Access to all articles, new health classes, discounts in our store, and more!
Karma Sickle Cell Project
Presented to Inland Sickle Cell Foundation, Riverside, California, October 20, 1980.
* * *
Introduction
Homozygous sickle cell anemia is remarkably variable in its clinical.expression; many affected individuals have frequent and severe manifestations of this genetic alteration) whereas others may have a virtually crisis-free course. Karma Miller has the former physiological state and thus has been frequently hospitalized for primary and secondary manifestations of sickle cell anemia. Karma Miller was brought to us by her parents on 21 April 1980 during the middle of her last serious crises. She was admitted to Kaiser Foundation Hospital, Fontana, CA on 22 April 1980. At that time, according to medical record, she was suffering from sickle cell crises, acute pulmonary edema and congestive heart failure, with possible osteomyelitis versus cellulitis of the left eye–no history of trauma. Since her discharge from the hospital 2 May 1980, Karma has been on a dietary supplement program which consists of unique algae-based products which were obtained from the blue-green algae Aphanizomenon flos-aquae (Klamath strain). Herein, we describe our successful results with this four-and-one-half year old black female. This effort has involved growth and harvesting of the organism, product preparation, and administration of the material by the child’s parents. The study is a continuing effort.
Experimental
Cell Culture–The blue-green algae Aphanizomenon flos-aquae were obtained from Upper Klamath Lake which is located in Klamath County, Oregon. The Klamath strain of A. flos-aquae grows abundantly in the lake during the summer months and exists in the water at concentrations of 30 to 60 mgℓ-1. The algae are removed from the lake water (Fig. 1), washed with tap water, and concentrated to a thick paste (Fig. 2). The algae are flash frozen in plastic bags (30 cm x 30 cm x 2.5 cm) and stored at -20°C.
Fig. 1. Algae harvesting on Upper Klamath Lake. The continuous belt microsieving screens are shown mounted on a support barge. Each screen removes about 40 kg of algae per h from the lake.
Fig. 2. Secondary algae concentration equipment is located onshore about 45 m from the primary dewatering equipment. Algae is pumped from the barge onto the screens where a vacuum is applied to remove additional water. The algae are removed from the micromesh screen (shown in figure), placed in plastic bags, and flash frozen.
Cell Analysis–Each harvest of algae is chemically analyzed to determine its quality and purity. Cellular protein, carbohydrate, lipid, and trace mineral content is determined by described methods and procedures.1,2 The algae were analyzed for the presence of heavy toxic metals (lead, cadmium, mercury, selenium, and arsenic). Further, the algae were analyzed for the presence of lindane, aldrin, other chlorinated hydrocarbons as p,p-DOE and PCBs (as Arochlor 1260). The analysis for the presence of microorganisms included aerobic plate counts, yeast, mold, coliform (MPN), coagulase positive staphylococcus, and salmonella.
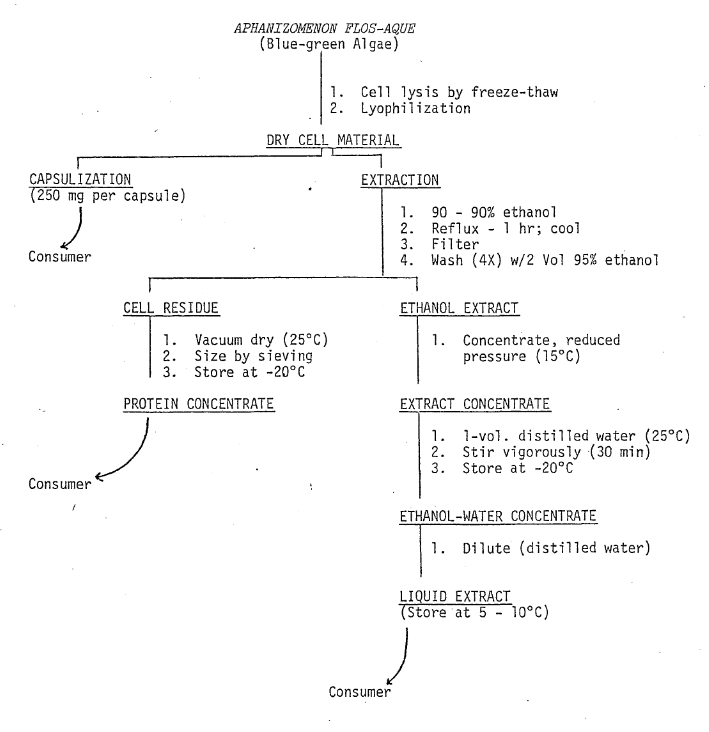
Isolation and Preparation of Materials for Oral Ingestion–The fresh frozen algae were lyophilized using a virtus U.S.M.-15 freeze-dryer equipped with stainless steel trays and automatic temperature controlling equipment which maintained the product at 15°C during the drying process. The freeze dried cell material to be encapsulated (Fig. 3) was passed through a fine mesh stainless steel screen to ensure uniform material size. The algae (250 mg per capsule) was subsequently packaged into size 0 gelatin capsules by Cosmo-Pharm, Inc. (North Hollywood, CA).
Fig. 3. Flow Diagram: Biochemical Preparation of Administered Material
Freeze-dried algae to be used for preparing protein concentrates and concentrated liquid extracts was suspended in ethanol (10:1 w/v) and refluxed. The suspension was filtered and the cell residue was washed with ethanol (Fig. 3). The cell residue was dried in vacuo (25°C), sieved, and supplied to the consumer in powder form. The alcohol extract was concentrated using reduced pressure and steam. During volume reduction, the product temperature did not exceed 15°C. After the volume of the extract had been sufficiently reduced (80 g solids ℓ -1), sterile distilled water was used to increase the volume (40 g solids ℓ -1). For consumer use, the concentrate was diluted with sterile distilled water to the desired concentration (~8 mg mℓ -1–Karma Miller).
Preparation of Material for Nasal and Bronchial Application–Liquid concentrate containing 40 g solids per ℓ of solution was diluted with buffered normal saline (pH 7) to give a final concentration of 0.4% solids. The buffered solution was filtered and stored (4°C) in sterile glass containers. For dispensing to the consumer, the solution was placed in a nasal aspirator. The aspirator is similar to those used to dispense common nasal decongestants.
Consumer Dosage–Karma T. Miller–During a severe sickle cell crisis, Karma was placed on a regimen of liquid algae concentrate which was termed Mannacol for identification. From 22 April 1980 through 1 June 1980, a dosage of one teaspoon three times daily was administered. Beginning 21 May 1980, Karma was given the specially prepared nasal/bronchial mist. She continues to use this material two to four times daily. The nasal spray was termed Mannamist for Karma’s identification. On 2 June 1980, Karma’s intake of Mannacol was increased to one teaspoon four times daily, and she was started on two 250-mg capsules of freeze-dried algae daily. The capsules were named Blue-Green Manna. On 17 June 1980, Karma’s intake of algae and algae-related material was changed to its present level which is as follows: one teaspoon Mannacol five times daily; four 250-mg capsules Blue-Green Manna; two to four applications daily of Mannamist, and four teaspoons daily of algae protein concentrate which was named Mannapro. Four teaspoons of Mannapro represent four to six grams of protein daily.
Results and Discussion
Preliminary studies of low molecular weight chemicals such as urea, potassium cyanate, carbamyl phosphate, dichloromethane, and others have been used at various times to attempt to help alleviate the painful symptoms associated with a sickle cell crisis.3,4 Several lines of evidence suggest that certain small molecules with a high affinity for specific sites in heme proteins could prove useful as anti-sickling agents. Dichloromethane, one agent of this group, at low concentrations can prevent completely and reverse partially the aggregation of hemoglobin S. It has been proposed by others that the search for useful therapeutic agents should focus on these low molecular weight non-heme ligands of hemoglobin.5 Presently, however, there is no recognized method either available now or in the testing stages that effectively alleviates the symptomology associated with sickle cell anemia.
Three approaches to the treatment of sickle cell anemia are generally used in experimental investigation; they are: replacement therapy, modification of the sickle cell or its environment, and modification of the molecular conformation of sickle hemoglobin.6 In understanding the rationale behind the administered treatment of Karma Miller, it is necessary to consider a fourth and possibly a new approach to treating sickle cell anemia; that is, the available free energy in the organism to effect a positive change and to activate the natural immunological responses. This concept of free energy is not generally applied to an entire living organism but is understood and accepted for individual chemical and biochemical processes. The relationship is defined by the following expression for the free energy change of a given process, ΔF = ΔH-T ΔS, where the free energy change is equal to the maximum useful work that can be done in excess of pressure-volume work, provided the chemical events are carried out reversibly and at constant temperature and pressure.
By understanding the concept of free energy as it is applied to an entire organism, it is then possible to understand an obscure Japanese report which describes the isolation of a specific nucleo-protein complex from Chlorella pyrenoidosa and the enhanced growth-promoting characteristic of the biological material. By stimulation of growth using this material, the investigators were able to increase the total net free energy available for useful biochemical work by the consumer organism.
The nucleoprotein complex isolated from Aphanizomenon flos-aquae is water soluble and is extracted as shown in Fig. 3. The complex has been chromatographically separated into four fractions, each of which appears to have growth enhancing effects on microorganisms. It has maximum optical absorption at 260 nm and is a mixture of sulfur-containing nucleotides and low molecular weight peptides. The nucleotides and low molecular weight peptides are complexed into sub units having various molecular weights, all of which are less than 15,000. One or more of these complexed units appears to be assimilable and transportable by the human organism. The liquid extract which was called Mannacol for identification purposes in the experimental section was administered to about 500 consumers prior to application in the Karma Miller study. Positive energy effects were observed in the pretest participants with no manifestation of negative side effects, probably due to the natural food base origin of the administered material. Analysis of the material for toxic/dangerous chemicals and foreign microorganisms indicated it was safe for human consumption.
Since we began our work with Karma Miller in late April 1980, she has not exhibited any of the complex symptoms which were common to her physiology. On 27 June 1980, a blood screening test was performed on Karma by the Inland Sickle Cell Organization. At that time, her blood had a dark red color and was thick-normal in appearance. The blood sample had the appearance of not being anemic. In the opinion of the individual performing the test, Karma was doing well and had good skin coloring. At that time, Maxine Miller informed the examining official that Karma was taking a special type of vitamin. The chronic history of nasal and bronchial congestion which has been indicative of the patient since birth has all but disappeared. Her vital signs are near normal, pulse about 60, breathing not labored. Karma has had a very mild head cold which accompanied an outbreak of chicken pox; this lasted one to two days. In June, she had what appeared to be a minor crisis when she developed an aching shoulder; this, too, disappeared quickly. Karma’s general physical appearance was reported as good. My observation is that she is a positive, radiant, and aware individual. Interestingly, she consumes her daily regimen of algal products voluntarily–as though she realizes that she has an improved energy-state. At this point, I am not aware of the status of her heart involvement. Her parents are continuing the regimen of algae materials. Evidence to date indicates that the various biological isolates used in this study should be applied to a larger experimental group for further verification of their utility in relieving the general symptomology associated with sickle cell anemia.
Implementation of Clinical Testing Program
Description–The clinical program of treatment is unusual in that it does not call for drug therapy. The substances which will be offered to homozygous and heterozygous genotypes are essentially foods, not classical medicines. To improve our understanding of the physiological and psychological benefits of these unique algae-based foods, we propose a limited group study of homozygous sickle cell patients who would ingest them just as Karma Miller has for a period of one year. During this time period, each individual would be closely monitored for his/her progress.
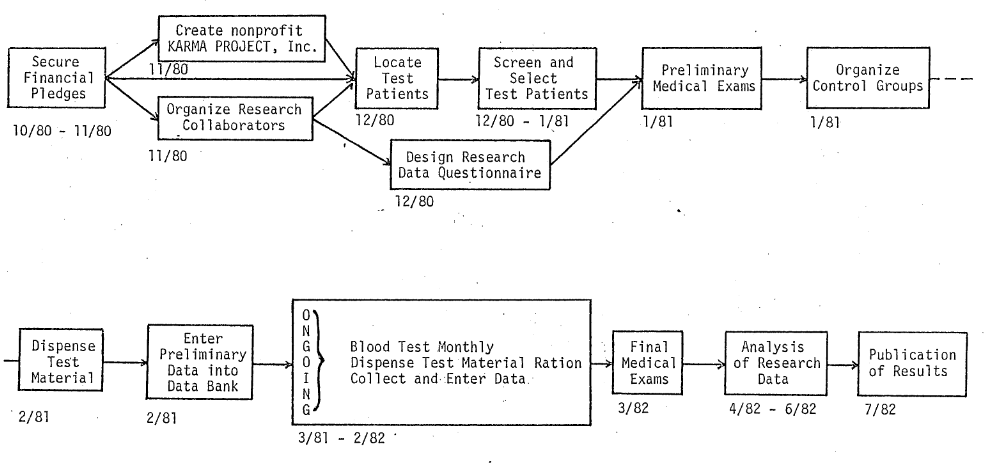
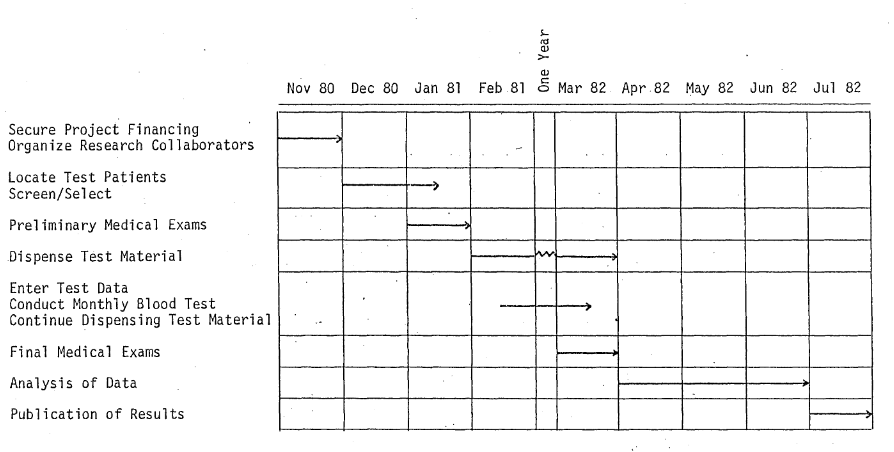
We propose to call this limited research effort “THE KARMA PROJECT” and to coordinate it with the efforts of the National Association for Sickle Cell Disease, Inc., Los Angeles, CA and the National Sickle Cell Disease Program, NIH, Bethesda, MD. THE KARMA PROJECT calls for selection of 100 volunteer sickle cell patients. These 100 “Karma” patients would be selected and screened from within the State of California, but primarily in the Los Angeles Metropolitan area. They would first be given medical examinations and the relevant medical information compiled and entered into a project computer file.
Under the supervision of our own research team, in cooperation with the local participating entity, the 100 “Karma” patients would be divided into four control groups and given adequate test materials to last for a one-month period. Each month, the patients would be given blood tests at the appropriate center and at that time, they would receive their next month’s supply of test material. Consumption levels would be varied each month depending on the patient’s progress. Clinical data on the “Karma” patients would be compiled and analyzed continuously by our research team which would be located at computer facilities in Los Alamos, NM and Los Angeles, CA. After 12 consecutive months of test food consumption and clinical monitoring, the “Karma” patients would again be given a complete medical examination and the final data compiled for review by participating groups. We anticipate a three to six month analysis period following completion of the testing period. Results would be published and distributed at that time.
Follow-up monitoring and test food distribution would continue for an additional period of time for any “Karma” patient who desires it. Judging from our experience with Karma Miller, we would be prepared for the continued use of these materials by all of the “Karma” patients.
Objectives–The set of objectives for THE KARMA PROJECT could be infinite if everyone’s desires were included. However, the proposed project budget simply does not permit an inclusive compilation; thus, only those objectives considered to be the primary target areas are described. The objectives are as follows:
- To identify differences in effect of the test food materials among different sickle cell types. Isolate areas for further research.
- To identify differences in effect among similar sickle cell types for different test food substances (i.e., the algae processed in different ways as shown in Fig. 3). Isolate areas for further research.
- To launch large-scale second-stage research to refine our understanding and to test for regional-environmental effects. This objective assumes that there will be a statistically significant success rate during the first-stage experiments. Success in this context means satisfactory control of sickle cell crises.
- We assume that the majority of the “Karma” patients will be black children. We would extend our second-stage research to sickle cell patients who are older and of other races which are subject to sickle cell disease.
- To isolate, characterize, and possibly synthesize the active constituent(s) in Aphanizomenon flos-aquae. Isolate areas for further investigation.
- To stimulate other independent research into the effects of nutrition on sickle cell disease as a result of this effort and the resulting publications.
Proposed Budget–The costs of operating this project are low relative to the value of the research results we anticipate.
Second-stage research will be considerably more expensive. By undertaking now a more limited program which can be closely monitored at a moderate expense, we will be able to carefully target research areas for the more expensive secondary work. And, we will do so in the happy knowledge that we are on track.
The first stage of THE KARMA PROJECT will cost $333,100 by our best estimates. The funds would be solicited from private sources and would be managed through THE KARMA FOUNDATION. The test materials will be provided by Kollman Consulting Laboratories at cost.
Source of Funds–We are here today to explain THE KARMA PROJECT and to ask your assistance in raising funds to make the project a reality. We intend to demonstrate to you how this project can provide the knowledge we need in order to save the lives of people and to relieve the excruciating pain of sickle cell crises, first here in our country and soon, throughout the world.
We seek the financial assistance of organizations and individuals who have a vital interest in helping to understand sickle cell anemia. Because sickle cell anemia is a disease which occurs most commonly among dark-skinned peoples, the white population generally has not perceived it to be a problem and, in many cases, have never heard of it. However, as humans, it concerns all of us when any of us must suffer–especially when the solution may be so close at hand. We will make arrangements for contributions to be processed through the appropriate agency to ensure that they qualify as charitable contributions under the U. S. Internal Revenue Code.
Publicity–In order to increase awareness of sickle cell anemia among the public, especially the dark-skinned communities, we intend to publicize THE KARMA PROJECT in both the black-oriented media and the general media. Our publicity goal is to sensitize the public to the suffering of sickle cell patients such as Karma Miller and to encourage large-scale government-supported testing programs and research efforts.
Our second publicity objective is to demonstrate that there are concerned individuals and organizations eager to help sickle cell victims who want and need such help.
We anticipate a completed project within 18 months after actual dispensing of the test material begins. Published results should be in-hand by July 1982 providing we begin soon.
The exciting possibilities that Karma Miller’s case presents make this project a must. All we need is your help.
References Cited:
- Kollman, V. H. and Fowler, E. B., “Kinetic Studies of C. pyrenoidosa Using 94% 13C CO2,” Biotechnol. and Bioeng. 14, 819 (1972).
- Milner, H. W., “The Chemical Composition of Algae, in Algal Culture from Laboratory to Pilot Plant,” ed. John S. Berlew, Carnegie Institute of Washington, Publication 600, Washington, DC (I.S.B.N. 0-87279-611-6), chp. 19 (5th printing May 1976).
- Freedman, M. L., “New Drugs in the Treatment of Sickle Cell Anemia,” Amer. J. Med. Sci. 265, 371 (1973).
- Lubin, B. H. and Oski, F. A. “Oral Urea Therapy in Children with Sickle Cell Anemia,” J. Pediat. 82, 311 (1973).
- Milosz, A. and Settle, W., “New Approach to the Treatment of Sickle Cell Anemia,” Res. Commun. Chem. Pathol. Pharmacol. 137 (1975).
- Zarkowsky, H. S., “Sickle Cell Anemia: Therapeutic Considerations,” Heart Lung 3, 962 (1974).
The Karma Project Budget–18 Months
- Scientific and Medical
- Direct Test Expense
- Medical Examinations ($265/patient)–$26,500.00
- Test Materials ($1848/patient) x 50%–$92,400.00
- Establish Patient Records ($50/patient)–$5,000.00
- Direct Test Expense
Sub Total – $123,900.00
-
- Indirect Test Expense
- Test Patient Selection–$5,000.00
- Data Processing–$5,000.00
- Personnel Services–$62,000.00
- Project Director–$18,000.00
- Research Assistant–$24,000.00
- Medical Collaborator–$10,000.00
- Consultant Fees–$10,000.00
- Indirect Test Expense
Sub Total $72,000.00
Scientific and Medical Total $195,900.00
- Administrative Overhead
- Personnel Services–$71,000.00
- Administrative Director–$36,000.00
- Financial Director–15,000.00
- Secretarial–20,000.00
- Rent ($1200/mo)–21,600.00
- Equipment–3,600.00
- Supplies and Printing–15,000.00
- Telephone–9,000.00
- Travel and Per Diem–12,000.00
- Miscellaneous, Legal, etc.–5,000.00
- Personnel Services–$71,000.00
Administrative Total $ 137,200.00
PROJECT TOTAL $ 333,100.00
Percent Scientific and Medical = 59; Percent Administrative = 41; Percent Total = 100
Flow of Project Events