Access to all articles, new health classes, discounts in our store, and more!
A Study of the 1946 Poliomyelitis Epidemic by New Bacteriologic Methods
Published in The Journal-Lancet, Minneapolis. July 1948, Vol. LXVIII, No. 7, pp. 265–277.
* * *
A review of results of previous studies made by others and myself on the importance of a specific type of streptococcus in the etiology of poliomyelitis, and by myself on the relation of the streptococcus to the filterable virus and references to the original reports have been set forth in a monograph.1
Owing to the widespread occurrence of poliomyelitis during the summer of 1946, the exhaustion of the supply of the poliomyelitis antistreptococcus serum with which diagnostic and curative action had been obtained,1 and because of the encouraging results I have had in the preparation and use of artificial antibody2,3 in the diagnosis and treatment of other diseases due to or associated with specific types of alpha streptococci, I could not do otherwise than again study this still unsolved problem from the streptococcal and viral standpoint by the use of new methods.
Persons suffering from poliomyelitis studied by me in 1946 were being cared for in hospitals in Cincinnati, Ohio, Madison, Wisconsin, Rochester, St. Paul and Minneapolis, Minnesota, Rockford and Chicago, Illinois, and Cleveland, Ohio. The diagnosis was made by the respective attending physicians.b
Methods
Serial dilution cultures4 were made in dextrose brain broth from washings of nasopharyngeal swabbings of persons suffering from poliomyelitis, from contacts and non-contacts, and from NaCl solution washings of bronze turnings which had been exposed to outdoor air in glass tubes with screened ends on the hood of an automobile driven from 50 to 300 miles for different samplings in and Old Side of the epidemic zone.7
The swabbings of the nasopharynx were made from behind and above the soft palate without touching the tongue with aluminum wire wrapped swabs bent to a suitable angle. The material thus obtained was washed off in 2 ml. of a 0.2 per cent Gelatin-Locke solution and 0.15 ml. of the washings was inoculated with a 1 or 2 ml. pipette into the first of a series of six tubes, each containing 15 ml. of dextrose brain broth 0.15 ml. was then transferred and thoroughly mixed with the same pipette in rapid succession from tube to tube through the series. A like amount of the undiluted saline washings was inoculated on the surface of a blood agar plate. All cultures were incubated at 33° to 35°C.
Pure cultures of the streptococcus were selected for study, not from colonies of the primary growth on blood agar plates but from the end point of growth of serial dilution culture in dextrose brain broth. This usually occurred in the fourth, fifth, or sixth tube of the serial dilutions from nasopharyngeal swabbing and in the second to the fourth dilutions from the washings of the bronze turnings which had been exposed to outdoor air.
Mice were inoculated intracerebrally in the right frontal lobe while under ether anesthesia with 0.03 ml. of 10 to 1 NaCl solution suspensions of the primary pure culture of the streptococcus from the end point of growth in the serial dilution cultures in dextrose brain broth, and large volumes 3500 ml. of freshly prepared 0.2 per cent dextrose broth contained in bottles of 4 liter capacity were inoculated usually with the second or third subculture. The young 4 to 8 hour culture in tubes of dextrose brain broth containing 15 ml. Was inoculated into each of the large volumes of dextrose broth. After incubation, usually overnight, the growth was harvested in the bowl of a continuous feed super centrifuge and the putty-like mass of the centrifugated organisms from the inside of the bowl was suspended in glycerol 2 parts and saturated NaCl solution 1 part, so that each ml. contained approximately 1000 billion organisms, or the growth from 500 ml. of dextrose broth.
Two types of artificial antibody were used in cutaneous and agglutination tests and for therapeutic injection. One was prepared by autoclaving at 17 lbs. pressure, NaCl solution suspensions containing 20 billion streptococci per ml. for 96 hours,2 the other by autoclaving suspensions containing 10 billion streptococci per ml. but for 1 to 3 hours after adding 1.5 per cent H2O2.3 For cutaneous tests the former was diluted with an equal volume of NaCl solution containing 0.4 per cent phenol, and the latter was used undiluted after neutralizing with sodium hydroxide and adding 0.2 per cent phenol. The former type of antibody was used routinely in cutaneous tests. The suspensions from which antibody was prepared were made from streptococci preserved in the dense suspensions in the glycerol-NaCl solutions menstruum.
Studies on the in vitro production and nature of artificial antibody from NaCl solution suspensions of streptococci by the application of prolonged heat in the autoclave and much less heat after adding oxidizing agents such as H2O2 or O2 under 2000 lbs. pressure have shown that the organisms disintegrate, the remnants became sharply agglutinated, toxic components diminished or disappeared, and substances resembling specific antibodies appeared in solution. Hence the bacteria-free supernatant of the heated suspensions will be designated as thermal or artificial antibody.
The neutralizing action of artificial antibody on poliomyelitic virus, virulent for mice, was studied by the methods usually employed. Four parts of centrifugated 5 per cent emulsions of the spinal cord and brain of mice that had succumbed to poliomyelitis were mixed respectively with 1 part of undiluted antibody, freshly prepared with H2O2 and heat, and as a control with 1 part of NaCl solution. The mixtures were incubated at 37°C for 1½ hours, kept in the refrigerator overnight, and 0.03 ml. inoculated intracerebrally under ether anesthesia far forward in the right frontal lobe. Three highly virulent virus strains were used: I am indebted to Dr. Charles Armstrong, of the National Institute of Health, Washington, D. C., for the strain of virus he adapted to the mouse and to Dr. C. W. Jungeblut of New York City for two murine strains isolated by him.
After it was found that parallel reactions were obtained on intradermal injection of natural antibody consisting of 10 per cent solutions of the euglobulin fraction of the serum of horses that had been immunized with the streptococcus and artificial antibody and that late reactions due to sensitization did not occur following injection of the latter, artificial antibody was used routinely for the detection of specific streptococcal antigen in skin or blood. Approximately 0.03 ml. was injected intradermally as superficially as possible about 4 cm. apart, usually over the volar aspect of the forearm. A sharply blanched bleb 5 mm. in diameter was taken to indicate that the right layer and the right amount was injected. An immediate erythematous reaction at the site of intradermal injection of antibody was taken as indicating the presence of antigen in the skin or blood immunologically related to the antigen or streptococcus from which the injected antibody was prepared, resembling the Foshay phenomenon discovered in tularemia.5 Antibody prepared from streptococci isolated from nasopharynx of well persons, of persons ill with diseases other than poliomyelitis remote from epidemic poliomyelitis, and NaCl solution containing 0.2 per cent phenol were injected as controls.
A similar immediate erythematous reaction occurred if uncombined antibody was present in skin or blood following intradermal injection of solutions of specific polysaccharide or streptococcal antigen in the bacteria-free supernatant of NaCl solution suspensions containing 10 billion streptococci per ml. after heating at 65° or 70°C. for one hour. This reaction was taken to indicate antibody in skin or blood specifically related to the streptococcus from which the injected antigen was prepared, resembling the Francis test for pneumococcal antibody in pneumonia.6 Of the two materials containing antigen, that in the supernatant of the heated suspensions proved the more satisfactory and hence was used routinely for detecting specific antibody in skin or blood. The maximal erythema following injection of antibody and antigen was outlined with pen and ink and the size in square centimeters was determined and recorded by superimposing circles of predetermined size on a transparent card 10 x 15 cm. in size.
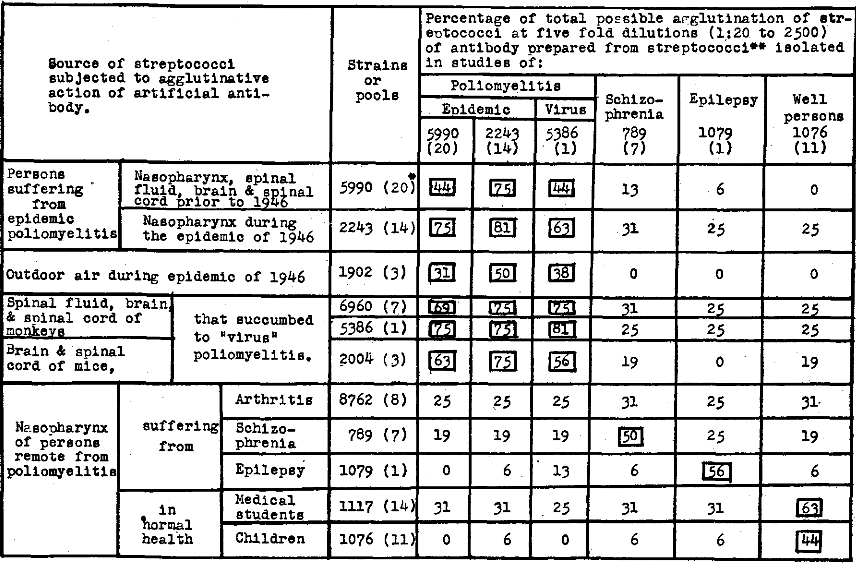
The agglutinative titer of natural antibody in the serum from immunized horses and artificial antibody was determined at five-fold dilutions of 1-20 to 1-2500, and that of serum of persons before and after subcutaneous therapeutic injections of artificial antibody was determined at five-fold dilutions of 1-10 to 1-1250. The dilutions of artificial antibody and serums and of suspensions of streptococci were made in NaCl solution containing 0.2 per cent phenol. Two-tenths ml. of the dilutions of antibody or serum and 0.2 ml. of the suspensions containing approximately 5 to 6 billion streptococci per ml. were added to test tubes 3 x 3/8 inches. The mixtures were thoroughly shaken and incubated at 48° to 50°C. for 18 hours. Readings were made under the edge of the shade of a 100-watt light bulb against a nonreflecting black velvet background. The degree of agglutination for the four dilutions was recorded according to the usual scale of 0 to 4 plus. In order to make comparisons readily possible and to avoid needless repetition of figures in tables and charts, the total possible agglutination for the four dilutions was determined in percent. Thus, a 4+ agglutination for each of the four dilutions would be 16/16 or 100 per cent. If the total observed agglutination was 7+, the percent of the total possible would be 7/16 or 44 per cent, and so forth.
Results of Cultures of Precipitation Tests and of Inoculation of the Streptococcus in Mice
Cultures from the NaCl solution washings of nasopharyngeal swabbing on blood agar uniformly revealed large numbers of colonies of greening or alpha streptococci from all of 37 persons suffering from poliomyelitis in 30 contacts and 32 non-contacts. Usually no —or only occasionally a few— hemolytic colonies of streptococci and relatively few colonies of micrococcus catarrhalis and staphylococci grew.
Short chained streptococci of the viridans type of morphology were always found in greatly predominating numbers in the first tube of the serial dilution cultures in dextrose brain broth and nearly always in pure culture at the endpoint of growth. Platings of the growth at the endpoint always yielded green producing or alpha streptococci and almost always in pure culture. In these, hemolytic streptococci were not obtained.
Cultures and precipitation tests were made according to the methods previously described from nine samplings of outdoor air.7 Five of these were made in the general region where poliomyelitis occurred but not in epidemic proportions and four in the heart of the epidemic in Minneapolis and St. Paul, Minnesota, and surrounding environs. The average distance driven for the five samplings was 198 miles, for the four samplings 75 miles, a total of 1290 miles. Streptococci were isolated from the endpoint of growth of serial dilution cultures in dextrose brain broth that had been inoculated with centrifuged sediment of the NaCl solution washing of the exposed bronze turnings in seven of the nine samplings, and streptococci grew in dextrose brain broth into which was planted the exposed bronze turnings in two of the five and in all of the four samplings. Each of the four strains of streptococci isolated from the four samplings made in the heart of the epidemic and two of the five strains isolated remote from the epidemic were agglutinated specifically by artificial antibody and immune horse serum prepared from streptococci isolated in studies of poliomyelitis and by convalescent serum. Cultures made after the samplings had been kept at room temperature for one month yielded essentially the same results, while cultures made 10 months later proved free from streptococci.
Interface precipitation tests made with the NaCl solution washing of the exposed bronze turnings and the “poliomyelitis” antistreptococcal horse serum were positive for each of the four samplings made in the heart of the epidemic and negative for the five samplings made remote from the epidemic. Precipitation tests made with the pooled serums of persons in whom reactions indicating antibody were low were negative for all samplings. While with the pooled serums of persons whose reactions indicating antibody were very high following therapeutic intramuscular injection of artificial antibody, precipitation at the interface was positive for three of the four samplings made in the heart of the epidemic and negative in the rest.
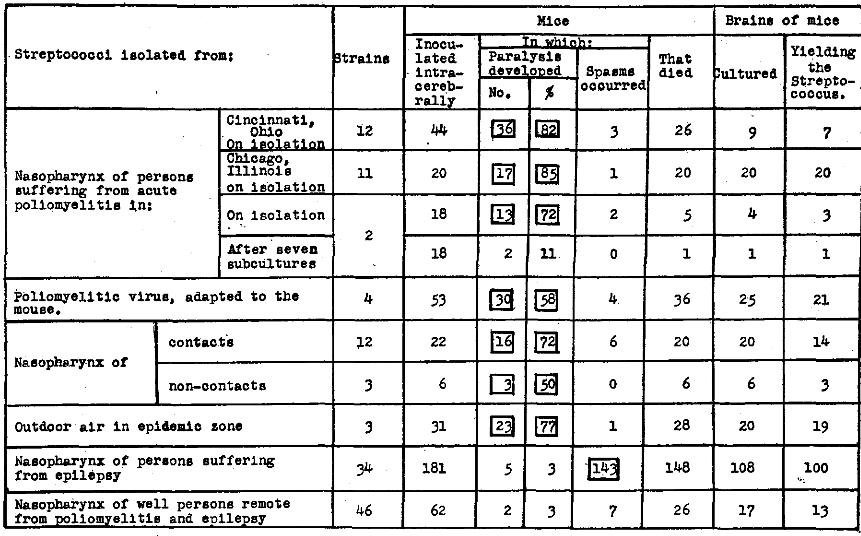
The marked tendency of streptococci, freshly isolated in studies of poliomyelitis, to produce flaccid paralysis in monkeys, rabbits, and guinea pigs on appropriate inoculation has been reported.1 The effect of intracerebral inoculation into mice of strains freshly isolated from persons ill, from contacts and non-contacts, from poliomyelitic virus virulent for mice, and from outdoor air in the epidemic zone, was studied and is summarized in Table 1. Paralysis, usually flaccid in type, developed in high incidence in the mice receiving streptococci freshly isolated from nasopharynx of persons suffering from poliomyelitis, from the “virus” virulent for mice, from contacts and non-contacts, and from outdoor air in the epidemic zone. Thus, of a total of 47 strains inoculated into 194 mice, paralysis developed in 138 or 71 per cent and spasms were seen to have occurred in only 17 or 9 per cent. After cultivation of two of the strains on the usual type of mediums for only a short time, the incidence of paralysis dropped from 72 per cent to 11 per cent.
Table 1. Incidence of flaccid paralysis in mice following intracerebral inoculation of streptococci isolated in studies of epidemic poliomyelitis, 1946.
(The squares in this and the following tables are used to indicate specificity.)
Thirty-four strains of streptococci isolated in studies of epilepsy were inoculated in like manner into 181 mice (Table 1). In only 5 or 3 per cent did paralysis develop, and convulsive seizures developed in 143 or 79 per cent. Paralysis developed in only 2 mice or 3 per cent and spasms were seen to have occurred in 7 or 11 per cent of the 62 mice inoculated with 46 strains of streptococci isolated from the nasopharynx of well persons remote from poliomyelitis.
In accord with previous studies, pleomorphic diplococci sometimes in short chains were found after special staining in or at the periphery of the lesions in the spinal cord in each of six persons who had died of acute poliomyelitis.
Neutralization of Virus by Artificial Antibody
Since the streptococcus isolated from the brain and spinal cord of mice that succumbed to poliomyelitis following inoculation of virus was agglutinated specifically by artificial antibody prepared in vitro and natural antibody prepared in horses with the streptococcus isolated in studies of epidemic poliomyelitis and by convalescent human serum, and since it produced paralysis in mice in high incidence, the action on virus, virulent for mice, of artificial antibody was studied.
Three solutions of artificial antibody were used. All were freshly prepared, two from streptococci isolated in studies of epidemic poliomyelitis, one in previous epidemics and the other in the epidemic of 1946, and one from the streptococcus isolated from natural poliomyelitic virus that had been passed successively through monkeys for many years, the Flexner MV strain.
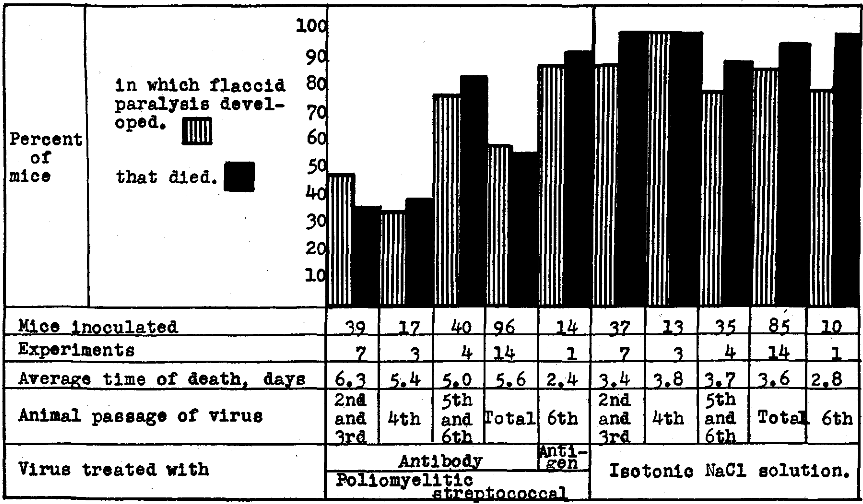
The neutralizing action of solutions of artificial antibody in 14 different experiments on a large number of mice was comparable for the three solutions and is summarized in Figure 1. It will be seen (1) that the incidence of flaccid paralysis and of deaths was far less and the average duration of life significantly longer after inoculation of virus that had been treated with antibody after the second or third animal passage than following control inoculations of virus that had been treated in NaCl solution; (2) that the neutralizing action of antibody became progressively less after additional passages of the virus through mice, and that the supernatant of NaCl solution suspensions, or antigen, corresponding to suspensions from which antibody was prepared had no neutralizing action. The somewhat irregular results in neutralization experiments with artificial antibody in these experiments were similar to those on the neutralization of virus virulent for monkeys with convalescent serum reported by others.8
Figure 1. Neutralization of virus virulent for mice by artificial antibody prepared from streptococci isolated in studies of epidemic poliomyelitis and experimental (virus) poliomyelitis in monkeys.
Results of Cutaneous Tests for Specific Streptococcal Antigen and Antibody
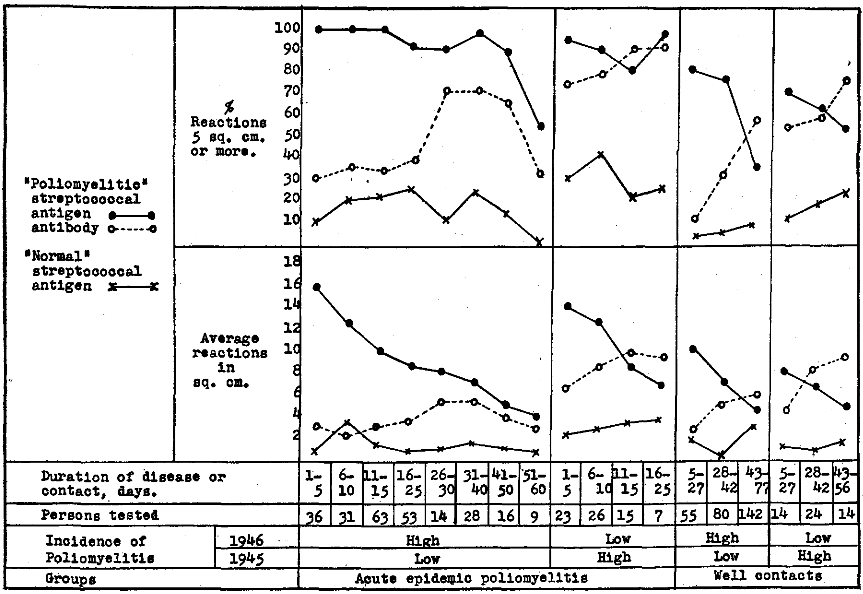
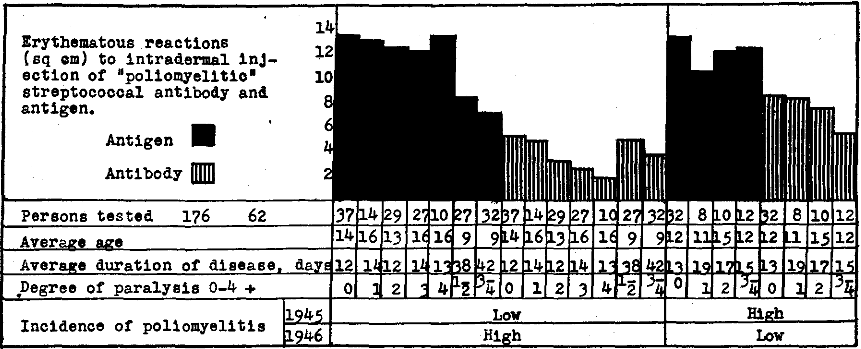
In Figure 2 are depicted graphically the results obtained following intradermal injection of “poliomyelitic” and “normal” streptococcal antibody and “poliomyelitic” streptococcal antigen in altogether 250 persons suffering from poliomyelitis, in 277 well contacts in regions where the incidence of poliomyelitis was low in 1945 and moderately or very high in 1946; in 71 persons suffering from comparatively mild attacks of poliomyelitis and in 52 well contacts in regions where the incidence was high in 1945 and low in 1946. It will be seen (1) that the average size of the reaction in square centimeters and the reactions 5 sq. cm. or more to intradermal injection of antibody, indicating antigen, was highest at the onset of the disease in regions where the incidence of poliomyelitis was low in 1945 and high in 1946 and next highest in regions where the incidence was high in 1945 and low in 1946, and that the reactions became gradually less with time as recovery ensued, far more slowly in the former than in the latter group; (2) that reactions indicating antigen in well contacts soon after exposure, while lower than in persons stricken, was highest in regions where the incidence of the disease was low in 1945 and high in 1946 and that reactions indicating antigen diminishes with prolonged contact; and (3) that reactions to control antibody prepared from streptococci isolated from the nasopharynx of well persons remote from poliomyelitis were uniformly low and had little or no relation to the disease nor to contact.
Figure 2. Erythematous reactions to intradermal injection of “poliomyelitic” and “normal” streptococcal antibody, indicating antigen and “poliomyelitic” streptococcal antigen, indicating antibody in skin or blood of persons suffering from epidemic poliomyelitis and of well contacts during the epidemic in 1946 in relation to the incidence of poliomyelitis in 1945.
Reactions to intradermal injection of antigen indicating specific streptococcal antibody, were lowest early in the course of the disease and soon after contact, far lower in regions where the incidence of poliomyelitis was low in 1945 and high in 1946 than in regions where the incidence was high in 1945 and low in 1946, and increased more slowly in the former than the latter groups with recovery or with prolonged contact. Since the cutaneous reactions indicating streptococcal antigen were highest at the onset of poliomyelitis and gradually diminished as recovery ensued, and since the reactions indicating antibody were low at the onset of the disease and increased as recovery occurred, the reactions to antibody and antigen were summarized according to age in persons suffering from poliomyelitis.
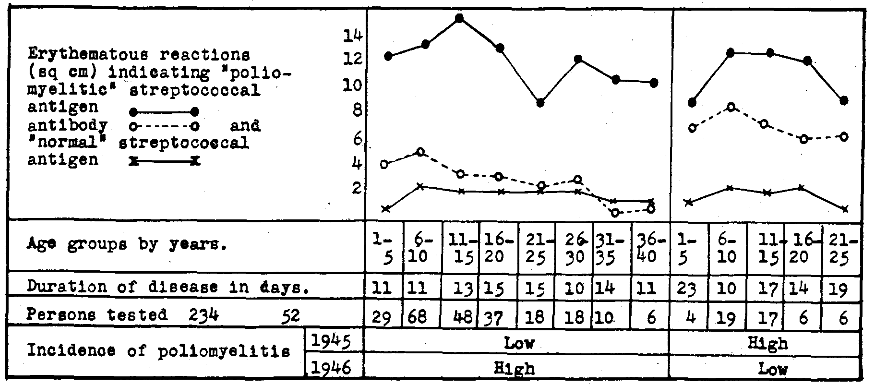
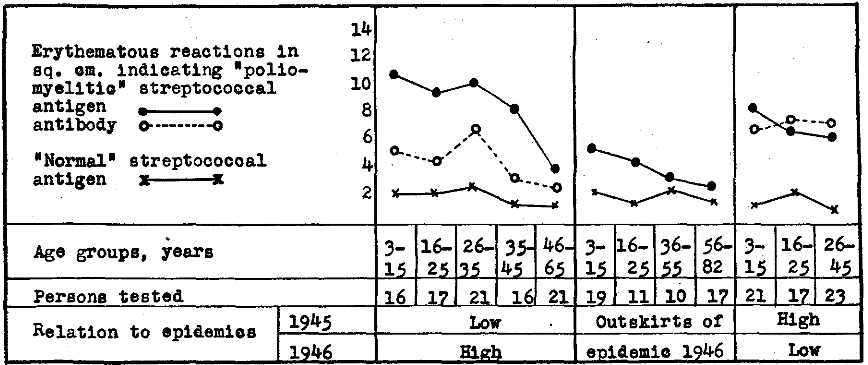
A summary of such reactions are depicted graphically in Figure 3. It will be seen that the reactions indicating streptococcal antigen were highest in age groups most susceptible to poliomyelitis, both in regions where epidemic poliomyelitis had and had not occurred in 1945.
Figure 3. Erythematous reactions to intradermal injection of “poliomyelitic” and “normal” streptococcal antibody indicating antigen and “poliomyelitic” streptococcal antigen indicating antibody in skin or blood of persons suffering from epidemic poliomyelitis according to age in relation to the incidence of poliomyelitis in 1946 and 1945.
Reactions indicating antibody were far lower throughout the different age groups where poliomyelitis had not occurred or occurred in low incidence in 1945 and in high incidence in 1946 than where the incidence of poliomyelitis was high in 1945 and low in 1946. Moreover, in both regions antibody was highest in the very age groups where mortality to poliomyelitis is low, and diminished with increasing age, corresponding roughly to the increase in mortality rate according to age. The reactions to “normal” streptococcal antibody were minimal and bore no relation to age.
As shown in Figure 4, the cutaneous reactions indicating streptococcal antigen in skin or blood in well noncontact persons within epidemics and at the outskirts of epidemics paralleled roughly susceptibility to poliomyelitis according to age. Well persons who resided in Rockford, Illinois, during the epidemic in 1945 and the slight recurrence in 1946 showed less antigen and much more antibody in skin or blood than well persons who resided in regions where the incidence of poliomyelitis was low in 1945 and high in 1946.
Figure 4. Erythematous reactions indicating “poliomyelitic” streptococcal antigen and antibody in skin or blood of well non-contact persons according to age who resided within or at the outskirts of the epidemic of 1946 and well non-contacts who resided in Rockford, Ill. during the severe epidemic of 1945 and the slight recurrence in 1946.
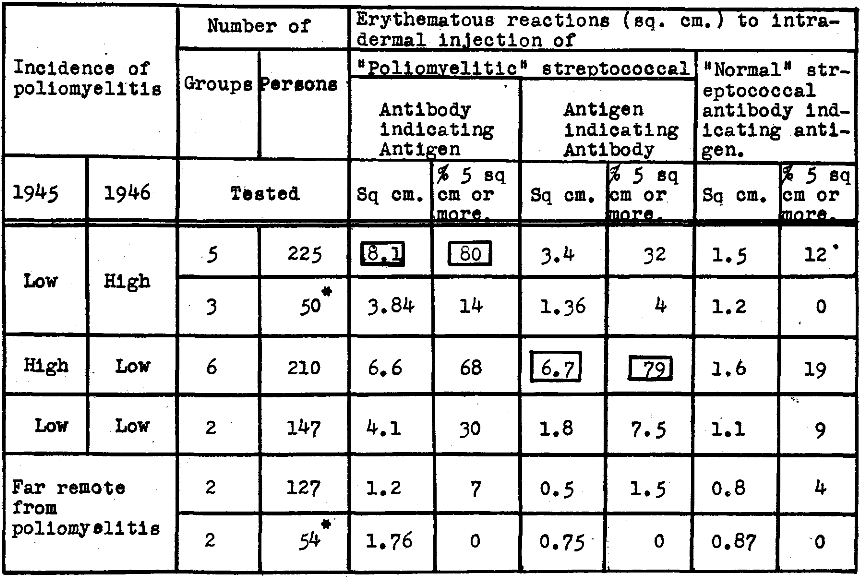
The results of cutaneous tests made on well non-contact and on non-contact patients in hospitals within and remote from epidemics of poliomyelitis are summarized in Table 2. It will be seen that reactions in square centimeters and percent of reactions 5 sq. cm. or more, indicating specific streptococcal antigen in skin or blood, were greatest in well non-contact persons residing in regions where the incidence of the disease was low in 1945 and high in 1946, lower in regions where the incidence was high in 1945 and low in 1946, still lower in regions where the incidence was low both in 1945 and 1946, and least in regions far remote from poliomyelitis. Reactions to the control antibody prepared from streptococci isolated from the nasopharynx of well persons remote from poliomyelitis were consistently minimal or negative. Reactions indicating specific streptococcal antibody in skin or blood were relatively low in regions where the incidence was low in, 1945 and high in 1946 and by far higher in regions where the incidence was high in 1945 and low in 1946. Antibody and antigen were low where the incidence of poliomyelitis was low both in 1945 and 1946 and least in well persons far remote from poliomyelitis. Cutaneous reactions indicating antigen and antibody were far less in non-contact patients cared for in hospitals for non-infective ailments than in well non-contacts in regions where the incidence of poliomyelitis was low in 1945 and high in 1946 and reactions in similar hospitalized patients after the epidemic had disappeared were minimal and comparable to well non-contacts (Table 2).
Table 2. Erythematous reactions to intradermal injection of “poliomyelitic” and “normal” streptococcal antibody indicating antigen and “poliomyelitic” streptococcal antigen indicating antibody in skin or blood of well non-contact persons in 1946 in relation to the incidence of poliomyelitis in 1945.
*Non-contact bed patients in hospitals having illness other than poliomyelitis.
In accord with the isolation of the “poliomyelitic” type of streptococcus from outdoor air during the epidemic, it was found that reactions indicating antigen in two otherwise comparable groups of patients were twice as great in the group cared for in an “outer” ward of a general hospital in which windows were widely open, some facing hospital wards where cases of poliomyelitis were cared for, than in the group in an “inner” ward more shielded from outdoor air.
Similarly, in the epidemic zone the average reaction indicating antigen was 6.82 sq. cm. and the reaction indicating antibody was 2.69 sq. cm. in 66 nurses living in nurses’ home, and 9.07 sq. cm. and 4.69 sq. cm. respectively in 62 nurses doing comparable work in hospitals and living in private homes at some distance from the hospital.
The reactions indicating antigen in skin or blood of persons who had had poliomyelitis long before residing in the epidemic zone were slightly less, and reactions indicating antibody were slightly greater than in similar groups that had not had poliomyelitis.
Cutaneous tests were made with altogether nine different solutions of artificial antibody and of corresponding antigen prepared from streptococci isolated in studies of poliomyelitis, with three control solutions of antibody prepared from streptococci isolated from the nasopharynx of well persons remote from poliomyelitis, and with five control solutions of antibody prepared respectively from streptococci isolated from the nasopharynx of persons suffering from acute respiratory infection, chronic encephalitis, arthritis, epilepsy, or schizophrenia, all remote from poliomyelitis. Three of the nine solutions of antibody and antigen prepared from streptococci isolated in studies of poliomyelitis were prepared from streptococci isolated from the nasopharynx, and two from the spinal fluid and spinal cord of persons who suffered from or who succumbed to epidemic poliomyelitis. Two were prepared from streptococci isolated from spinal fluid or brain and spinal cord of monkeys that were ill with or that had succumbed to poliomyelitis following inoculation of passage virus; one was prepared from streptococci isolated from the brain and spinal cord of mice that died of poliomyelitis following inoculation of virus virulent for mice and one was prepared from three strains of the streptococcus isolated from outdoor air in the epidemic zone. Reactions to antibody prepared from streptococci isolated in studies of epidemic and experimental poliomyelitis and from outdoor air in the epidemic zone were roughly comparable and were uniformly far greater than reactions to intradermal injection of the different control antibody solutions. Reactions to control injections of NaCl solution, containing 0.2 per cent phenol made in 157 persons suffering from poliomyelitis, contacts and non-contacts were uniformly minimal or entirely negative.
Some months after these tests were made, my attention was called to the work of Jensen9 who found that the titer of poliomyelitic viral neutralizing antibodies of convalescent serum was by far lower in paralytic than in non-paralytic or abortive cases. The reactions indicating antigen and antibody in skin or blood of the large number of patients tested were summarized according to the presence or not of paralysis, to the degree of paralysis and to the incidence of poliomyelitis in 1945 and 1946. The data are summarized graphically in Figure 5. Reactions indicating specific streptococcal antigen in skin or blood were high, quite regardless as to whether paralysis was absent, slight or marked, and the average reactions were slightly greater in regions where the incidence of poliomyelitis was low in 1945 and high in 1946. In sharp contrast, the reactions indicating specific streptococcal antibody were far less in the group that contracted poliomyelitis in 1946 where the incidence of poliomyelitis had been low in 1945, as compared with the small group that contracted poliomyelitis in 1946 where the incidence had been high in 1945. In both groups, especially in the former, the average reactions indicating antibody were greatest in persons with little or no paralysis and least in persons severely paralyzed, corresponding to the viral titers reported by Jensen.
Figure 5. Erythematous reactions in persons suffering from poliomyelitis 1946 to intradermal injection of “poliomyelitic” streptococcal antibody, indicating antigen and antigen indicating antibody in skin or blood according to the degree of paralysis and to the incidence of poliomyelitis in 1945.
Results of Cutaneous and Agglutination Tests
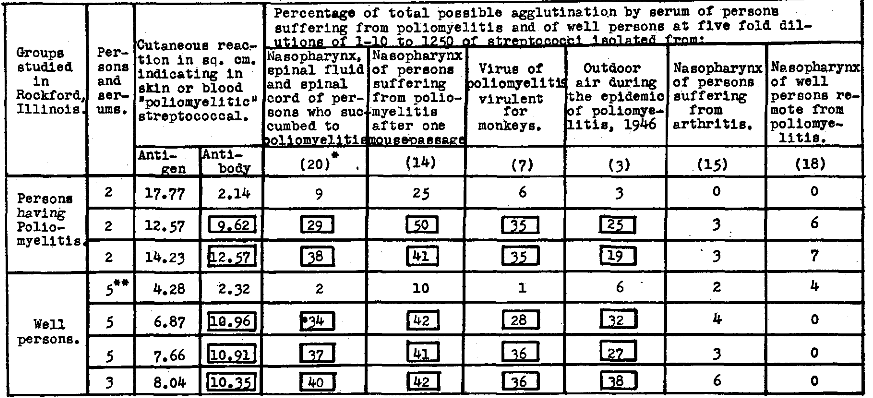
In Table 3 is shown the parallelism between reactions to intradermal injection of antigen indicating antibody and the agglutinative titer for streptococci isolated in studies of epidemic and experimental poliomyelitis and from outdoor air during the epidemic in 1946 of the serum of persons suffering from poliomyelitis and of well persons who lived in Rockford, Illinois, during the severe epidemic of 1945 and the slight recurrence in 1946. The comparative lack of antibody in skin or blood and in the serum of the five persons who resided in regions where poliomyelitis had not occurred in 1945 is noteworthy. The relatively low agglutinative titer of the serum from persons whose cutaneous reactions indicating antibody was low, and the high agglutinative titer of the serum from persons whose reaction indicating antibody was high for strains isolated in studies of epidemic and experimental poliomyelitis and outdoor air and the lack of agglutinins for the control strains are all strikingly shown.
Table 3. Erythematous reaction to intradermal injection of “poliomyelitis” streptococcal antigen, indicating antibody in skin or blood, in relation to the agglutination titer of the serum of persons suffering from poliomyelitis and of well persons who lived in Rockford, Illinois during the epidemic of 1945 and the slight recurrence in 1946.
*Numbers in parenthesis indicate strains of streptococci in respective suspensions.
**Persons living in Rockford in 1946 but who resided in regions in 1945 where poliomyelitis did not occur.
The agglutinative titers of artificial antibody prepared from streptococci isolated in studies of epidemic and experimental poliomyelitis and as controls of antibody prepared from streptococci isolated in studies of schizophrenia, epilepsy, and well persons over the respective streptococci are summarized in Table 4. A high degree of specificity of artificial antibody was shown for the homologous types of streptococci isolated in studies respectively of poliomyelitis including strains isolated from “virus” poliomyelitis in monkeys and mice and in studies of schizophrenia, epilepsy and well persons.
Table 4. Agglutinative titer of artificial antibody prepared from streptococci isolated in studies of epidemic and experimental poliomyelitis.
*The figures in ( ) indicate number of strains.
**The antibody solutions as used in these experiments consisted of the bacteria free supernatant of 1:100 dilutions of isotonic NaCl solution of the dense 1000 billion streptococci per ml suspensions in glycerol two parts and saturated NaCl solution one part.
The serum of horses that had been immunized with the streptococcus isolated in studies of poliomyelitis also agglutinated the streptococcus specifically.
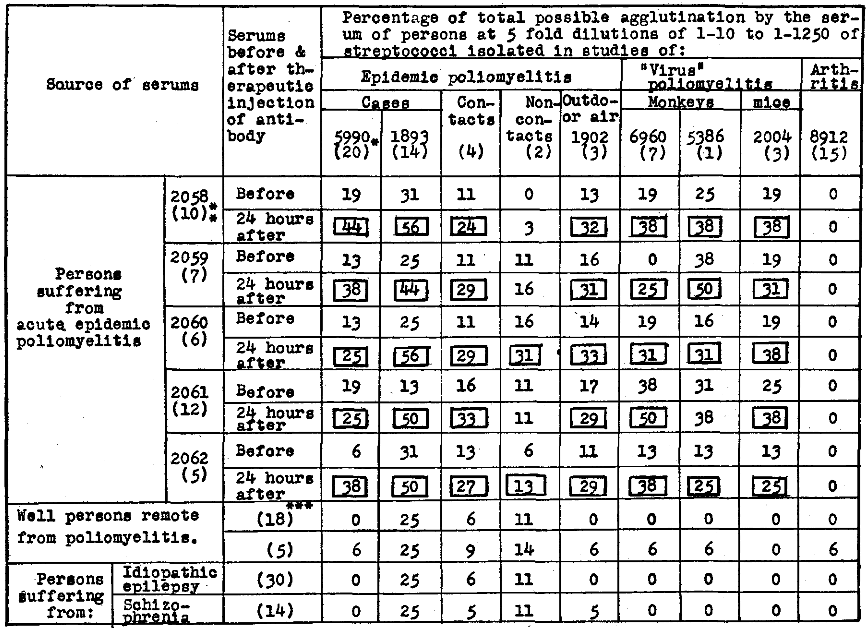
The agglutinative titers for streptococci of the serum of persons suffering from poliomyelitis before and 24 hours after therapeutic injection of artificial “poliomyelitic” streptococcal antibody are summarized in Table 5. A consistent increase in titer was found in these serums 24 hours after therapeutic injections of artificial antibody for streptococci isolated in studies of epidemic poliomyelitis, of contacts and non-contacts, of outdoor air, and of experimental “virus” poliomyelitis in monkeys and mice, and which was not found for streptococci isolated in studies of arthritis.
Table 5. Agglutination of streptococci isolated in studies of poliomyelitis by the serum of persons suffering from acute poliomyelitis before and 24 hours after therapeutic injection of “poliomyelitic” streptococcal antibody.
*Figures within parentheses indicate the number. of strains of streptococci pooled; **day of disease; and ***number of serums per pool.
Results of Therapeutic Injection of Artificial Antibody
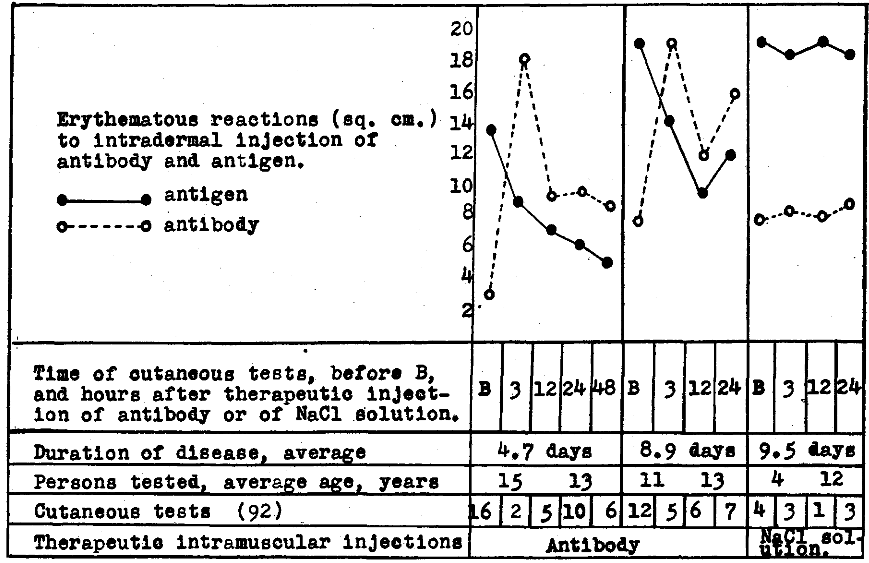
A summary of the effects of therapeutic injection of artificial antibody on cutaneous reactions indicating antigen and antibody respectively is shown graphically in Figure 6. Only one therapeutic, intramuscular, or subcutaneous injection of artificial antibody was given to each patient. This usually consisted of 2 ml. of the solution of antibody from 5 billion streptococci for children five years of age or younger and two additional milliliter for each additional year of age up to 20 years. A truly remarkable diminution of antigen and increase in antibody uniformly occurred in from one to three hours after the injection and remained at low and high level respectively for 24 or 48 hours or longer. Control injection of NaCl solution had no apparent effect.
Figure 6. Erythematous reactions to intradermal injection of antibody and antigen indicating antigen and antibody respectively in skin or blood of persons suffering from poliomyelitis following therapeutic intramuscular injection of “poliomyelitis” streptococcal antibody.
Of the 26 patients each receiving one therapeutic injection of the “poliomyelitic” streptococcal antibody, 19 showed clinical improvement, apparently attributable to this new treatment, as cutaneous reactions indicating antigen promptly diminished, and reactions indicating antibody increased. In two of these, both advanced cases of bulbar poliomyelitis, the improvement was temporary and death from respiratory failure ultimately occurred. In no instance of the 26 patients given therapeutic injection were the symptoms made worse. No clinical change attributable to the injection of antibody occurred in seven; all were severely paralyzed and four of the seven died. All of the four who died had been in the respirator because of advanced bulbar poliomyelitis and beneficial action was not to be expected. The details of the results obtained in treatment will be reported elsewhere.
Comments and Summary
The results of a bacteriologic and epidemiologic study of poliomyelitis made by new methods are reported.
The presence in the epidemic of poliomyelitis of 1946 of a specific type of streptococcic infection was indicated (1) by the consistent isolation from persons stricken, of streptococci that produced flaccid paralysis associated with degeneration of anterior horn cells in high incidence in mice; (2) by the specific agglutinative titer of convalescent serum, the serum of horses that had been immunized with the streptococci isolated in previous epidemics and the artificial antibody prepared from the streptococcus isolated in previous and the current epidemic; (3) by the erythematous reactions to intradermal injection of natural and artificial antibody and to antigen prepared from the streptococcus isolated in previous and the current epidemic indicating, respectively, specific streptococcal antigen and antibody in skin or blood; (4) by the progressive diminution of antigen and the increase in antibody in skin or blood and by the increase of the agglutinin titer in the serum as spontaneous recovery occurred; (5) by the abrupt drop in antigen and abrupt increase in antibody in skin or blood of persons suffering from poliomyelitis and the improvement in symptoms following therapeutic injection of the “poliomyelitic” streptococcal antibody and by its neutralizing action of poliomyelitic virus virulent for mice; and (6) by the demonstration of pleomorphic diplococci in the lesions in the spinal cord of persons who succumbed to poliomyelitis and their absence remote from the lesions.
The epidemiologic importance of the streptococcus was indicated (1) by the demonstration of specific streptococcal precipitinogen in and the isolation from outdoor air within the epidemic zone of a streptococcus which produced flaccid paralysis in mice in high incidence and which was agglutinated specifically in high titer by artificial antibody prepared from streptococci isolated from persons suffering from poliomyelitis and by convalescent serum; (2) by the isolation of the streptococcus true to type from the nasopharynx of well contacts and non-contacts in the epidemic zone; (3) by the general presence of this type of subclinical streptococcal infection in contacts and non-contacts as indicated by cutaneous tests; and (4) by the decrease in antigen and increase in antibody in skin or blood on prolonged contact; and (5) by the absence of this type of streptococcus in the air and in persons remote from poliomyelitis.
The production in mice of the all-important symptom, flaccid paralysis, associated with edema, hemorrhage and degeneration of ganglion cells in the anterior horns of the spinal cord with the streptococcus isolated from persons ill, well contacts and outdoor air is considered of fundamental importance even though some of the clinical and histological findings as seen in epidemic poliomyelitis in humans and experimental “virus” poliomyelitis in monkeys were lacking.
The reactions indicating a far higher streptococcal antibody content of skin or blood and in the serum of well non-contact persons in regions where poliomyelitis was epidemic in 1945 than in regions where the incidence of poliomyelitis was low in 1945 parallel the increase in viral neutralizing antibodies in the serum of well persons10 after exposure11 and after epidemics reported by others.12,13,14
The high titer of specific streptococcal antibody in skin or blood during convalescence from poliomyelitis and in well persons during mild outbreaks in 1946 where poliomyelitis was epidemic in 1945 reported herewith indicates it would seem the etiologic role of the streptococcus for the same fundamental reason that the presence of viral neutralizing antibodies in the serum of convalescents and well persons in relation to the occurrence of epidemics reported by others indicates the etiologic role of the virus.8,15,16,17
Moreover, the greatly lower titer of streptococcal antibody in skin or blood reported herewith and the greatly lower titer of viral neutralizing antibodies reported by Jensen9 in convalescent serum of paralytic poliomyelitis than in abortive or non-paralytic poliomyelitis likewise indicate etiologic significance of both the streptococcus and virus and that perhaps paralysis developed because of the lack of adequate antibody formation. Whether this striking parallel lack of viral and streptococcal antibody formation in paralytic poliomyelitis is expressive of a familial, physiologic or autarceologic defect and hence heightened susceptibility, as suggested by Aycock18 and as emphasized by Anderson,19 or whether due to a particularly severe infection by the streptococcus or virus is not clear.
Cutaneous reactions obtained in epidemic poliomyelitis before and after therapeutic injection of artificial antibody were similar to those obtained in studies of influenza and other epidemic respiratory infections, and in arthritis, epilepsy and schizophrenia with antibodies prepared from streptococci isolated from persons suffering from these respective diseases. Interestingly, intradermal injections elicited reactions indicating specific streptococcal antigen for six to eight weeks following attacks of poliomyelitis and for only two to three weeks following attacks of epidemic respiratory infections, corresponding to the enduring immunity in poliomyelitis and the transient immunity in influenza and epidemic respiratory infections.
The much higher specific antibody titer in well persons and in persons stricken in regions where poliomyelitis was epidemic in 1945 and where few cases occurred in 1946, than that of well persons and persons stricken in regions where few or no cases occurred in 1945 and where poliomyelitis was epidemic in 1946, the general presence of the specific type of streptococcus in both regions in 1946 and the absence of specific antigen and antibody in persons far remote from poliomyelitis indicate that the immunity to poliomyelitis following epidemics may be due to a greatly accelerated response in antibody formation on antigenic restimulation by the “seasonal” recurrence of the specific type of streptococcus and perhaps the accompanying virus.
The question whether the abrupt decrease in specific antigen and increase in antibody following therapeutic injection of artificial antibody is due wholly to the antibody injected, as would seem, or whether due in part to the release of preformed sessile antibodies from lymphocytes, reticulo-endothelial and other cells remains undetermined. This question, the further effects of therapeutic injections and the chemistry of artificial antibody are under study.
The data adduced in this study by the use of the new methods are in accord with previous studies and indicate anew that a specific type of alpha streptococcus, in some way, is etiologic in epidemic poliomyelitis. Experiments indicating the nature of the apparent relationship of both the streptococcus and the virus to poliomyelitis are not lacking. By the use of special methods the streptococcus has been consistently isolated from or demonstrated in the spinal fluid at the onset of symptoms of epidemic and experimental poliomyelitis when polymorphonuclear leukocytes are the predominating cell in the spinal fluid and in the lesions in the spinal cord,20 in the lesions after death, and in filtrates and emulsions of poliomyelitic virus.1 Filtrable transmissible poliomyelitic virus resembling natural virus in infectivity and antigenicity has been produced experimentally from “neurotropic” streptococci, including strains isolated from material wholly remote from poliomyelitis.1, 21, 22,23
The studies on the virus made now for nearly 40 years have been so alluring that the solution of the problem has quite naturally been sought from this standpoint, almost to the exclusion of forthright bacteriologic studies. It is hoped that the results, reported herewith, will lead to a broader approach at the solution of this problem than that hitherto employed, to a study of both the streptococcus and the virus.
aGrateful acknowledgment is hereby made for the cooperation of superintendents of hospitals, directors of Boards of Health, attending and resident physicians, and pathologists and nurses which made this study possible.
bFrom the Bacteriologic Research Laboratory, Longview Hospital, Cincinnati, Ohio.
References Cited:
- Rosenow, E. C.: Poliomyelitis. The Relation of Neurotropic Streptococci to Epidemic and Experimental Poliomyelitis and Poliomyelitis Virus, Diagnostic Serologic Tests and Serum Treatment. The International Bulletin, New York, vol. A-44, 1944.
- Rosenow, E. C.: Production in Vitro of Substances Resembling Antibodies from Bacteria. of Inf. Dis. 76:163-178, 1945.
- Rosenow, E. C.: Studies on the Nature of Antibodies Produced in Vitro from Bacteria with Hydrogen Peroxide and Heat. of Imm. 55:219-232 (March) 1947.
- Rosenow, E. C.: Isolation of Bacteria from Virus and Phage by a Serial Dilution Arch. of Path. 26:70-76, 1938.
- Foshay, Lee: The Nature of Bacterial-Specific Intradermal Antiserum Reaction. of Inf. Dis. 59:330-339, 1936.
- Francis, T., Jr.: Value of Skin Test with Type-Specific Capsular Polysaccharide in Serum Treatment of a Type I Pneumococcus Pneumonia. Exp. Med. 57:617-631, 1933.
- Rosenow, E. C.: Specific Types of Alpha Streptococci and Streptococcal Precipitinogen in Air in Relation to Epidemic Infections of the Respiratory, Tract and Nervous System. Journal-Lancet 65: 108-122, 1945.
- Schaeffer, M., and Muckenfuss, R. S.: Experimental Poliomyelitis. Published under Auspices of the National Foundation for Infantile Paralysis, 1940.
- Jensen, C.: The 1934 Epidemic in Denmark. Roy. Soc. Med. (Sec. Path.) 28:13-32, 1935.
- Shaughnessy, H. J., Harmon, P. H., and Gorden, F. B: Prev. Med. 4:463, 1930.
- Jungeblut, C. W.: The Power of Normal Human Sera to Inactivate the Virus of Poliomyelitis in Its Relation to Blood Grouping and to Exposure. Imm. 24:157, 1933.
- Aycock, W. L., and Kramer, S. D.: Immunity to Poliomyelitis in Normal Individuals in Urban and Rural Communities as Indicated by the Neutralization Test. Prev. Med. 4: 189, 1930.
- Brodie, M.: The Antibody Titer of so-called “Normal” Adult Serum Compared to That of Convalescent Poliomyelitis Serum. Bact. 23:102, 1932.
- Schultz, E. W., and Gebhardt, L. P.: Incidence of “Normal” Persons Possessing Demonstrable Antibodies for Poliomyelitis Virus in Their Serum. Soc. Exp. Biol. and Med. 28:409, 1931.
- Aycock, W. L.: A Study of the Significance of Geographic and Seasonal Variations in the Incidence of Poliomyelitis. Prev. Med. 3:245, 1929.
- Aycock, W. L., and Kramer, S. D.: Immunity to Poliomyelitis in a Southern Population as Shown by the Neutralization Test. Prev. Med. 4:201, 1930.
- Hudson, N. P., and Lennette, E. H.: Incidence of Poliocidal Sera in Regions Where Poliomyelitis Epidemics Are Infrequent. Jour. Trop. Med. 18:35, 1933.
- Aycock, W. L.: Nature of Autarceologic Susceptibility to Poliomyelitis. Jr. of Public Health 27:575-582, 1937.
- Anderson, G. W.: Epidemiology of Poliomyelitis. The Journal-Lancet 67: 10-13, 1947.
- Wolbach, S. B.: Personal communication.
- Rosenow, E. C.: The Production of a Filtrable Infectious Agent from Alpha Streptococci. Jour. Clin. Path. 14:150-167, 1944.
- Rosenow, E. C.: Studies on the Virus Nature of an Infectious Agent Obtained from Four Strains of “Neurotropic” Alpha Streptococci. Nerv. & Ment. Dis. 100:229-262, 1944.
- Rosenow, E. C.: A Filtrable Infectious Agent Obtained from Alpha Streptococci Isolated in Studies of a Case of Poliomyelitis. Jour. Clin. Path. 14:519-533, 1944.