Access to all articles, new health classes, discounts in our store, and more!
Electrolytic Medication (Physiological and Dental Aspects)
Published in The Journal of the National Dental Association, 1918, pp. 601-28. Co-Author: Samuel E. Pond.
* * *
From the Laboratories of the Research Institute of the National Dental Association, Cleveland, Ohio.
During the past year S. C. Brooks and the writers, working in the Research Institute laboratories in Cleveland, have attempted to throw light upon some very important questions which have arisen in the practice of the so called “Electrolytic Medication.” The purpose of this and succeeding papers is to study the action of electrolytes and electric current upon bacteria and body tissues under conditions simulating the treatment of infected oral tissues in the human body. It is hoped in this way to aid the dental profession to clear away some misconceptions regarding “Electrolytic Medication” and to more readily establish such a therapeutic method upon a firm basis.
The treatment of various diseases and diseased areas by medicaments carried into the tissues thru the aid of a galvanic current has been known, in dentistry, as “Cataphoresis” (W. J. Morton), “Ionic Medication” (Jones), “Electrolization” (Rhein), “Electro-Sterilization” (Prinz), “Electrolytic Medication” (Fette), and “Iontophoresis” (Koller). There has been no uniform choice of any one of these terms. Indeed, to select one descriptive of the method is somewhat arbitrary, depending largely upon the conception of what takes place. Since the diffusion of ions in a mean direction (not specifically toward the cathode) is considered the important factor, and not always to effect sterilization but, as well, the stimulation tending toward the regeneration and repair of periapical and other tissues, we may suggest that the term “Electrolytic Medication” appears to be satisfactory and the most applicable of those which have been used. Fette has previously indicated1 that the term “Electrolytic Medication” is preferable to “Ionic Medication” or “Ionization” as it implies a method of treatment with an electrolyte and that electrolysis (not alone “Ionization”) is one of the associated phenomena. The term may not be wholly applicable but it appears to more nearly approach a descriptive one and be less confusing than the others referred to. We shall use, in this paper, electrolytic medication synonymously with the phrase electro-chemical method, and discriminate between the names of associated phenomena, e. g. dissociation, electrolysis, polarization, etc.
The use in dentistry of galvanic current and externally applied medicaments was suggested in 1858 by B. W. Richardson. In 1887 Dr. McGraw demonstrated, at a clinic in Chicago, some apparatus in this connection. In 1895 Gillett, one of us (Price), Van Woert, and others, perfected apparatus and studied what was then known as “Cataphoresis.” L. P. Bethel, W. J. Morton, M. L. Rhein, E. Sturridge, and others, treated pyorrhea and attempted from 1896 on to correct various septic conditions with the aid of this agent. In 1903 Hoffendahl, after working with Miller in Germany, recommended that electricity be used without externally applied electrolytes. Since then, in this country and abroad, considerable prominence has been given to the use of electricity with and without externally applied electrolytes to cure and correct certain abnormal or.pathological conditions, i. e., to sterilize infected tissues and to stimulate and lead to the replacement of tissue destroyed as a result of infection.
An electro-chemical method has been advocated for various reasons. One of the chief of these is apparently the aim to reach deep seated infections with different medicaments which, it has been assumed, could be driven into the tissues to “seats of infection” by an easily controlled reagent, electricity. Some of these medicaments have disinfecting properties but, what is essential to note here, the combination of electrolytes and electric current has been definitely credited by Prinz,2 and perhaps others, with having sterilized infected areas (presumably by direct action upon the bacteria). Some articles on this subject have, also, given the writers of this paper the impression that electrolytic medication has led to the stimulation of the body tissues to repair destroyed portions.
The usual treatments with electrolytic medication vary considerably. They involve the use of a galvanic current with an approximate range of one-tenth milliampere to five milliamperes, over a period of from five to forty-five minutes. Electrodes of copper, zinc, platinum, silver and iron have been used with either anode or cathode in the tooth. An “indifferent” (larger) electrode serving as the opposite pole is placed by some operators on the gum near the tooth to be treated, by others on the cheek, back of the neck, on the forehead, or on either wrist. The electrolytes which have been recommended and used are copper chloride, copper sulfate, magnesium chloride, potassium iodide, sodium chloride, silver nitrate, zinc chloride, zinc sulfate. and possibly others. Iodin in alcohol and in water has also been recommended in connection with electrolytic medication but the halogen is not an electrolyte, i. e., does not conduct electricity to any appreciable extent. Studies to determine the effect of differences of potential, current density, the value of one electrode over another, or the superiority of an electrolyte for a specific purpose have not been prosecuted extensively enough to satisfy the writers. This fact along with the variety of technic, the great differences of opinion among the profession and some conflicting clinical data which we have secured, has led us to undertake the present work. The clinical data included the culturing of the fluid from the periapical area in a large number of cases, including immediate aspirations, one hour after, and one and several days after, electrolytic medication, and in each and every case we obtained vital cultures. These clinical tests also included the extracting of a tooth and the culturing of its root apex and the periapical tissues after the treatment had been given for thirty minutes at 4 ma. The current was again applied to this tooth, after extraction, for one hour at a higher current strength in a normal salt and the apex still gave vital cultures.
A study of the literature on this subject has involved the construction of a bibliography, based mainly upon the review of twenty different dental journals (cf. “Bibliography of Electrolytic Medication”). The oldest of these journals have been reviewed since 1881 and the rest since their beginning. In many cases the indexing has been found to be so poorly done that volume after volume has had to be scanned serially. The result has been the review of the development of the method, the gaining of a quite definite idea as regards the present standing of electrolytic medication and the recognition of the complexity of the problem. Besides the dental literature we have frequently referred to the medical journals, and those of the more exact sciences, especially physical chemistry, for recent contributions to the subject and original investigations. From this review we have been impressed with the need of an analysis of the whole problem and have, consequently, conducted the experimental work and considered the theoretical aspect under the following four divisions:
- Direct Effects
- Toxicity of electrolytes, to answer the questions (a) What concentration of electrolytes, acting for a unit time, are toxic so that they cause irreversible changes, i. e., death of bacteria? (b) In these concentrations for the same unit of time, is there destruction of human tissue beyond the limit of body tolerance and power of regeneration?
- Effect of galvanic current upon living tissues, to answer (a) Will galvanic currents kill pure or mixed cultures of bacteria and what is such an irreversible change a function of (i. e. time, current-density, electromotive force, etc.)? (b) What is the effect upon human tissues of a galvanic current acting under conditions which will kill bacteria in that locus?
- Electro-chemical effects, to throw light upon (a) What effects are produced when electrolytes are used in connection with galvanic current upon bacteria and upon human tissues under conditions which would kill bacteria and under conditions simulating the present practice with regard to electro-sterilization of root canals and periapical tissues? (b) What is the depth to which the ions penetrate under a fall of potential which is used in connection with electrolytic medication, and what is the penetration a function of?
- Indirect Effects
- Tissue repair factors, to answer (a) What are the essential factors in the repair of periapical tissues? (b) Can electric current, or electrolytes, or both, change dying cells to recuperating or living cells?
General Considerations
Before proceeding to the presentation of the experimental data regarding the action of electrolytes and electric current on tissues, the writers believe that it is necessary to consider some general and related questions.
It has been intended in this paper to point out the value of quantitative: determination and care in the recording of experimental results. To aid in making this clear, the analysis of the problem includes largely the determination of certain quantitative factors with which we are especially concerned. To put this view in other words, we believe that it is not sufficient to know what changes take place, but also how they take place and how one factor is related to another, in a given reaction. The medical profession has already realized the necessity for quantitative methods in research. Recently, for example, studies on the “rate of cicatrization” have been reduced to mathematical expression by Carrel, DuNouy, and others.3 The subject of immunity, or immunization, probably better known to the dental profession, is being developed more rapidly since the quantitative studies of Madsen and Jorgensen on the blood’s content of agglutinin and Levin’s studies on the concentration of diphtheria antitoxin in goat’s blood.4 In dentistry Prinz5 has proposed, altho with unreliable data, an expression to calculate the time of current application in “Electro-Sterilization.” It is essential, we wish to point out, that this need of quantitative expressions be more fully realized in dentistry.
In this connection Arrhenius4 wrote:
“As long as only qualitative methods are used in a branch of science, this cannot rise to a higher stage than the descriptive one.”
His point may be taken as implying that medicine today consists of countless descriptions of known diseases, observations of the treatments, of drugs used, and the clinical results, and to form an opinion of the manner in which these substances react it is essential to utilize the methods introduced by modern physical chemistry. Descriptive and qualitative chemistry are of very limited value. Physical and theoretical chemistry allow us to follow quantitatively the influence of temperature and of foreign substances upon the interesting organic products; which are of greatest importance in medicine, in the physiological processes of daily life and in the diseases and their therapy. Biological chemistry and general physiology in applying the physico-chemical methods have paved the way to a clearer understanding of the vital processes upon which is based the advance in medicine. The literature reviewed for this study indicates that the dental profession has not realized the significance of care in the recording of results and of quantitative measurements.
There have been a few notable exceptions to the last statement. We would refer here to the studies in metallurgy, cocain anesthesia, teeth stains, and, recently, the studies with relation to bacteria and the internal secretions. With reference to the problem of electrolytic medication, the early observations and conclusions from studies in cataphoresis 1895-1900, regarding the chemical and physical factors, have, however, passed unimproved and untested in spite of the fact that investigations in medicine and physiology destroy, to a great extent, the value of these old results and must change, as will be later seen, our concept with regard to such phenomena.
In the main the investigators of the question of “Electro-Sterilization” and those who have striven to perfect technic in the application of such a method have neglected, furthermore, to appreciate fully the significance of characteristic differences between living and non-living systems. A knowledge of electricity, deemed important by Sturridge6 and many others,7 and even a knowledge of electro-chemistry, is not sufficient. Electrical response in living tissue, the role of electrolytes, polarization phenomena, the significance of the semi-permeability of membranes, surface-tension, etc., have not been considered sufficiently in the past when drawing conclusions as to the efficacy of such an application of electro-chemical phenomena as an “Electrolytic Medication.” In 1900 Cook, A. Pont, and one of us (Price), discussed the significance of certain of these physiological questions. No further consideration of such matters has been prominent in the dental literature.
With living matter, we must not overlook the fact that we are dealing with two types of reactions, (known in chemistry as the “reversible” and the “irreversible”). The characteristic of the living state is that reactions are not in stable equilibrium and are largely of the reversible type. For example, we are not dealing with reactions as
H2 + ½ O2 = H2O or NaOH + HCl = NaCl + H2O
but more of the type
C2H5OH + CH3COOH ↔ CH3COOC2 H5 + H2O
(the last known as “esterification” and “saponification”). The reactions in the body further, have been considered, by some investigators, as similar to the change in gelatine from solution to jelly and the reverse (sol ↔ gel). In the body we presume that changes occur constantly in one or the other direction, hence we speak of them as reversible. In a large measure the effect of electrolytes and electric current upon the living cells leads to the type of reversible reactions (such as alterations in the semi-permeability of membranes, temporary changes of the hydrogen-ion concentration, narcosis, or anesthesia, and the like). In physical and organic systems an increase in the tensity of such reagents or a longer reaction time will often lead to irreversible changes; resulting, in the body, in the destruction or death of the tissue. This statement applies to bacteria as well as to body cells.
It will be worth while to point out some of the reactions of this type with which we are concerned in this connection. Constant electric current, e. g. galvanic electricity, passing thru the living organism, alters many physiological processes. Where the current enters the tissue (in the region of the anode) there is decreased irritability, depression, or inhibition (“anelectrotonus”); where the current leaves the tissue (the region about the cathode) there is excitation or heightened irritability (“catelectrotonus”).
“The essential basis of the effect appears to be an altered polarization of the cell surface” (Lillie).8
Reversible changes in the semi-permeable membranes and other secondary effects are known to accompany this polarization effect in many cases. This is attributed by Nernst to the accumulation of the ions of the dissolved electrolytes of the cell at the membrane, under a constant fall of potential.
As regards the actions of ions and molecules of electrolytes in contact with the living cell there are many characteristic effects of the reversible type. Magnesium sulfate is known to produce anesthesia. Similar effects are produced by potassium salts. Calcium and strontium salts cause reversible desensitization in nerve and muscle (Lillie, above). Calcium, also, antagonizes the stimulating action of pure sodium salts, hence its use in Ringer’s and Locke’s solutions.
Considerable significance is attached to the effect of the neutral salts upon the colloids of the cell, especially of the surface layer or plasma membrane (probably a solution effect or surface tension change). Czapek, Osterhout, Macallum, Lillie, Hober, Overton, and others, have studied the effects of electrolytes and, as well, other chemicals upon the surface membranes. Some of the effects were found to accompany an increase or decrease of the surface tension and are closely connected with the electrical potential difference existing between colloidal (or other cell particles) and the medium by which the cell is bathed. The significance of this surface effect, of alterations of the semipermeability of the plasma membranes, is important: Lillie considers that “a surface effect is sufficient to set in motion the whole complex apparatus of response in the cell-interior.”
Whatever effects have been produced during electrolytic medication are apparently, we believe, of the reversible type. In other words, electrolytes or electric current in low concentrations or for brief periods of time may intensify or suppress certain functions. Presumably, however, increase, either in the intensity of the reagent or the amount of reaction time, should lead to irreversible destructive changes. Our experiments indicate this irreversibility in reactions with bacteria has not been secured. The reversibility or irreversibility, it must be noted, is the same in character with living cells of whatever type or function, i. e. bacteria, epithelial, bone, or other cells, differing only in degree, due probably to different rates of metabolism, or to constitution, characteristic of the individual cell.
- Direct Effects
1. Action of Electrolytes upon Bacteria and Other Organisms.
Studies of electrolyte action on bacteria have been made necessary because investigations in the past have not taken into account (1) exact quantities of the reagents, (2) the time over which the reaction was allowed to take place, or (3) the conditions were not comparable, with those which we meet in electrolytic medication. For instance, many of the investigators working with bacteria “in vitro” allowed the electrolytes to act for twenty-four hours. It is essential for our purpose to know what direct action there is in every minute, or at least after five, fifteen and thirty minutes.
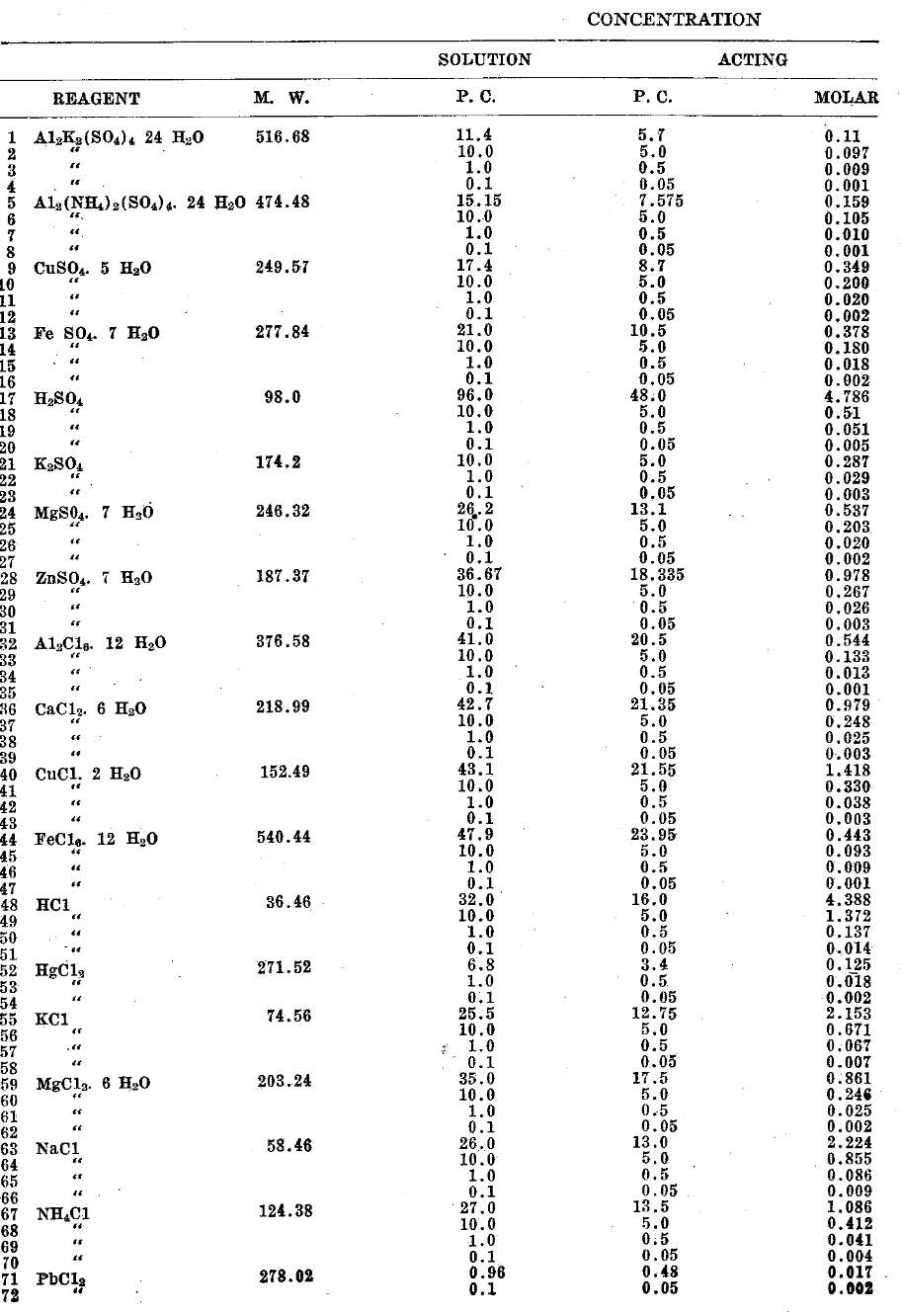
For this purpose, a series of electrolytes (chlorides and sulfates of eleven different elements) were selected and used in different concentrations for different periods of time.
Saturated and ten per centa solutions were made up at twenty-five degrees centigrade. One per cent and one-tenth per cent solutions were made up from the latter by diluting a pipetted sample (i. e. ten cc and one cc respectively) to one hundred cubic centimeters at the same temperature.b
The procedure was to use one-half cubic centimeter of each salt or acid solution and an equal quantity of an active culture of mixed oral bacteria in dextrose agar broth. (Hence, there was a total concentration of one-half that made up.)
Certain experimental errors, it is hoped, were avoided in this series. Every precaution was taken in the series to prevent contamination of the salt and acid solutions as we were attempting to study the direct action of pure electrolytes. The culture of bacteria was grown fresh and was used within twenty-four hours. A freshly extracted infected tooth was ground up in a mortar and mixed with a sterilized dextrose agar broth, about one quarter of a liter. In twelve hours, at thirty-seven and one-half degrees centigrade, an active culture was observed in every case. The mixture of electrolyte and bacteria was placed in small test tubes and kept throughout the reaction time at constant temperature, thirty-seven and one-half degrees centigrade. After five minutes had elapsed a sample on a flamed platinum loop, or tungsten-gold wire, was transferred to each of four tubes of sterile dextrose agar broth. This was also done after fifteen, thirty, sixty and one hundred and twenty minutes. Controls with the solvent of the electrolyte, in this case water, were used in every case. Observations of the incubated samples were made after twenty-four and forty-eight hours, and in some cases four days. If, in this incubation time, a tube was still clear it was called negative. (See Table II). Controls in every case indicated (cloudy tube) that the growth of bacteria was active and alive throughout the longest reaction period of over two hours, under the same conditions as the electrolyte-bacteria reaction was proceeding.
Table I–Action of Electrolytes on Bacteria in Mass.
Table II
The results are significant. The following decreasing order of toxicity was recorded for neutral salts: Mercury > Iron > Copper > Aluminum
With the acids which were possessed of toxic properties, concentrated hydrochloric was slightly greater in effect than concentrated sulfuric. In the case of mercuric chloride of all concentrations used, i. e. saturated and as low as 0.002M, all the bacteria in the mixture were killed during five minutes. A saturated solution of ferric chloride was slightly effective after fifteen minutes and reached a maximum in about thirty minutes. No dilutions of the salt were effective. A saturated solution of copper chloride required one hour before it could be said to have a disinfecting action, and a saturated solution of copper sulfate required two hours. The saturated solution of aluminum chloride inhibited but did not prevent growth throughout two hours. Concentrated hydrochloric acid was effective as a disinfectant within five minutes, but ten per cent and one per cent solutions required one hour. The bacteria withstood the action of concentrated sulfuric acid for more than five minutes but were killed in something less than fifteen. Ten per cent solutions required two hours before they were effective. Lower concentrations of the reagents than those mentioned had no perceptible effect. There was no disinfecting, or even antiseptic action, with the following cations: Ammonium; calcium; ferrous iron; lead; magnesium; potassium; sodium and zinc. The anions chloride and sulfate exhibited no toxic properties. (In regard to the terms antiseptic and disinfectant we follow the accepted use in bacteriology. The term antiseptic is taken to mean either neutralization of toxin or to inhibit bacterial growth, while disinfectant means to kill or cytolyze the bacteria and stop growth, permanently, of bacteria present.)
We are thus led to conclude with regard to the electrolytes in their direct action upon bacteria that, to disinfect or sterilize chemically, it would be necessary in the case of mixed cultures of bacteria to use concentrations which would be prohibitive in connection with electrolytic medication. It would not seem wise to attempt to use about the teeth the concentrated hydrochloric or sulfuric acid which would be necessary to kill any large quantity of bacteria in a short period of time. Mercuric chloride is not advisable to use except externally, unless it can be reduced by the body to HgCl (mercurous chloride or “calomel”). The time and concentration necessary to disinfect with ferric chloride or copper sulfate, for this reason, makes these electrolytes almost prohibitive. This statement of the case applies under the conditions which the bacteria were killed in these experiments. It is obviously impossible in practice to bring about the total concentration with which these results were obtained in these tests. The diffusion of a solution from the pulp chamber to the periapical or other tissues surrounding the teeth would require considerably longer time even with the electric current, and then it is doubtful if the distribution could be anywhere nearly as effective or uniform as in our experiments.
Referring to the work of others for comparison Kronig and Paul9 found that the number of colonies of bacteria depended upon the time during which the toxic reagent acted and upon the concentration of the solutions. They found that the more dilute the solution the less was the action. They used pure strains of bacillus anthracis and staphylococcus pyogenes aureus distributed on garnets to insure uniform amounts of the flora. The only other results known to us which are comparable are those of Sternberg, who used five cubic centimeters of reagent and five cubic centimeters of a bacterial culture in media, allowing the action, in every case, to proceed for two hours. (Other investigators allowed the reaction to proceed for twenty-four hours and most of them introduced only an amount of bacteria which could be carried on a platinum loop into relatively large quantities of electrolyte solutions, thus making it difficult to compare results.) Sternberg found that the minimum concentration of five reagents required to kill bacteria was as follows:10
Boer11 found that in two hours it required HCl in the following concentrations to kill:
Kitasoto12 found HNO3 required as follows:
KOH was found to require
NaOH Kitasoto found to be the same as KOH.
While it is somewhat out of place in this discussion it will be well to mention here the action of chlorin as a disinfectant. Prinz13 writes:
“The ionization of a one per cent sodium chloride solution by means of the galvanic current within a root canal furnished free chlorin in ‘status nascendi’ in the presence of moisture and body temperature which is most active, and admirably suited for our purposes. The free chlorin sterilizes the walls of the root canal–and bleaches the discolored dentin. Incidentally, the ionization of salt water furnishes an appreciable amount of hydrochloric acid which acts as a superficial solvent of tooth structure, and thereby enlarges the root canal.”
From this quotation we might understand that the chloride ion is a disinfectant because of the author’s use of the word “ionization.” We believe, however, after careful inspection, that Prinz does not mean ionization, but electrolysis. Regarding the action of electrolytic products we will speak later under the head of electro-chemical action upon bacteria but we will here make clear the difference between the action of ionic (charged molecular or atomic) chlorin and gaseous (electrically neutral) chlorin. There is danger of confusion in the use of the terms, by authors in the dental literature.
The chloride ion as dissociated from any neutral salt or hydrochloric acid in water solution (that is, ionic chlorin) is not a disinfectant. Our experiments, for example, referred to above (where we used highly dissociable chlorides) exhibited no toxic reaction with bacteria. In all cases (in one series) the chloride ion (anion) was present and in suitable concentration to have disinfected, had the ion possessed that property. This result is in agreement with the findings of many investigators. The chloride ion (likewise the sulfate or nitrate ion) is non-toxic. Plant and animal cells will live for days, in mixture of neutral salts (Ringer’s or Locke’s solution) where the anion is chloride (also true of sulfate or nitrate).
On the other hand, chlorin in gaseous or molecular (electrically neutral or non ionic) form is a disinfectant. It has an affinity for hydrogen and releases nascent oxygen. It must act, however, in the presence of moisture, as Prinz indicated. (loc. cit.) We may illustrate here that anthrax spores, which had been previously dried and exposed to 44.7 per cent dried chlorin were not destroyed. Moistened spores were killed in the same time with four per cent chlorin.10
We do not understand how “ionization” of a sodium chloride solution will liberate chlorin in such a form that it will disinfect. It must be understood that “nascent” chlorin (indicated by Prinz) refers to the gas and not. to the ion, and ionization does not produce the former, but the latter. It is the former upon which the action of the hypochlorite solution, suggested by Prinz, depends for its disinfecting property, and we wish to point out here that this action is not direct but in reality by a secondary or indirect reaction. The Dakin-Daufresne hypochlorite solution is slightly soluble in water and yields Ca(++), Cl(-) and (ClO)(-) ions. In the presence of catalizers the hypochlorite solution would decompose as follows:
hypochlorite
2 CaCl (OCl) → 2 CaCl2+O2
The germicidal action of the hypochlorite solution is dependent, according to Dakin, upon the formation of a chloramine group (NCl)14 which is strongly bactericidal and non-irritating to animal cells. With regard to the employment of electrically decomposed chlorin from sodium chloride solution to sterilize root canals, we shall discuss later.
One may conclude from these statements regarding the direct action of electrolytes in common use that, as used in the experiments cited when acting alone, i. e. without electric current, are either (1) not effective in killing bacteria or (2) are effective (a) only after considerable time or (b) in short periods of time, in concentrations which are not easily used in dental practice. The several electrolytes, the effects of which have been thus studied, are those most used in electrolytic medication or accessible for use. Other electrolytes which may be effective in usable concentrations, i. e. so as not to injure body cells beyond repair, and to kill bacteria in periods of time of two hours or less, have not been studied within the knowledge of the writers. It appears to us, then, that if, with the aid of electrolytic medication, up to the present, certain infections have been checked, infected areas sterilized, or repair instituted as a result of the action of electrolytes or their ions, it is not a direct independent effect of electrolytes upon the bacteria.
Galvanic Current and Living Tissues
The action of direct current upon bacteria has not been studied carefully to any appreciable extent. Becquerel, Davey, Fabre, Paleprat, Guardana, d’ Hahn, Hassenstein, Mangani, Priestly, Sigard and de la Fond investigated the action of galvanic current upon tissues between 1870 and 1875. Cohn and Mendelsohn in 1883 reported some studies of the effects of low galvanic currents on bacteria in nutrient solution, and this work was continued by Prochowink and Spaeth in 1890. Apostoli and Laquerriers in 1890 tried currents of 100-150 milamperes in nutrient medium and reported them efficient in killing bacteria, attributing the results to electrolytic products. Versoogen reached the same conclusion in 1891 after similar experimental work. Zierler and Lehmann (Wurzburg) in 1900 gave a detailed account of the action of galvanic current on bacterial infection of root canals. None of the evidence from these investigators is as conclusive in telling us about the action of the current as is that from the researches of Loeb, Maxwell, Jennings, Kiihne, Nernst and others working to give a physico-chemical explanation of the phenomena involved. The results of older investigators were conflicting; sometimes indicating that electric current would kill bacteria, at other times it would not. The physico-chemical studies have given us very definite ideas as to how the current acts and have established the fact that it may stimulate, depress, or even cytolyze tissue cells. We now know that the ions move under the influence of the electric current in the living cells much as they do in a battery–going to the opposite faces of the membranes–the physiological “poles” of the cells acting like electrodes. In this manner we would suppose that the ions in the medium surrounding the cells can be accumulated at the membranes. We have thus an effect not unlike polarization in a battery. We now know also that other particles suspended in liquids move with the current. Colloidal particles and even bacteria may be carried in a definite direction by such means (cataphoresis).
In the case of the body with which we are especially concerned, it conducts electricity like a liquid “in which,” as Loeb says, “the current is carried by the dissociated ions or such colloidal particles as possess an electric charge. The concentration of the colloidal particles is very small compared with that of the electrolytes in solution, so that the conduction of the latter one is mainly or practically exclusively responsible.”15
Two effects are produced by the constant current in living tissue–one following any rapid change in the intensity of the current, the other “throughout the whole duration of the current and consists in an increase of irritability at the cathode and a decrease at the anode.”
The work done by the current in passing thru a liquid conductor “consists, first, in the pulling of the ions thru the liquid to the electrode and, second, in the withdrawal of the charges from the ions and the transformation of the latter into uncharged atoms at the electrodes.”
Loeb and others believe that the latter is unimportant: Köller16 attempts to make use of the transference of the charge from the ion to the tissue, considering that the charge now given up to the tissue cell (not to the electrode) is the principal factor. On this question there is no evidence known to the writers. It seems from the evidence which is at hand that the physiological actions of electric current are due almost wholly to the increase of the concentrations of ions at the electrodes. Now, it is especially significant that the membranes may serve as electrodes, in many cases where the radicals or ions are precipitated in their uncharged form unlike the action at the poles of a battery where polarization phenomena occur.
Two other effects are recognized in connection with the passage of electric current thru living tissues. One of these is the rise in temperature. In 189817 one of us (Price) considered that the rise in temperature was important and calculated this rise in degrees centigrade, per second. Without considering opposing forces such as radiation, the increase was calculated as 4.04 degrees centigrade per second. This was in the tissue in the apical foramen. The effects of temperature are, of course, important in living phenomena, heat rigor in muscle beginning at 42 degrees centigrade, and probably some type of coagulation changes taking place in other tissues. The action of heat is reversible up to a certain point but if the temperature is changed suddenly, or maintained for any length of time beyond the normal range, it is irreversible. The temperature to kill bacteria, however, is considerably beyond what the body will tolerate, requiring a temperature above 50 degrees centigrade for some time. In some cases, with spores, a temperature of 120 degrees centigrade must be maintained for thirty minutes. No such temperature would ever be reached in the body without destruction of vital cells. In this connection we have found in a series of tests that, if the temperature were maintained constant and below 37.5 degrees, the passage of direct current of 100 volts thru a culture of bacteria for fifteen minutes is of no effect. Without preventing the rise in temperature the same current inhibited the growth in one minute and disinfected it in five minutes. This current is, of course, impossible of attainment with a patient for any length of time, not even an instant.
There is also the effect of very weak currents in living cells. In the case of Actinospherium (a single celled animal used to demonstrate this electrical effect), Kühne reports thus:
“When subjected for some time to constant current It begins to disintegrate on the anode side. This is a purely electrolytic effect, not one which could be considered stimulating.”
Phenomena of secretion are also produced by the constant current which, in the case of Amblystoma (a salamander) reported by Loeb was shown to be a direct effect on the gland cells of the body surface.
“When a current is sent thru a trough filled with water which contains an Amblystoma, a secretion of whitish mucus appears on the skin wherever the outside of the latter Is struck by the current waves emanating from the anode.”
If paramecia (another single celled foam) are subjected to constant current “the effect is such that the position of the cilia on the side of the cathode is altered while on the side of the anode the cilia retain their normal position, provided the current is not too strong.”
This effect is produced mechanically, and chemically as well. Effects of this kind are presumed to occur with bacteria, and like effects probably take place in the body during infection.
We believe from this that there is no evidence to justify the assumption that galvanic current can sterilize infected tissues in the body by killing bacteria directly, without serious destruction of body cells. The current would act as we have seen by precipitating the ions at the bacterial membranes if it would act at all and thus, by changing the electrical and chemical condition, lead to cytolysis. But this same action is going on with the body tissue cells. There appears in the case of the body, as we know, a characteristic regulation and change during the passage of perceptible weak galvanic currents, (such as are used in electrolytic medication). The cells presumably accommodate themselves to the movement of the ions while the current is flowing. They are affected only by a sudden increase or decrease in the intensity of the current (sudden accumulation of ions). As we have found no evidence to the contrary and in our experiments bacteria were not killed during the passage of constant current, we presume the bacteria, likewise, take care of the polarization.
Electro-Chemical Effects
In possible contrast to the action of electrolytes and constant electric current, independent of each other, we shall now be concerned with their combined (electro-chemical) action. The impression of most of those who use electrolytic medication seems to be (1) that electric current will drive ions into tissues which they could not otherwise penetrate, (2) that the electric current dissociates (“ionizes”) the electrolytes in the solution, or the electrode in the tooth (root canal), or both, and (3) that there is an electrolytic action in the tissues. This impression has been favored by experiments of Prinz, Sturridge, Jones, Finzi, and others, in the following way:
Experiments by Finzi18 are reported by Jones19 and Sturridge20 where potassium ferricyanide ions were carried into the tissues of “cats, rabbits, a monkey and a dog,” by electric current. Without the current it is reported that the ions had not penetrated. Leduc is reported by Sturridge21 as having used colored ions and demonstrated that they penetrated into the body. Permanganate of potash ions were employed on both arms where the electrodes were in contact with the flesh. At the positive pole no staining took place while at the opposite pole a brown punctate rash appeared. Sturridge22 demonstrated the movement of copper and iron ions into coagulated albumen, noting the movement of green (cupric) ions and blue (Turnbull’s, not “Prussian” blue,) indicating ferrous ions. Prinz has introduced two experiments to indicate the migration of chlorin. In reality he has demonstrated the movement of the hydrogen ion. In one case a tube of an alkaline gelatine solution, “containing 1% of sodium chloride, colored pink with phenolphthalein,” has a platinum electrode in each end. (This is an adaptation of Sir Oliver Lodge’s experiment to show the velocity of the migration of ions.)
With the passage of an electric current hydrogen ions migrate from the anode toward the cathode and hence make the solution in the region of the anode colorless. This change of color is due to the transformation of the phenolphthalein. In alkaline solutions a sodium salt of phenolphthalein is formed which dissociates into the cation sodium and a colored complex organic anion. The presence of hydrogen ions forms undissociated (colorless) molecules, a quantity of which leads thus to the change of the color of solution. (This cannot be attributed to chlorin). In a similar experiment Prinz has the anode passing thru the foramen of a tooth. As before, the change of color is due to the migration of the hydrogen ions from the anode. Jones, further, in an experiment, has demonstrated that ions will penetrate bibulous paper to a considerable depth.
The experiments cited are unsatisfactory because (a) definite concentrations of reagents are not reported, (b) the destruction of body tissue cells and the disinfection of bacteria are not taken into account, and (c) the conclusions do not apply directly to the conditions as met with in electrolytic medication. In other words, these experiments do not demonstrate that the ions employed are toxic to bacteria in the concentrations used nor that they will sterilize infected tooth or periapical tissue-the objectives of electrolytic medication. Hence, we believe that it is essential to consider the electro-chemical effects from a different angle than heretofore.
It is surprising that, in discussions regarding electro-chemical actions accompanying electrolytic medication, the structure and functions of the body tissues and their role as physical and chemical forces have been overlooked. This has led to many misconceptions. For example, the results of Finzi and others have been taken to indicate that ions would penetrate to the periapical tissues from the pulp chamber and root canal with a direct current flowing. We have found that this is not wholly true for some ions will not penetrate the tooth wall and their conduction thru the periapical foramen is liable to be altered by the ebullition of gas. The migration velocity of the ions in use appears on calculation to be so slow that it is doubtful if the ions in the solution in the pulp chamber actually reach the periapical tissues. A further consideration of the results of some investigators has led us to presume that the reported effects of electrolytic medication must be referred to something other than the ions contained in the solution, which is placed in the pulp chamber, or the electrode in use.
It will be necessary, to make this clear, to discuss the questions in the following way. So far as the physical effect of the electric current upon the ions, in a liquid, is concerned it is to increase the diffusion in a certain direction. One result of the passage of the current is thus to increase intensity of the ions at certain points in a mean path. The electric current thus directs the ions, those ions moving to the anode are called anions, those to the cathode cations, after the original nomenclature of Faraday. The electricity cannot flow thru a liquid conductor unless there are ions to conduct it.
It will be of value here to review the development of this conception. The determinations of osmotic pressure by Pfeffer, De Vries, Van t’ Hoff and Arrhenius indicated that there were two classes of substances, one.group which conformed to a theoretical conception of the chemicals, and another group which were extreme exceptions to this formulation. The former group includes the sugars, the latter, acids, bases and salts. It was observed that the latter group in water solutions conducted electricity while the former did not, and thus it appeared that there must be some connection with the acids, bases and salts and the galvanic stream. The acids, bases and salts, it was also noticed, did not conduct electric current in other solvents to any marked degree. In water it was found that HCl exerted twice the effect upon the freezing point (a physical test of osmotic pressure) that it would have if it acted as a gas and thus it seemed as tho it must be split up into two parts, H and Cl. But hydrogen and chlorin were known to exist when gases as H2 and Cl. It was at first difficult to understand what happened to the HCl in the solution. It had been observed that the passage of electric current thru a liquid is accompanied by the transference of matter; for example, the ions, to which we have referred. It is associated also with either the solution of the metal of the metallic electrodes or the separation of the substance in the solution and its deposition on the electrode. Dry HCl gas does not conduct electricity. There is no conduction with very pure water between the poles, say, of a battery. The current, however, does pass freely when the HCl is dissolved in the water. Now free chlorin separates at the anode where the current enters the solution and free hydrogen at the cathode where the current leaves the solution. One component, then, wanders in one direction, the other in the other direction. Faraday called these wandering particles “ions.” The passage of positive electricity into the solution at the anode is associated with the separations of anions and the passage of the positive electricity out of the solution into the cathode is associated with a separation of cations. The term dissociation had been early applied to the separation or cleavage of larger into smaller molecules, or molecules of one into molecules of a different species and was utilized here, the phenomenon being termed “electrolytic dissociation.” Some have spoken of it as “ionization” but this term is reserved by a growing group of scientists to apply to the formation of ions in gases produced by the action of Röntgen rays and the like. From experiments, it is known that molecules do not always split up into the ions in the same way, nor do all of the molecules entirely split up. Some portions of the bodies remain in the solution as undecomposed molecules which are electrically neutral and do not, then, conduct electricity. H2SO4, for instance, splits up into H+, and HSO4, or in more dilute solutions into 2H+ and SO4–.
If, now, in a water solution of HCl we place two platinum electrodes and connect these with a source of galvanic current one plate, connected with the positive pole of the battery, is charged positively and the other, negatively. The free electricities of the electrodes act upon the free ions which are charged with free electricity attracting the positive ion by the negative electricity of the cathode to the negative pole, and the negatively charged ion to the anode, the positive pole. No such force will be exerted upon the neutral molecules. The same quantity of electric current sets free chemically equivalent quantities of the most various ions from the most various solutions. This is Faraday’s law. This law, of course, applies to the quantity of each ion which is deposited as neutral molecules at the electrodes and not as Prinz23 would have us understand to the production of ions in the solution. Electrolytic dissociation and the deposition of ions at the electrodes with the passage of electricity is one thing; the production of ions by the cleavage of a salt, acid or base, in the solution apart from, and independent of, the passage of electric current is distinctly another. Confusion here makes a tremendous difference in one’s conception of electrical and ionic action. The ions, then, do not depend upon the current but the current depends upon the ions.24
Since Sturridge25 suggests that one can “dissociate a particular ion” at the anode and send it into the tissue by the aid of the current, we may consider this point. This author states that the zinc ion will dissociate from the chlorin ion at the anode. In his text he specifies a zinc electrode in a zinc chloride solution. Apparently he neglects the zinc ion dissociated from the zinc chloride and proceeds to speak of the therapeutic value of the zinc ion from the electrode. If the electrolytic dissociation theory is true there are zinc ions throughout the solution wherever the zinc chlorid is dissolved. A zinc electrode placed in this zinc chlorid solution throws off a few ions–before the current is turned on–and then no more of the metal ions go into solution. A balance exists, or as we speak of it physically, there is “equilibrium.” No more zinc ions, beyond these few, will go into solution even if the solution be pure acid or there be acid in the zinc chloride solution. But if in the solution a platinum strip be placed and connected with the zinc strip, the equilibrium is disturbed. More zinc ions go into solution. With acid present hydrogen ions from the acid give up their charge to the platinum and escape as gas. The platinum now becomes positive with respect to the zinc and a positive charge flows from it to the zinc. The zinc having received positive electricity can begin dissolving anew and continue to pass into solution as long as it receives positive electricity from the platinum, that is, as long as there are any hydrogen ions in the solution to furnish positive electricity to the platinum. The passage of an electric current as from a battery thru a zinc chlorid solution with a zinc electrode as anode, and, let us suppose, a platinum electrode as cathode results in a similar transformation. As fast as the zinc ions are separated out of the solution at the cathode, zinc would dissolve at the anode, the metal electrode throwing off cations. Even in a sodium chloride solution this would result. For as fast as the cations (sodium in this case) in the vicinity of the cathode are precipitated upon the electrode the zinc electrode would dissolve, sending into solution just as many ions as are separated at the cathode.
Since Prinz doubted the presence of zinc in a sodium chloride solution under the above conditions, and there is no theoretical explanation why they should not be present, we experimented as follows: A zinc and copper electrode were immersed in a beaker with twenty-five cubic centimeters of a one per cent sodium chloride solution (Prinz, loc. cit.), and a current of five milliamperes was passed thru the solution in the direction of zinc to copper. One cubic centimeter samples were removed every fifteen minutes for two hours. One cubic centimeter of an ammonium sulfide test solution was added to the portion. In one hour traces of zinc were visible and the precipitate was increased noticeably in the samples taken after a longer time of action. (Tests of the sensitivity of this particular reagent indicated that one part of zinc in 20,500 parts of solution could be detected). This appears to verify the statement that zinc ions exist and indicates that they can be detected in a sodium chloride solution after the passage of an electric current of 5 ma, if sufficient time is allowed. Six minutes allowed by Prinz26 was too short a time for enough zinc to dissolve from the anode so that it could be detected by the reagent. As will be seen in our tests, it required one hour for more than one part of zinc in 20,000 parts of solution to be present.
It will be of interest here to discuss the migration velocities of the ions, since in electrolytic medication it is presumed that the ions (the supposed disinfecting agents) will travel from the root canal to the periapical tissues during the time of medication. Now hydrogen, the fastest moving ion, penetrates a gelatine solution (where water is in the usual physical sense the “continuous” phase) to a depth of 0.18 cm, or 0.02 of an inch per minute with a gradient of one volt. Other ions move from five to ten times as slowly or one-fifth to one-tenth as fast. Zinc would penetrate under the same conditions a little faster than 0.003 of an inch per minute. If a metallic ion traveling as fast as zinc actually diffused thru the tooth structure or the apical foramen, the amount in fifteen minutes at this rate would be small; but as we shall see presently the physical conditions of the tooth are not the same as in a gelatine or water solution.
We have, thus far, in dealing with the electro-chemical phenomena, considered the condition as similar to a battery, that is, two electrodes separated only by a liquid. We must now observe what conditions obtain where the electrodes are separated by liquids either side of a semi-permeable membrane, a porous partition like the tooth wall, by a capillary like the root canal, and by body tissues.
We began our experimental work upon this question by assuming that the devitalized tooth was a porous partition not unlike a clay wall. We reasoned that if this were true we could determine approximately the flow of cations or anions thru the pores in unit time, and, by calculating from the amount of disinfecting ions necessary to sterilize a definite quantity of infected tissue, we could determine the time necessary to disinfect under more or less definite electrical and chemical conditions. But not only did we find that the ions we used would not kill bacteria, we found that the cation zinc and the anion iodide (two supposedly reliable disinfecting agents) would not penetrate the porous partitions we used. Copper we found would penetrate where zinc would not. It made no difference what concentrations or what quantity of solution we used, similar results were obtained.
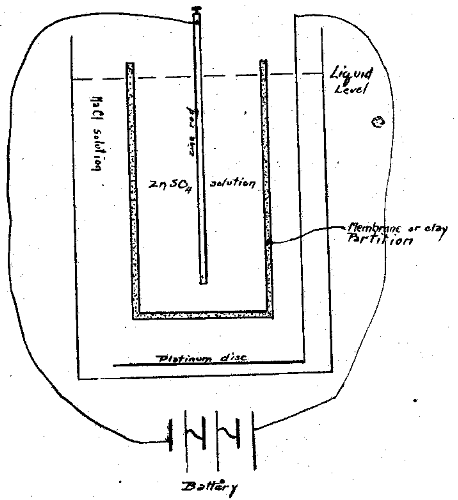
Our experimental cells were as follows: (a) “porous cup” of a battery; (b) Berkfield (clay) and Alundum filters; (c) porcelain tubes of the average volume capacity of a tooth; (d) freshly drawn teeth with apex sealed. The “set up” may be illustrated as follows:
Schematic Arrangement: Experimental Diffusion Cells
Figure I
The solutions in the inner and outer chambers were always adjusted to the same level and were made up of equal osmotic pressures. The quantities were reduced to very small volumes in some cases and relatively large in others. For example, the teeth and small porcelain tubes would hold 0.1 to 0.5 c. c. and were immersed in 0.5 c. c. to 1 c. c.; the filters held 2 c. c. and were immersed in 10 c. c.; the porous cups holding 25 c. c, were in 30 c. c. of solution. The apparatus available was not ideal but the best at hand. The osmotic pressure which the porous partitions would sustain, and the total cross sectional area of the pores, could not be determined with means and time at hand. In every case a constant current was passed thru the cells for nearly three hours. The current used varied from 0.2 ma to 5 ma and the potential was from 2.4 volts in some cases to 28.5 volts in others. 0.5 c. c. samples of the sodium chloride solution were removed at intervals of five minutes up to thirty minutes and every 15 minutes thereafter. To this was added an equal quantity of one of three reagents of known sensitivity, viz. normal Na2CO3, normal potassium ferrocyanide, and an ammonium sulfide solution prepared by saturating 300 c. c. 6-N NH4OH with H2S, adding 200 c. c. 6-N NH4OH and diluting to a liter. In spite of careful tests where the reagent was sensitive to as little as 1:300,000, no zinc was detected after the current had passed for more than two hours. With copper the tests with the potassium ferrocyanide indicated its presence after fifteen minutes. Potassium iodide could not be detected with soluble starch solution–(0.5 gm in 25 c. c. of boiling water) under similar conditions to those with zinc and copper. These experiments indicated to us that the tooth and clay or porcelain wall was impermeable to the zinc and iodide ions, but not to copper. Other investigators have reported similar results.27
An explanation of these results involves a consideration of the size of the pores, the size of the ions, the speed of migration, the electrical condition of the porous partition and the chemical condition of the ions. It has been found that the tooth wall bears a negative electric charge to water like clay and porcelain. This would account for the opposition to the anion iodide under some conditions. In experiments where Al Cl3 was added, the charge was reversed and the iodide was found to penetrate, demonstrating that this is presumably correct. Zinc ions would not reverse the charge of the partition. With zinc sulfate in the anode chamber in the case of large pored partitions, it is conceivable that the positively charged zinc ions are attracted to the partition and held there, forming a positive layer, blocking the passage of any more cations thru the pores. The bulk of the current would be, in such cases, conducted by the anions. In the tooth the pores are infinitely smaller and the anions would be repelled by the negative charge of the wall. As the current flowed thru the wall and the zinc ions were detected chemically as having passed thru (verified also electrometrically), we assume that the current was carried mainly by hydrogen and chlorin ions. It is possible that hydration of the zinc ions occurred and hence the ions so enlarged were not able to penetrate the pores. Copper supposedly is not enlarged and thus may penetrate readily. These questions must be further investigated.
To determine whether the ions had free access thru the apical foramen and what factors were involved in the conduction of the current from the pulp chamber to the surrounding tissues, particularly the proportion of current passing thru the apical foramen to that thru the lateral walls to the periodontal membrane, we have made the following experiments.
In the literature on ionic medication the authors seem to have taken for granted that the current was being delivered to the tissues involved. This problem of current path had previously been studied by one of us, (Price, Items of Interest, May 1898) in connection with cataphoresis.
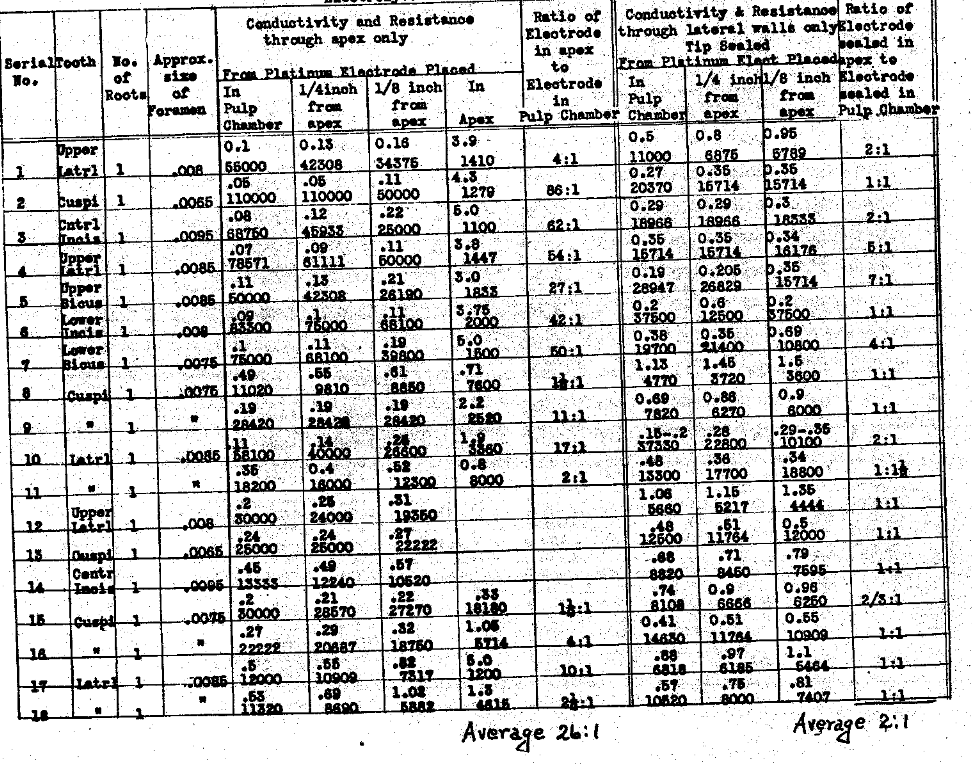
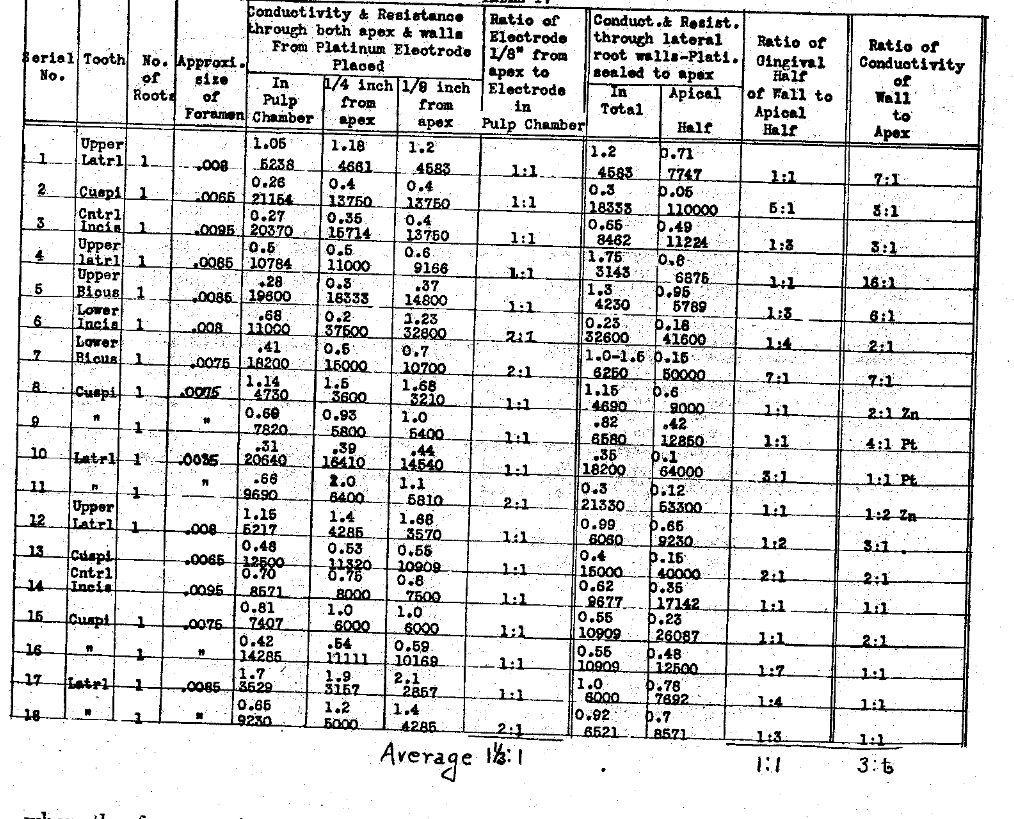
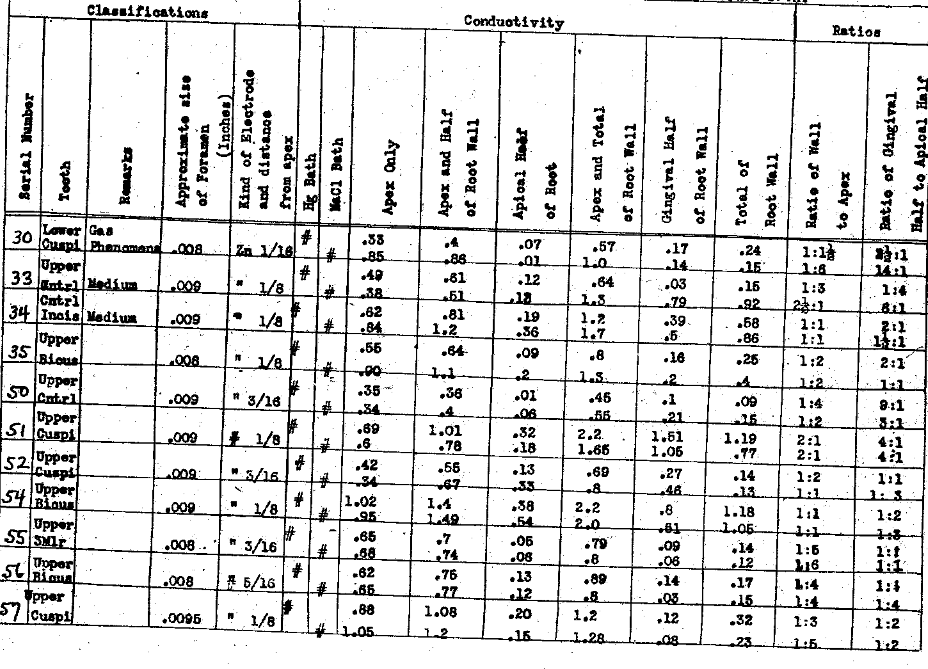
In tables III and IV we have determined the influence of the position of the electrode on the conductivity thru each the apex and lateral walls, and the ratio of the current carried thru the apical foramen when the electrode is within one-eighth of an inch of the apex, to the current carried by that same column of normal salt solution when the electrode is placed in the pulp chamber, usually producing a column about one-half an inch, or a little less, in length. In these series we have also studied the influence the various positions of the electrode have had upon the current conductivity thru the lateral walls of the tooth when the apex was sealed. These observations demonstrate that the position of the electrode has a great deal to do with the quantity of current that can be carried thru each the root apex foramen and the root walls. In the former if the electrode is placed just to exit of the foramen, tho not beyond, the ratio of current to that carried when it is within the pulp chamber ranges from two to one to eighty-six to one, with an average ratio of twenty-six to one. This demonstrates the importance of extending the electrode to the apex. The influence of these variations of position of the electrode upon the conductivity thru the root walls is not nearly so great, averaging only as two is to one. This emphasizes the necessity that the electrode extend to the apex. The results of the series also indicate the actual current conductivity thru each the apical foramen and the lateral walls and express them relatively to each other; the actual carrying capacity of the lateral walls as compared with the foramen, using for the ratios the position of the electrode one-eighth inch from the apex. It is much the greater, varying from equal to sixteen to one in the extreme, with an average in the series of three to one. Since the pathological condition is presumed to be directly related to the foramen and quite limited to that area of the tooth, in such cases as would be considered favorable for this method of treatment, we must conclude that with the electrode placed to about one-eighth of an inch from the apex, only one-fourth of the total current could be passing thru the pathological tissue, and the other three fourths would probably he penetrating normal tissues. In this series we have reduced the conductivities thru the various paths to ohms resistance, as well as expressing them in milliamperes carrying capacity. We have also determined the ratio of the conductivity thru the gingival half of the root wall to the apical half, not including the foramen. These show a wide variation but in the majority of cases the conductivity is greater thru the gingival half of the root walls than the apical half.
Table III–Electrolyte-Normal Salt Solution
The Electrical Conductivity of Different Structures and Channels of the Teeth.
Influence of The Position of the Electrode of the Conductivity Through Each the Apex & Lateral Walls
Table IV–The Electrical Conductivity of Different Structures and Channels of the Teeth.
Influence of the Position of the Electrode on the Conductivity Through Each the Apex & Lateral Walls
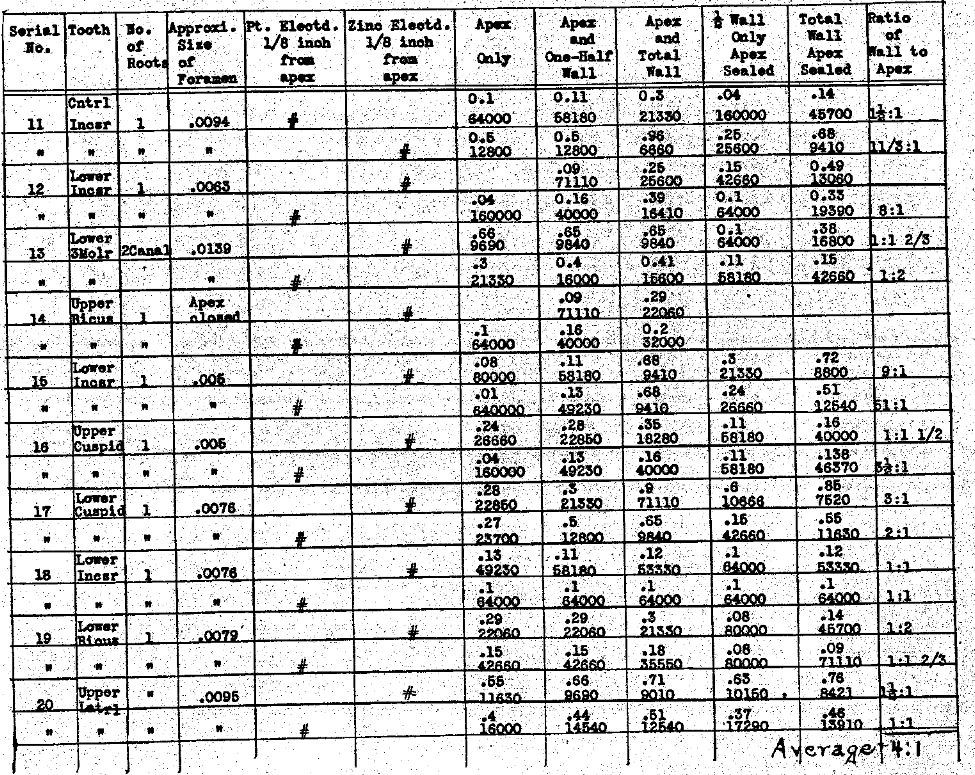
In Table V we have determined the difference in the conductivities, whether a platinum electrode is used or a zinc electrode, and also the amount of current carried thru each the apical foramen, the apical half of the root wall and the gingival half of the root wall, as well as to tals. The rate of generation of gas is a little greater with platinum than with zinc in a sodium chlorid solution, the gas being different and the conductivity is, accordingly, generally greater with the zinc electrode than with the platinum. While this series shows a wide variation in the ratio of conductivity thru the lateral walls of the root to that of the apical foramen, the average for this series is as four is to one. In other words, one-fifth of the total current flowing would, on the average, be passing thru the foramen to the pathological tissue and four-fifths to the healthy periodontal membrane thru the lateral walls.
Table V–The Electrical Conductivity of Different Structures and Channels of the Teeth.
Comparison of Zinc & Platinum as Electrodes and of Conductivity & Resistance of Different Current Paths
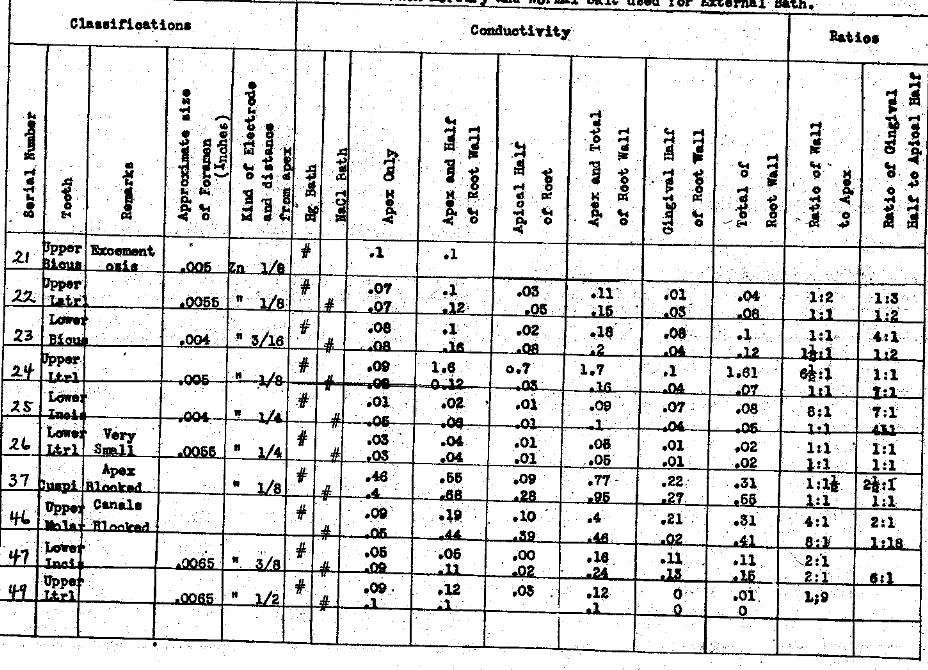
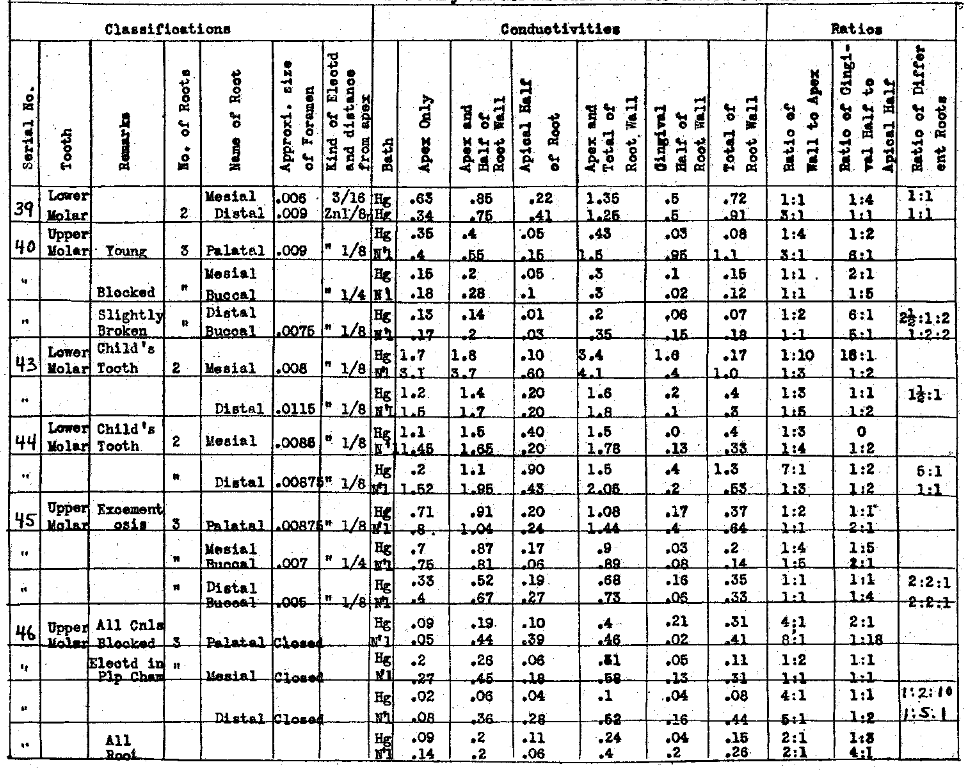
In Table VI we have studied the conductivities thru apices and lateral walls when the foramen is very small or apparently blocked. These have shown a very high resistance thru the apex. It will be noted that in the fourth column of each of these charts we have recorded the approximate size of the main apical foramen. We could conveniently measure only one, namely, the one nearest in alignment with the pulp canal. In this series (Table VI) the apices were from .004″ to .0065″, or entirely blocked in Nos. 37 and 46. It is interesting to note that, while the apex of No. 38 was blocked against the wire used, the conductivity indicates that the foramen is at least medium in size and the shape of the canal undoubtedly prevents passing an instrument thru it. This series demonstrates that when the foramen is small it is a matter of wide variability as to what the ratio of conductivity of the wall will be to the apex. The same conditions that produce the closing of the foramen probably cause an increase in the density, therefore the lowering of the conductivity of the lateral walls of the tooth as well. There is also a very wide variation in the conductivity of the gingival half to the apical half. No. 21 (Table V), for example, had a foramen .005″ in diameter and the tooth had extensive excementosis. The walls of the tooth were so poor a conductor that practically no current was carried and only a very small amount thru the apex. Excementosis greatly reduces the conductivity thru the root wall and, since it is apparently in many cases the result of a local irritation in the form of an infection, we are not able to reach that infection by this means, even if it were competent to destroy the organisms.
Table VI–Each Mercury and Normal Salt used for External Bath.
The Electrical Conductivity of Different Structures and Channels of the Teeth.
The Conductivity of Small Size Apical Foramina and Their Ratio to Other Paths
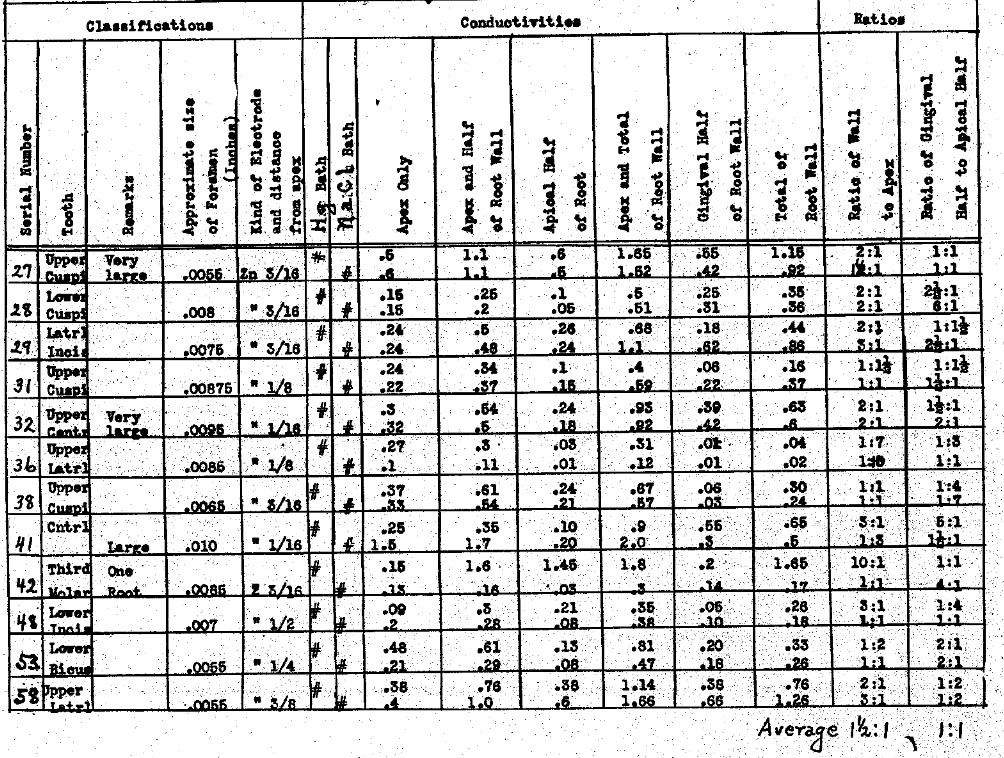
In Table VII we have studied the conductivity of medium sized apical foramina and their ratios to other paths. In this class of tooth we have included foramina ranging from .0055 to .0010 of an inch in diameter. We have placed the electrode, in this case zinc, as far into the canal as possible and in no case did it penetrate the apex, distant from about one sixteenth to three-sixteenths of an inch. Two baths were tested, namely, sodium chlorid and mercury. It will be noted that the conductivity of the root wall is very much greater in the teeth in Table VII than in Table VI, which emphasizes the point mentioned relative to the teeth with small foramina, having also an increased resistance to the current in the root wall. Generally, the conductivity thru the wall is greater than thru the apical foramen. In this series the ratio of the gingival half to the apical half of the root has considerable variation, the chances to passing current streams being about equal.
Table VII–Each Mercury and Normal Salt used for External Bath.
The Electrical Conductivity of Different Structures and Channels of the Teeth.
The Conductivity of Medium Size Apical Foramina and Their Ratios to Other Paths.
With Table VIII we have studied the conductivity of large apical foramina and their ratios to other paths. This has included those between .008″ and .009″. These naturally show a marked increase in the amount of current passing thru the apex and also a marked increase in the conductivity of the lateral walls. The ratio of the conductivity laterally thru the walls of the tooth to that passing thru the apical foramen varies in quite a wide range, yet the average for the series shows twice as much going thru the foramen as laterally thru the walls. The ratio of the gingival half of the root wall to the apical half is as two is to one.
Table VIII–Each Mercury and Normal Salt used for External Bath.
The Electrical Conductivity of Different Structures and Channels of the Teeth.
The Conductivity of Large Size Apical Foramina and Their Ratios to Other Paths.
In Table IX we have made a comparison of the conductivity of different roots of multi-rooted teeth. The foramina ranged in size from no opening to .011″ diameter, the latter being a child’s tooth in which the conductivities were large thru each the apex and the lateral walls. No teeth were used that were extreme. The ratios of conductivity show a very wide range, suggesting that it is exceedingly problematical in each and every case in the mouth which, of different roots, is carrying the most current, and what their ratios will be. The same is true of the ratios between apex and lateral walls, which vary from one to one to one to ten. There is a greater variation, however, than in any other series between the ratios of the gingival half and the apical half, they being as high as one to eighteen. The ratios between the different roots range from five to one to ten to one. When an electrode is passed thru the apex into a fluid beyond the root, the conductivity is enormously increased, both because of the elimination of the resistance of each the small column of liquid in the root canal and foramen and of the lateral wall, and is limited only by the voltage and the ability of the gas, formed at the electrode, to escape.
Table IX–Each Mercury and Normal Salt used for External Bath.
The Electrical Conductivity of Different Structures and Channels of the Teeth.
A Comparison of the Conductivity of Different Roots of Multi-Rooted Teeth.
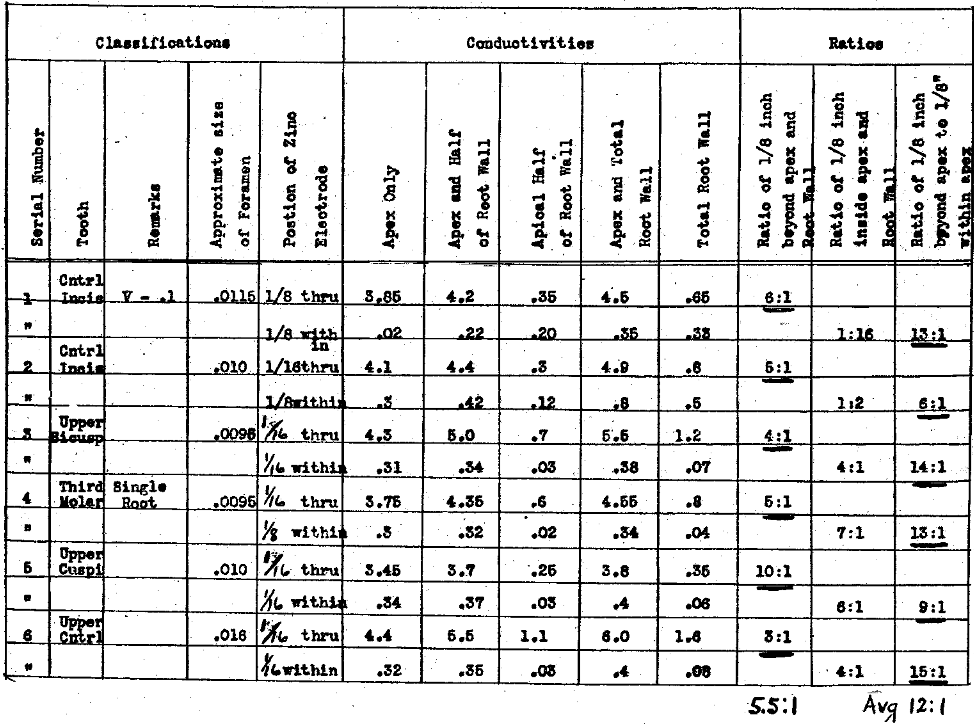
In Table X we have made determinations of the difference in conductivity when a zinc electrode is extended thru the apex, for from 1/16 to 1/8 of an inch to the same when placed about that distance within the apex. We have also determined the ratios to the root walls under these conditions. Owing to the very greatly reduced resistance of the circuit, when the electrode was passed thru the apex, it was necessary to reduce our voltage to 0.1. The figures in Table X, therefore, cannot be compared directly with those in the previous charts where voltages ranging from 5.5 to 6.6 were used. This will, however, not disturb the ratios. It will immediately be noted that the ratio of the conductivity thru the lateral walls to that thru the apical foramen has been reversed from the condition shown in previous charts, the average being 5.5 to 1, whereas the ratio of conductivities, when the electrode was withdrawn to 1/8 of an inch within the apical foramen to that of the root walls, averages one to one, even with so large an apical foramen. The most significant factor is the ratio of the conductivity with the electrode 1/16 to 1/8 of an inch beyond the apical foramen to 1/16 to 1/8 of an inch within, the average for which is twelve to one. This very strongly emphasizes the necessity for placing the electrode, if possible, not only to the apical foramen but thru and beyond it if the effects to be produced are to be delivered to the periapical tissues. This condition, however, is difficult with a zinc electrode where the foramen does not exceed in size .008″ to .010″ of an inch. Such a foramen will be found in the teeth of young people or produced artificially by enlarging. We must not, however, consider that our difficulties are eliminated by passing the electrode beyond the apex, for in so doing the gas cannot readily be eliminated as it does automatically from the pulp canal. This will be discussed presently.
Table X–The Electrical Conductivity of Different Structures and Channels of the Teeth.
A Comparison of the Conductivity of A Zinc Electrode Extended Through the Apical Foramen About One Eighth Inch to the Same in Equal Distance Within the Apex. V-.1
Another condition that must not be overlooked is that of the production of heat by the passage of a concentrated current. One of us, Price,28 has previously estimated the heat generated in the different paths of an electric current passing thru the tooth and periapical structures. It is not probable, however, that a sufficient concentration of current may be used to considerably raise the temperature for any considerable distance from the electrode. If it were possible the application of heat in this form would be an ideal way to sterilize.
The age of the patient has a very marked influence on the conductivity thru the lateral walls of the tooth, as well as on the size of the apical foramen.
When we apply these facts to the actual and relative conductivities of different paths, as determined by different conditions of apex and the root wall, to our practical problems, we have some of the following conditions to meet. Let us suppose a lower molar has an opening thru the distal root of .008″ to .010″ thru which an electrode is inserted. A second electrode is passed into the mesial root as far as possible. Unless the mesial canal has been greatly enlarged, it will not be possible to go beyond the apex with a zinc electrode in a large number, if not the majority, of cases. We attach our current to both these electrodes. What is the relative amount of current that will be applied to the periapical tissues of each root? Since the distal electrode is passed beyond the foramen we have eliminated the resistance of the small column of fluid and its obstruction with the gas of electrolysis. The ratio of the conductivity of an apical foramen of small size to one of large will be as one to five or ten. (See Charts Tables VI and VIII.) Passing the electrode beyond the apex would increase this conductivity still another ten to fifteen fold (See Chart Table X). The ratio of the current passing to the tissues about the apex of the distal root to the tissues about the apex of the mesial root would probably be anywhere from fifty to one to one hundred and fifty to one. The logical procedure is to place the electrode in one root at a time and thru the apical foramen, if possible. (This, however, does not yet provide for the elimination of the gas, which, of necessity, forms about the electrode. We see little evidence that gas is a good form of medicament to be applied in the tissues at the apex of the root, but this may be left an open question for further research.) The conditions that obtain with a three rooted molar with the electrode passed thru the apex of any one root would in general correspond, the current always following the path of least resistance, entirely irrespective of distance or direction. We wish to emphasize this because some exponents of this method have erroneously assumed that the current is projected from the end of the electrode much as a high tension static spray or brush.
(Continued in a later issue.)
(a) Ten grams by weight diluted with distilled water to one hundred grams of solution.
(b) We have used “per cent” concentrations because this term occurs in the dental literature on this subject. We have calculated all of the concentrations in terms of the molecular weight in a liter of solution. It is to be hoped that in dental research of the future, investigators will utilize the latter (molecular) concentrations and, as well, the chemical equivalent (normal) concentrations, terms used by the analytical chemist. It would then be possible without difficulty to recognize equivalent chemical action, and compare, readily, results from different hands.
References Cited:
- Journal of the National Dental Association. March 1917.
- Prinz, H. “Electro Sterilization,” Dental Cosmos, April 1917, page 373.
- Carrel and Hartman, Jr. Experimental Medicine. ’16, XXIV, 5,429; DuNouy, Ibid, 451; Same XXV, 5,721; Vincent, Ibid XXVI, 1,83.
- Arrhenius, Immunochemistry, N. Y. 1907, Macmillan; Madsen and his pupils “Memoirs in the Communications de I’ Institute serotherapione de l’ Etat danois 1907-13.”
- Prinz, Cosmos, LIX, 4,393, April 1917.
(4) Arrhenius, Quantitative Laws in Biological Chemistry, London, 1915, G. Bell and Sons, Ltd. - Sturridge, E. “Dental Electro-Therapeutics,” Lea and Febiger, N. Y. 1914.
- Tousey, Sinclair, “Medical Electricity”–Saunders, 1915; Jones, Lewis. “Ionic Medication,” Blakiston, 1914, 2d ed.; Fette, G. T., Cosmos, March 1917, and this Journal, March, 1917.
- Lillie, R. S. Biological Bulletin, Vol. 5,311, May 1916.
- 1896–Zeitschr–Phys. Chem. 21, 414.
- Sternberg, G. M., Manual of Bacteriology, N. Y. 1893.
- Boer, cf. Sternberg loc. cit.
- Kitasoto, cf. Sternberg loc. cit.
- Prinz, H. Dental Cosmos, April 1917, page 388.
- Dakin, H. D., Proceedings Royal Society, England, 1916, B. 89, 234.
- Loeb, J., Dynamics of Living Matter, Macmillan, N. Y. 1906, page 98.
- Köller, H., Correspondenz-Blatt für Schweiser Aerzte, Basel, April 28, XLVII, No. 17, pp 513-44 and No. 16, p 485.
- Items of Interest, May 1898.
- Finzi, N. S., Brit. Med. Journal, November 2, 1912.
- Jones, H. Lewis, Ionic Medication, 1914, 2d Ed.
- Sturridge, E., Dental Electro Therapeutics, 1914, 216.
- Sturridge, E., loc. cit. p. 218.
- Sturridge, Royal Soc. Med. Odontological Section, 1912, and loc. cit. 214, Vol. 5, p. 102.
- Prinz, loc. cit., p. 379.
- Adapted from Nernst, 6th Edition, Theoretical Chem. p. 356.
- Loc. cit., p. 245.
- Prinz, loc. cit., p. 381.
- McClendon, Physical Chem. of Vital Phenomena, 1917, pp. 104-08.
- Price, W. A., “Foundation principles of cataphoresis,” Items of Interest. May, 1898, p. 344-376.