Access to all articles, new health classes, discounts in our store, and more!
Carbohydrate Fermentation Reactions of Staphylococci
Published in American Journal of Clinical Pathology, Vol. 10, No. 6, June 1940. Co-Author: Merritt H. Stiles.
* * *
A survey of the literature indicates that wide variations in technic and in the incubation period may have been chiefly responsible for extreme differences in the proportion of staphylococci fermenting certain carbohydrates, and differences in the association between carbohydrate fermentation reactions of staphylococci and other properties of the cultures.
Hoffstadt and Youmans,1,2 Pinner and Voldrich,3 Haberman,4 and others have shown that staphylococcal cultures may dissociate into variants which differ in their fermentative properties. Thus, it is possible that, during the prolonged incubation period (5 to 30 days) often used in studying the fermentation reactions of staphylococci, dissociation of the cultures may occur which might affect the fermentation reactions. The advantages of a shorter incubation period do not appear to have been recognized until Chapman, Lieb and Curcio5 reported that staphylococci which ferment mannitol on the surface of plates of Bacto phenol red mannitol agar usually produce detectable acid after overnight incubation. This brief interval eliminated the influence of variants having fermentative properties differing from those of the parent cultures. Thygeson6 found that this technic gave more reliable results than did previous methods. The results agreed with those of the coagulase test in 98.2 per cent of 168 strains and with the power to produce conjunctivitis-producing toxin in 97.4 per cent of 196 cultures.
In the investigation to be described the method was applied to the study of the fermentation of other carbohydrates by staphylococci and the results were compared with certain reactions of the cultures usually associated with pathogenicity.
Technic
The base medium used in the fermentation studies (Bacto phenol red agar base) had the following composition:
| grams | |
| Bacto beef extract
Bacto tryptose Sodium chloride Bacto agar Bacto phenol red |
3
10 5 15 0.025 |
Enough water was added to make the total volume 1000 cc. after adding the carbohydrate solution. The reaction was adjusted so that after sterilization and when ready for use the pH was 7.4.
Concentrated solutions of the carbohydrates were filtered through Seitz filters and added to the cooled base medium to make 1.0 per cent concentration and poured immediately into Petri dishes. The poured plates were set aside in the refrigerator until the following day to reduce running of growths caused by condensation.
The cultures were streaked across the surface of the media and incubated at 37°C. After overnight incubation the plates were inspected for evidence of fermentation, as indicated by the appearance of yellow zones around the growths. If the results were negative the plates were returned to the incubator and re-examined daily. The plates were discarded after 5 days because only an occasional change occurred after 48 hours, and later changes were likely to have been caused by the development of variants in the cultures.
Because there is general agreement that the power of a culture to coagulate human or rabbit plasma, produce orange pigment, hemolyze rabbit blood agar in 18 hours and produce orange or violet growths on crystal violet agar* is closely associated with the pathogenic properties of the culture for man and rabbits, these reactions were used as “in vitro” indicators of “pathogenicity.”
(*It has been called to our attention that Epstein7 described a crystal violet agar reaction, usually referred to as the “Epstein phenomenon,” which is similar to the crystal violet agar reaction of Chapman and Berens.8)
Cultures Used
Staphylococci recently isolated from persons suspected of having chronic infection were classified as follows:
Pathogens (70 cultures): Producing orange pigment. Hemolytic on rabbit blood agar after overnight incubation. Coagulating human or rabbit plasma within 18 hours. Producing violet or orange growths on crystal violet agar.
Degenerate pathogens (22 cultures): Coagulating human or rabbit plasma within 18 hours, but lacking one or more of the other properties characteristic of pathogenic staphylococci.
Non-pathogens (33 cultures): Not coagulating human or rabbit plasma.
Results Obtained
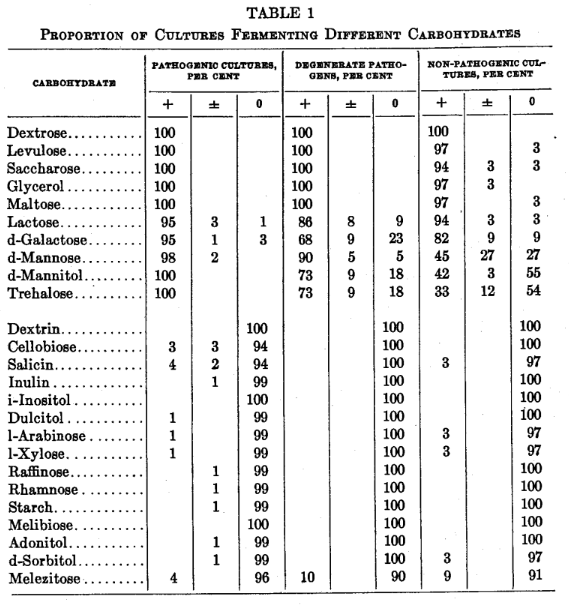
The carbohydrates were fermented more easily by the pathogenic group, although there was no close relationship between the fermentation and in vitro reactions of a particular culture except in the case of d-mannitol, trehalose and, to a lesser extent, d-mannose.
All cultures fermented dextrose and glycerol, and all except one fermented maltose, saccharose and levulose. Lactose was fermented by 121 of the 125 cultures. d-Galactose was fermented less easily than lactose, 10 cultures giving negative results but the distribution, like that of lactose fermentation with which it was parallel in most instances, was unrelated to the in vitro tests of the cultures. d-Mannose, d-mannitol and trehalose were fermented more frequently by pathogenic type cultures (table 1).
Only exceptionally did a culture ferment dextrin, i-inositol, melibiose, inulin, dulcitol, l-arabinose, l-xylose, raffinose, rhamnose, starch, adonitol or d-sorbitol. Melezitose was fermented by 8 cultures but these included pathogens, degenerate pathogens and non-pathogens. Cellobiose was fermented by 4 pathogens. Salicin was fermented by 4 pathogens and 1 non-pathogen.
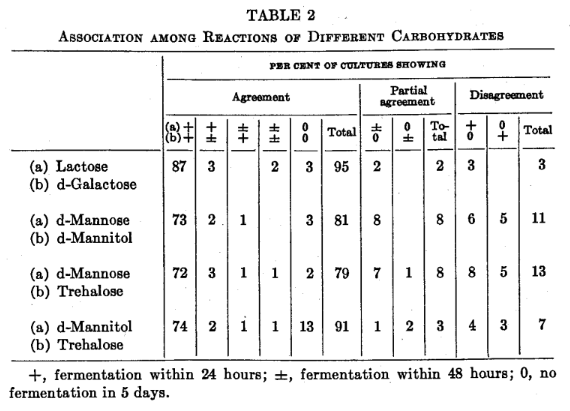
Certain associations between the fermentation of different carbohydrates appeared to be significant. There was agreement between lactose and galactose fermentation in 119 cultures and partial agreement in 2 cultures (table 2).
d-Mannitol and trehalose showed similar reactions in 113 cultures and closely related results in 3. d-Mannose and d-mannitol reactions agreed in 101 cultures and were closely related in another 10 (table 2).
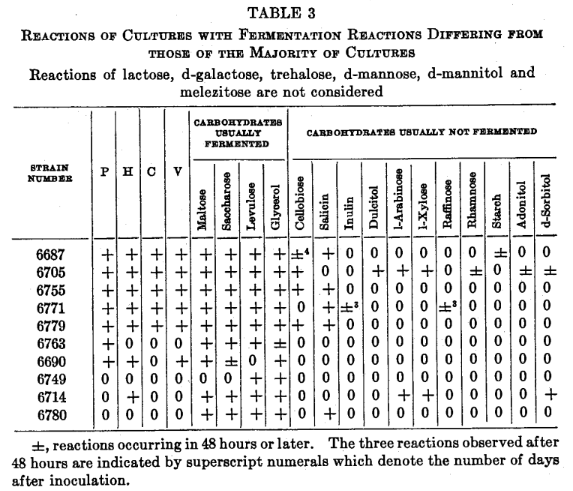
The two strains fermenting l-arabinose also fermented l-xylose and d-sorbitol. Two cultures (6705 and 6771) which produced atypical colonies fermented carbohydrates not fermented by most cultures. These associations are detailed in table 3.
Delayed fermentation (later than 18 hours) occurred in 3 of 103 fermenters of d-mannitol, 11 of 115 d-mannose, 6 of 103 trehalose, 4 of 120 lactose, 6 of 105 d-galactose and in 2 fermenters of raffinose. It occurred only once in each of the following: saccharose, glycerol, cellobiose, starch, rhamnose, adonitol, d-sorbitol, inulin and salicin. With three exceptions, all cultures that fermented any of the carbohydrates did so within 48 hours.
Discussion
A few pathogenic strains had such vigorous fermenting power that they fermented certain carbohydrates not fermented by the majority of strains. Conversely, the fermentative power of a few non-pathogenic cultures was so reduced that they failed to ferment certain carbohydrates readily fermented by the majority of strains. Although the power to ferment. carbohydrates was more active in pathogenic than non-pathogenic cultures, fermentation reactions were parallel with in vitro properties only in d-mannose, d-mannitol and trehalose.
The association of fermentation of d-mannose, d-mannitol and trehalose, particularly the two latter, is of considerable interest in view of the finding of many investigators that fermentation of d-mannitol is a characteristic property of pathogenic staphylococci. Fewer non-pathogens fermented trehalose than d-mannitol in 18 hours. All highly pathogenic cultures fermented d-mannitol and trehalose within 18 hours, while a number of degenerate pathogens failed to ferment these two carbohydrates. Because these three carbohydrates give such contrasting results among different strains, they should prove valuable in dissociation studies.
Contrary to common opinion, lactose fermentation was found unrelated to the in vitro properties. Lactose was fermented by 98 per cent of pathogens, 91 per cent of degenerate pathogens and 97 per cent of non-pathogens. Chapman, Berens, Nilson and Curcio9 found that slightly more pathogens than non-pathogens fermented lactose under similar conditions (100 and 80 per cent, respectively).
Conclusions
Most staphylococci, regardless of their in vitro reactions, ferment dextrose, levulose, saccharose, glycerol and maltose within 18 hours and fail to ferment dextrin, cellobiose, salicin, inulin, i-inositol, dulcitol, l-arabinose, l-xylose, raffinose, rhamnose, starch, melibiose, adonitol, d-sorbitol and melezitose within 5 days, when tested on the surface of a tryptose, beef extract agar with phenol red indicator.
Pathogenic strains as a group had greater fermenting power than the group of non-pathogenic strains, conforming to the generally accepted principle that pathogenic strains of staphylococci show greater metabolic activity than non-pathogens.
Lactose and d-galactose gave parallel results in 119 of 125 cultures but the reactions were not associated with the coagulating, chromogenic, hemolytic and crystal violet agar properties of the cultures.
Fermentation of d-mannose, d-mannitol and trehalose was more closely associated with these in vitro properties, the degree of correlation increasing in that order.
Two cultures, producing atypical growths, fermented carbohydrates which were not fermented by 97 per cent of the cultures.
The 4 cultures which fermented cellobiose and 4 of the 5 fermenters of salicin had in vitro properties characteristic of highly pathogenic staphylococci.
References Cited:
- Hoffstadt, R. E., and Youmans, G. P.: “Staphylococcus aureus. Dissociation and its relation to infection and immunity.” Jour. Infect. Dis., 51: 216, 1932.
- Hoffstadt, R. E., and Youmans, G. P.: “The genetic significance of the dissociants of Staphylococcus aureus.” Jour. Bact., 27: 551, 1934.
- Pinner, M., and Voldrich, M.: “Derivation of staphylococcus albus, citreus and roseus from Staphylococcus aureus.” Jour. Infect. Dis., 50: 185, 1932.
- Haberman, S.: Personal communication.
- Chapman, G. H., Lieb, C. W., and Curcio, L. G.: “The use of bromthymol blue agar and phenol red mannitol agar for the isolation of pathogenic types of staphylococci.” Amer. Jour. of Clin. Path., Tech. Suppl., 2: 3, 1938.
- Thygeson, P.: “Mannitol fermentation as an indicator of conjunctival pathogenicity of staphylococci.” Arch. Ophth., 20: 274, 1938.
- Epstein, S.: “Untersuchungen uber die Unterscheidung der Staphylokokken bei den verschiedenen Staphylokokkenerkrankungen der Haut.” Arch. f. Dermat. u. Syph., 170: 586, 1934.
- Chapman, G. H., and Berens, C.: “Crystal violet agar as a differential medium for staphylococci.” Jour. Bact., 29: 437, 1935.
- Chapman, G. H., Berens, C., Nilson, E. L., and Curcio, L. G.: “The differentiation of pathogenic staphylococci from non-pathogenic types.” Jour. Bact., 35: 311, 1938.