Access to all articles, new health classes, discounts in our store, and more!
A Simple and Rapid Urine Test for Estimating the Severity of Chronic Infection and for Controlling the Dosage of Vaccines
Published in The Military Surgeon, Vol. 106, No. 1, January 1950. Co-Author: Captain Albert F. Griffiths, M.C., U.S.A., Ret.
* * *
In 1947 one of us (G.H.C.)1 described a simple urine test which takes about 15 seconds, uses an inexpensive and easily available reagent, and indicates the amount of soluble bacterial products (probably streptococcal) excreted by the kidneys. Because of the simplicity of the method, a compact outfit has been prepared for use in the physician’s office. If he desires, the patient can do the test himself and report the results to his physician. The reagent is added to the urine until the precipitate which forms redissolves; the amount of reagent required to reach this endpoint being directly related to the severity of the infection.
This method has been used in private practice by one of us (A.F.G.) and found extremely valuable. The present communication is a résumé of his experience with it. It is a most useful simple method, not only for detecting unsuspected chronic infection but also for detecting an acute or subacute flareup of an infection, for investigating diseases of unknown origin, and particularly for following response to treatment. The method is so reliable that it can be used also to adjust the dosage of vaccine (provided that the “vaccine” is a specific antigen) so that the optimum dose and interval can be found quickly. In the two years that the method has been in use much has been learned about it and its limitations. A complete presentation2 will be submitted for publication late this year.
Method
The reagent consists of a 1.0 per cent aqueous solution of alkyl dimethyl benzyl ammonium chloride in which the alkyl chain has between 8 and 18 carbon atoms. In the laboratory, 1.0 ml. of urine is measured into a small test tube and the reagent is added from a 1.0 ml. pipette graduated in 0.01 ml. A precipitate forms, gradually increases in density, and then begins to dissolve. At this point the reagent is added cautiously with constant shaking until the mixture is opalescent. The volume in the pipette is noted and an additional drop or two of reagent is added until the opalescence is not reduced any further. Each 0.01 ml. of reagent used to reach this endpoint is considered provisionally as one unit. In case the endpoint is difficult to determine, either of the following methods may be used: (1) a slight excess of reagent may be added to one tube and a second tube titrated until the turbidity is reduced to that of the first tube, (2) a piece of “Q-Sharp test paper number 11”10 can be dropped into the urine and the reagent added until the paper turns pink.
Correlation with Other Laboratory Tests for Infection
It is well known that the blood sedimentation rate may be within normal limits even though the patient shows evidence of infection. Similarly, the blood count, which when properly done1,3 is one of the most reliable indicators of the severity of infection, may not in all respects reflect it accurately. Likewise, the urine titration method, which is positive (i.e., above 10 units) in 87 per cent of chronic invalids (who probably have a basic streptococcal infection in most instances9) does not in every case reflect the true degree of infection (or more probably the degree of streptococcal infection). Therefore, a close correlation cannot be expected among these tests. However, there is a good general relationship between the urine titer and other methods, as will be shown elsewhere.2
Fluctuations
If the titer is determined every time a person voids, it will be found that it varies at different times. Although the titer fluctuates, usually within rather narrow limits, a few persons show considerable differences. These fluctuations are caused by fleeting changes in the severity of infection. They vary inversely as the volume of urine excreted, which suggests that the bacterial products may be a major factor in controlling the output of urine. It is because of these variations that it is important, for comparative purposes, to make many control tests and calculate the average. Unless this is done, fluctuations following response to treatment may be difficult to interpret. Similar unexplained variations have been observed, for example, in blood counts. That they are not artefacts is shown by the facts that the titer is directly related to the color and specific gravity of the urine and is inversely related to its volume.
Nature of the Excreted Material
The precise nature of the bacterial products is unknown but they appear to consist mainly of mixtures of purines (probably from nucleic acid anabolism), precipitated reagent and bacterial nucleoproteins. The latter are relatively insoluble in the reagent and are responsible for the residual turbidity in the titrations. Usually, the higher the titer the greater the residual turbidity, and the deeper brown the urine.
Another curious property of these bacterial products is their antibacterial activity. The higher the titer the longer the urine can stand without showing signs of bacterial growth. Chapman and Liebb5,6 and Rawls and Chapman11 showed that, in the absence of achlorhydria, the number of coli group organisms in the feces is inversely related to the severity of upper respiratory streptococcal infection. Undoubtedly, the streptococcal metabolites are swallowed and, if coliforms are inhibited by gastric acidity, the streptococcal metabolites can prevent the coliforms from proliferating in the intestinal contents. It is probable that the substances responsible for the antibacterial activity of high titer urine specimens and for the inhibition of fecal coli are identical.. Another phase of the phenomenon is illustrated by the low urine titers of bacterial products in coli bacilluria. The coli group organisms are able to grow in the urine because of the relative absence of the bacterial (probably streptococcal) products. Some of the excreted soluble bacterial products may be nucleic acids in various stages of degeneration. Yeast nucleic acid itself is bactericidal, even for streptococci. This agrees with the observation5 that, in severe streptococcal intoxication involving the gastrointestinal tract, along with almost complete absence of coliform bacteria, there is almost a complete absence of streptococci in the feces.
Probable Significance of the Excreted Bacterial Products
Normally, a person in good health is able to prevent toxigenic bacteria from invading his body: or if they do penetrate the mucous membranes, the bacterial products are rapidly destroyed, most frequently by oxidation or conjugation as shown by the improvement when thyroid hypofunction and liver dysfunction are corrected. Under these circumstances, the urine shows a titer of less than 10 units. However, when the body defense is low, bacteria or their products are able to invade it. If the ability of the tissues to destroy bacterial products is unimpaired there will be very little of the bacterial products left and only traces (from 0 to 10 units) will appear in the urine. However, if the body is unable to convert the absorbed bacterial products into its own economy or if it is unable to destroy them, they are excreted in the urine and can be estimated by the urinary titration test under discussion. Under these conditions the titer rises and may reach 200 units or more. The method, therefore, is not a measure of the amount or severity of infectious material in different parts of the body but of the excess of-bacterial (streptococcal) products over the bacterial product destroying power of the body. Consequently, the reaction may be negative in some types of infection, particularly those that are localized (“walled off”). The titer, therefore, is a simple method for measuring the excess of circulating bacterial products (probably derived from the more pathogenic types), and consequently is extremely useful in screening chronic invalids.
Fluctuations of the Urine Titer in Disease
The titer of bacterial products in the urine rises sharply during colds7 and other acute infections, and in headaches. It is reduced by the administration of leukocyte stimulating agents.7 It responds rapidly to changes in severity in acute illnesses and might possibly be used in following the progress of such acute conditions as appendicitis. By testing consecutive samples of urine immediately (which can easily be done in 15 seconds by any operating room attendant) much time can be saved compared with the more than 15 minutes required for a leukocyte count. Since the titer rises sharply during acute illnesses it can be used effectively to follow the progress of treatment and reduces the delay waiting for laboratory reports. It is possible that disastrous conditions like poliomyelitis might be prevented if patients were to use the urine test daily and report high titers to their physicians as is being done successfully by Dr. C. M. Longstreth of Atlantic, Iowa.12
Value of the Urine Titer in Adjusting the Dosage of Vaccines
Because of extremely poor results with usual methods of “vaccine” therapy, one of us (G.H.C.) suggested that the urine titer might be used as a guide to dosage. To be successful, the “vaccine” should be a highly specific antigen.1,4,8 A few persons respond readily to nonspecific antigens but most chronic invalids, we have found in 24 years of intensive study, require highly specific, logarithmic phase antigens derived from toxigenic bacteria. First results were poor, but as the antigens became more specific the proportion of excellent results increased until, at the present time, about 90 per cent of the patients get excellent results. Some of the causes of failure were discussed by Stiles et al.4
The initial dose of antigen is estimated from the blood sedimentation rate and the urine titer and from the total number of filamented neutrophiles (normal 3,600 per cu mm1). The higher the first two and the lower the third the heavier the toxic load on the body, and the smaller should be the initial dose after reducing the toxic load as much as possible by treatment of apparent foci of infection.
In most instances after a focus of infection has been treated or eliminated, there is a sharp rise in the urine titer caused by manipulation of the focus which sets free bacteria or their toxins. Vaccine treatment must not be given until the elevated titers have dropped to the average level before the focus was treated. In a few cases the titer will be lower after treatment of the focus and the vaccine injections should not be given as long as the titers continue to drop, indicating progressively improved immunity.
After the control period in which the urine is tested at least once daily for a week and the average titer calculated, or after the post-surgical titers have become stabilized as just mentioned, the patient is given the estimated initial dose of specific antigen and instructed to continue the urine titrations and report to the physician a week later. If the titers show no change (and usually there are no changes in symptoms either) the dose was too small and should be increased. If the titers are lower after the injection, it was close to optimum, depending on how long and how low they were. The greater the reduction and the longer it lasted the closer the dose was to the optimum. If, on the other hand, the titers increased after the injection, the dose was too large and the next one must not be given until the titers return to their former level, no matter if it should take several months. No matter how small the next dose, if it is given before the titers reach the pre-dose level, it will likely produce an unfavorable, sometimes alarming result, due to overcoming the immune mechanism. Once the optimum dose has been determined the optimum interval can then be determined by observing how long the reduced titers remain low.
Examples of Application of the Urine Titrations to the Administration of Bacterial Antigens
The following examples illustrate the application of the method in adjusting the dosage of vaccines.
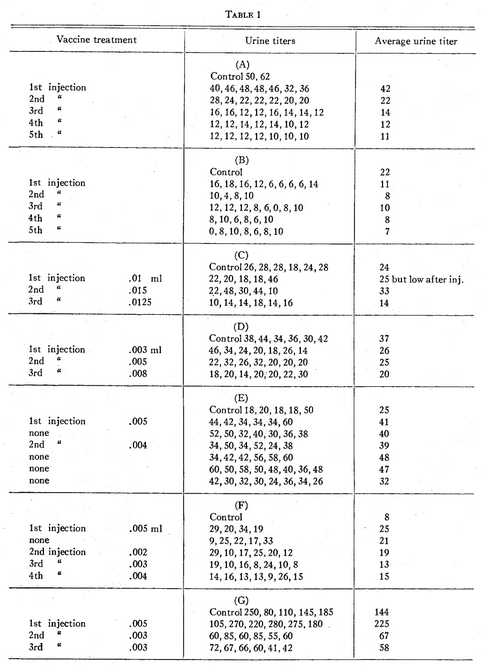
Patient P.H. (courtesy of Dr. Frank C. Combes, New York City) had dermatitis herpetiformis. Weekly injections of the specific bacterial antigen (Table 1A) brought the titers down from two individual control tests of 50 and 62 units to consecutive weekly averages of 42, 22, 14, 12, and 11, which were accompanied by progressive clinical improvement.
Patient M.B. (Table 1B) had a syndrome which we have come to associate with chronic bacterial intoxication (excessive lassitude, gastrointestinal disturbances, postnasal drip, sinusitis and autonomic dysfunction). Response was dramatic along with a gradual reduction of the urine titers.
G.W.C. had shown urine titers of 9, 11, 10, 18, 17, 7, 5, and 7. When he developed acute arthritis the titers were increased to 62, 46, 77, 84, 25, 17, 48, 51, 69, 52, 39, and 51.
F.H.H. (Table 1C) had osteoarthritis. The initial dose of antigen caused a drop in the urine titers for 4 days. The dose was increased 50 per cent and there was an increase in the average titer with some increase in symptoms. The third dose was a 25 per cent increase over the initial one and was followed by a considerable drop in titers and considerable relief from pain and stiffness.
P.M. (Table 1D) was given an extremely minute initial dose because of other findings, asthenia and a desire to avoid stirring up the multiple sclerosis. Cautious increases brought about a gradual decrease in the urine titers, and excellent symptomatic improvement.
J.P. (Table IE), with catarrhal deafness, is an example of a too early repeated, although slightly reduced, dose that caused an elevation of urine titers for several weeks along with an increase in deafness. Subsequently, when the urine titers had dropped to a low level, a smaller dose of antigen produced improvement in titers and deafness.
F.P. (Table 1F), who was difficult to control because of marked vegetative neurosis, responded unfavorably to the initial dose of antigen. After omitting the injection the following week, smaller, infinitesimal doses brought about progressive decreases in the urine titers and clinical improvement.
A.S. (Table 1G), with severe dermatitis bullosa and extremely high urine titers, had an exacerbation of symptoms from the initial dose and considerable increase in the urine titers. A slight reduction in the dose was followed by prompt clinical improvement and reduction in the urine titers.
Some patients with chronic infection are extremely sensitive to “vaccines” and get decidedly unfavorable results. The fault is largely with the interval, which in most instances is too short to allow for conversion of the antigen into antibody, and the residual (unconverted) antigen acts as a toxin. When this is repeated several times the accumulated antigen is sufficient to be a severe drain on the immune mechanism and is productive of distressing results. When the urine test is used, any unfavorable result is detected at the start and the optimum dose and interval can be readily determined.
Summary and Conclusions
When a 1.0 per cent solution of alkyl dimethyl benzyl ammonium chloride is added to urine from a person with certain types of infection, it produces a precipitate which, on further addition of the reagent, becomes increasingly turbid and then gradually clears leaving a slightly opalescent solution. The amount of reagent required to reach the latter endpoint is an indication of the severity of undestroyed absorbed bacterial products (probably streptococcal) in the body.
The titer of these bacterial products in urine changes rapidly with changes in the degree of infection. This makes the method extremely useful (1) as a simple office or bedside procedure for estimating the severity of infection, (2) as an indicator of the progress of acute and subacute infections, (3) as a guide to the progress of treatment of chronic or recurrent infections, and (4) to adjust rapidly the dosage of “vaccines” so that the optimum can be found quickly.
References Cited:
- Chapman, G. H. “Relationships of nonhemolytic and viridans streptococci in man.” Trans. N.Y. Acad. Sciences, 10:45, December 1947.
- Chapman, G. H. “Studies in chronic disease. Excretion in urine of substances resulting from increased bacterial activity in the body. A simple method for their measurement and a discussion of their significance in chronic infections.” Unpublished manuscript.
- Yetwin, I. J. “A new diluent for the enumeration of leukocytes.” Military Surgeon, 104:286, April 1949.
- Stiles, M. H., Berens, C., Rawls, W. B. and Chapman, G. H. “Attempts to obtain better results with the bacterial antigen (‘vaccine’) therapy of low grade chronic (‘focal’) infection. 1. Possible errors of usual methods.” Jour. Lab. & Clin. Med., 28:1447, September 1943.
- Chapman, G. H. and Lieb, C. W. “Bacteriology of the intestinal tract in certain chronic diseases. II. The possible inhibition of colon bacilli by pathogenic streptococci and staphylococci.” Rev. Gastroent., 5:234, September 1938.
- Lieb, C. W. and Chapman, G. H. “Bacteriology of the intestinal tract in certain chronic diseases. III. The possible role of upper respiratory infection.” Rev. Gastroent., 5:306, December 1938.
- Griffiths, A. F. and Chapman, G. H. “Effect of leukocyte stimulating agents on symptoms of the common cold.” Unpublished manuscript.
- Chapman, G. H. “The staphylococci.” Trans. N.Y. Acad. Sciences, 9:52, December 1946.
- Stiles, M. H. and Chapman, G. H. “Probable pathogenic streptococci and staphylococci in low grade chronic illness.” Arch. Otolaryngol., 31:458, March 1940.
- Cargille Scientific, Inc. New York 6, N.Y.
- Rawls, W. B. and Chapman, G. H. “Effect of gastric HCl and certain other factors on the number of colon bacilli in feces.” Rev. Gastroent. 6.:317, July-August 1939.
- Personal communication.