. Jun., Insull, W. Jun., Blomstrand, R., Hirsch, J., Tsaltas, T. T. and Peterson, M. L. (1957). Lancet, i, 943.
- Basnayake, V. and Sinclair, H. M. (1954). J. Physiol. 126, 55 p.
- Basnayake, V. and Sinclair, H. M. (1955). Proc. II Int. Conf. Biochem. Probl. Lipids (Ghent), 476-484.
- Bonham-Carter, R. E., Dent, C. E., Fowler, D. I. and Harper, C. M. (1955). Arch. Dis. Childh. 30, 399.
- Bronte-Stewart, B. (1957). Nutr. 11, 60.
- Evans, H. M. and Burr, G. O. (1928). Proc. Soc. Exp. Biol. N.Y. 25, 390.
- Hansen, A. E., Adam, D. J. D., Boelsche, A. N., Haggard, M. E. and Wiese, H. F. (1957). Proc. IV Int. Conf. Biochem. Probl. Lipids (Oxford). In press.
- Hartroft, W. S. and Thomas, W. A. (1957). J. Amer. Med. Ass. 164, 1899.
- Hollingsworth, D. F., Vaughan, M. C. and Warnock, G. M. (1956). Proc. Nutr. Soc. 15, xvii.
- James, A. T., Lovelock, J. E., Webb, J. and Trotter, W. R. (1957). Lancet, i, 705.
- Keys, A. (1953). J. Mt. Sinai Hosp. 20, 134.
- Kinsell, L. W., Michaels, G. D. and Dailey, J. (1957). Proc. IV Int. Conj. Biochem. Probl. Lipids (Oxford). In press.
- Kinsell, L. W. and Sinclair, H. M. (1957). Lancet, i, 883.
- Klein, P. D. and Johnson, R. M. (1954). J. Biol. Chem. 211, 103.
- Knisely, M. H. (1951). Postgrad. Med. 10, 15.
- Kramár, J. and Levine, V. E. (1953). J. Nutr. 50, 149.
- Lewis, J. M. (1935). J. Pediatr. 6, 362.
- Lightwood, R. (1952). Proc. Roy. Soc. Med. 45, 401.
- Mabrouk, A. F. and Brown, J. B. (1956). J. Amer. Oil Chem. Soc. 33, 98.
- McCann, M. B. and Trulson, M. F. (1957). J. Amer. Diet. Ass. 33, 358.
- Macmillan, A. L. and Sinclair, H. M. (1957). Proc. IV Int. Conf. Biochem. Probl. Lipids (Oxford). In press.
- Malmros, H. and Wigand, G. (1957). Lancet, ii, 1.
- Mead, J. F. and Howton, D. R. (1957). Proc. IV Int. Conf. Biochem. Probl. Lipids (Oxford). In press.
- Passmore, R. (1953). Lancet, i, 638.
- Ramalingaswami, V. and Sinclair, H. M. (1953). Brit. J. Derm. 65, 1.
- Robinson, D. S. and Poole, J. C. F. (1956). Quart. J. Exp. Physiol. 41, 36.
- Sinclair, H. M. (1952). Biochem. Soc. Symp. No. 9, 80.
- Sinclair, H. M. (1956a). Lancet, i, 381.
- Sinclair, H. M. (1956b). Lancet, ii, 101.
- Sinclair, H. M. (1956c). Lancet, ii, 893.
- Sinclair, H. M. (1957a). Proc. III Int. Conf. Biochem. Probl. Lipids (Brussels), 392-400.
- Sinclair, H. M. (1957b). Ann. Nutr. (Paris), 11, A147.
- Sinclair, H. M. (1957c). Roy. Soc. Hlth. J. 77, 234.
- Sinclair, H. M. (1957d). Proc. Nutr. Soc. In press.
- Sinclair, H. M. (1957e). Proc. IV Jorn. Diet. Madrid, 179.
- Sreenivasan, B. and Brown, J. B. (1956). J. Amer. Oil Chem. Soc. 33, 521.
- Witten, P. W. and Holman, R. T. (1952). Arch. Biochem. 41, 266.
- Yerushalmy, J. and Hilleboe, H. E. (1957). N.Y. State J. Med. 57, 2343.
- Yudkin, J. (1957). Lancet, ii, 155.