Access to all articles, new health classes, discounts in our store, and more!
Microdiplococci in Filtrates of Natural and Experimental Poliomyelitits Virus Compared Under the Electron Microscopes
Presented at a meeting of General Staff held May 21, 1941. Published in the Proceedings of the Staff meetings of the Mayo Clinic, 17, pp. 99-106, February 18, 1942.
* * *
The presence of small oval cocci and diplococci (microdiplococci) in filtrates of poliomyelitic and encephalitic viruses as revealed in unstained preparations by the Rife microscope under polarized light,1 and as revealed by the ordinary microscope when the preparations are stained by a special method,2 has been reported previously. Evidence adduced at that time indicates that these small forms represent stages in the development of the cultivable streptococcus which we have isolated consistently in studies of poliomyelitis and encephalitis. Since filtrates of poliomyelitic virus are still generally considered to be bacteria-free, further proof of the presence and nature of these organisms in filtrates of virus was considered highly desirable and probably obtainable by the new tool—the electron microscope.3,4
The results of the examination of these filtrates under a standard RCA electron microscope and of these and other filtrates stained by my special method and examined under the ordinary light microscope are so important that a report is considered advisable at this time.
Observations with the Electron Microscope
The following materials were selected for examination under the electron microscope: (1) glycerolated spinal cords of three monkeys and one mouse that had succumbed to, or were anesthetized during, acute attacks of typical poliomyelitis after inoculation of filtered poliomyelitic virus; one of the three monkeys that had died of poliomyelitis had been inoculated intracerebrally with natural poliomyelitic virus in the sixth animal passage; the other two monkeys received one virus strain each and the mouse one of these virus strains which were derived from streptococci5 and which were in the seventh and sixteenth animal passages, respectively; sections of the spinal cords of the monkeys and mouse showed characteristic lesions; (2) a fifteen day chick-mash culture of the streptococcus from poliomyelitis; (3) eighteen hour cultures in dextrose-brain broth of two strains of streptococci from poliomyelitis; and (4) as controls, the spinal cords of a normal monkey and mouse and of a monkey that had died after intracerebral inoculation of streptococci and sterile chick-mash medium.
Emulsions of spinal cords for filtration were made in distilled water in sterile mortars by thorough grinding with alundum powder. The amount of water added was such that the emulsions contained approximately 3 to 5 per cent of cord substance. The emulsions of the respective viruses, of control material, of the culture in chick-mash medium and of the sterile chick-mash medium were passed through new Berkefeld V, N or W and Seitz filters by applying moderate suction, and through new Swinney filter disks by pressure made with a syringe. At least two filtrates were made and examined of each of these materials. For examination under the electron microscope all filtrates were diluted 1 to 10 with sterile distilled water. Approximately 1 cu. mm. of the diluted filtrate was placed on the collodion disk and allowed to dry before being placed in the high vacuum of the microscope for examination. The magnification routinely used was 12,000 diameters.
Microdiplococci In Filtrates
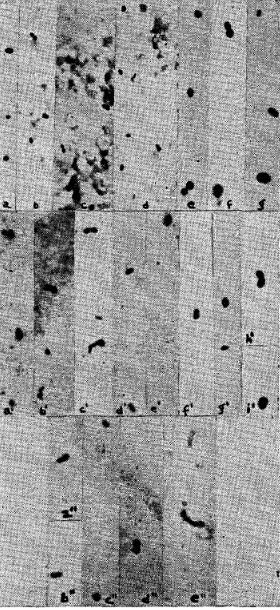
Microdiplococci, sometimes in short chains, of varying size and opacity, were found in each of the filtrates made of natural poliomyelitic virus (fig. 1a, b, c, d, e, f, g and h), of poliomyelitic virus derived from streptococci (fig. 1a’, b’, c’, d’, e’, f’, g’ and h’), and of the chick-mash culture of the streptococcus from poliomyelitis (fig. 1a”, b”, c”, d”, e” and f”). Exceedingly small ovoid and diplococcal forms and short chains of small diplococci (fig. 1a, b, c and d; b’, c’ and f’; d” and e”) some too small to photograph, were seen. The smallest forms seen resembling organisms approximated the postulated size of the virus particle. Some large forms seemingly were breaking into small diplococci (fig. 1e) similar to those seen in certain cultures of the streptococcus.6 The different forms, especially the larger ones, made visible by the ordinary microscope after staining (fig. 2), were distinguished easily from debris by their shape, intensity and smoothness of staining and regularity of margins (fig. 1a, c and h; e’, g’ and h’; a” and d”). As many as 200 microdiplococci were found in the examination of single specimens. It is calculated that this indicates the presence of at least 400,000,000 per cubic centimeter of filtrate.
Nothing resembling micro-organisms was found in the filtrates of emulsions of spinal cord of the two control monkeys, of the normal mouse and of sterile chick-mash medium.
Fig. 1. Large and small cocci, ovoids and diplococci, single and in short chains, resembling micro-organisms, revealed by the electron microscope in filtrates of emulsions of the spinal cord of a monkey that succumbed to natural poliomyelitic virus (a, b, c, d, e, f, g and h); in filtrates of emulsions of the spinal cord of a monkey (a’, b’, c’, and d’) and a mouse (e’, f’, g’ and h’) that succumbed to poliomyelitic virus derived from streptococci; and in filtrates of a fifteen day culture in chick-mash medium of a streptococcus from poliomyelitis (a”, b”, c”, d”, e” and f”). All photographs shown in figure 1 were taken at a magnification of 12,000 diameters.
Fig. 2. Cocci, ovoids and diplococci, single and in short chains, revealed by the ordinary light microscope in the same filtrates referred to in figure 1 after special staining. All photographs shown in figure 2 were taken at a magnification of 1,000 diameters.
Midget Forms Associated with the Streptococcus
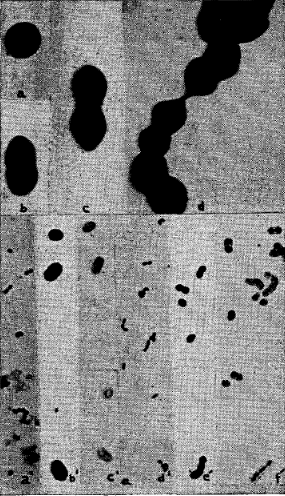
The unstained films of dextrose-brain cultures of two strains of streptococci isolated from poliomyelitic virus, one representing natural virus and one representing virus derived from streptococci, diluted 1 to 10 with distilled water just before examination, revealed the streptococci as very large, dense, round, oval cocci, diplococci and short chains of diplococci varying greatly in size (fig. 3a, b, c and d). Formalin (0.025 per cent) was added to the eighteen hour cultures and to 5 to 1 suspensions of the streptococci in distilled water forty-eight hours before examination. The films directly from the formalinized cultures in dextrose-brain broth revealed, in addition to the large organisms, numerous exceedingly small, elongated oval or diplococcal forms projecting radially from the margins, with few or no free forms (fig. 3c). The films from the formalinized suspensions in distilled water (fig. 3a, b and d) revealed few or none of these small forms at the margins of the large organisms, but showed similar more elongated forms dispersed in the surrounding medium, usually oriented in one direction (fig. 3a and d).
Fig. 3. Cocci, ovoids and diplococci, single and in short chains, in dextrose-brain broth cultures of two strains of streptococci from poliomyelitis, as revealed by the electron microscope at a magnification of 12,000 diameters (diplococci 1.2 to 1.6 microns in length) (a, b, c and d) and by the ordinary light microscope at a magnification of 1,000 diameters (diplococci 1.3 to 2.0 microns without capsule): a’ and d’ after staining by Gram’s method; b’ and e’ after staining by the special staining method as used in this study; and c’ and f’ after staining by the capsular stain as used previously.2
The location, radial orientation (fig. 3c), number and uniformity of size and shape of these “midget” forms suggest that they are dispersions of the large organisms and not artifacts due to possible shifting of the preparation while taking the picture.
If so, and if viable, this might be a reason for occurrence of growth of epidemic strains of streptococci in serial dilution cultures in dextrose-brain broth far beyond what would be considered possible on the basis of mathematical calculations of streptococci, as such, in the original culture.7
Observations with the Special Staining Method
Owing to the results obtained previously, and now with the electron microscope, search was made with the ordinary microscope at x 950 to x 1,000 for organisms in stained films of the same filtrates examined with the electron microscope. Similar diplococcal forms were found which, it is believed, represent the larger bodies revealed by the electron microscope (figs. 1 and 2). A study of filtrates of additional natural and experimental virus strains, of filtrates of chick-mash cultures of the streptococcus and of control filtrates by staining methods was undertaken.
The viruses and cultures studied by the special staining method were selected and the filtrates prepared in a manner identical to the way they were prepared for examination under the electron microscope, but the films were made from the undiluted filtrates.
We, as others, have found that staining of fixed films by ordinary methods, such as Gram’s method, Löffler’s methylene blue, Ziehl-Neelsen’s carbolfuchsin, Giemsa’s and Wright’s stains, as usually used, 1 per cent aqueous solutions of methyl violet (6B Grübler) or crystal or gentian violet, failed to show unmistakable micro-organisms. The special staining method previously used,2 slightly modified, was necessary to reveal micro-organisms consistently in stained filtrates of virus and filtrates of chick-mash cultures of the streptococcus. The method of staining consists of a combination of two nuclear staining technics used in the present study as follows: 1. Fix the air-dried film on a slide by heat in the usual way and while hot add a 5 per cent solution of chromic acid for fifteen seconds. Wash with a weak alkaline solution, such as a fresh 5 per cent solution of sodium bicarbonate, and water. 2. Stain with a 1 per cent aqueous solution of methyl violet (6B Grübler) for about fifteen seconds. Wash with water. 3. Cover film for about fifteen seconds with Gram’s solution of iodine. 4. Wash off iodine with a 2 per cent aqueous solution of safranine 0 (Grübler) and flush with water. Repeat steps 2, 3 and 4 once or twice, and after the last staining, decolorize slightly with 10 or 20 per cent alcohol. By this method small to large, diffusely stained brownish-purple, oval, coccal and diplococcal forms (microdiplococci) become clearly visible in supposedly bacteria-free filtrates (fig. 2). If staining and other conditions are just right, the center may be stained dark purple or may be unstained and surrounded or obscured by brownish-pink material. If carbolfuchsin is substituted for methyl violet in the staining method the center often appears unstained and is surrounded by a well defined brownish-pink capsule (fig. 2a and e”). If the films of filtrates that revealed unmistakable organisms were flushed once or twice with 95 per cent alcohol, the stained organisms became completely decolorized and invisible, except for a few exceedingly small indistinct forms. These microdiplococci revealed by the ordinary microscope are indisputably organismal, but the ordinary microscope seems to reveal only the larger micro-forms, while the electron microscope reveals smaller organisms which are probably beyond the range of the light microscope, even after special staining.
Typical microdiplococci of varying size, shape and density, resembling those revealed by the electron microscope (fig. 1), were found with the ordinary microscope in specially stained films of all sixty-seven additional filtrates of poliomyelitic virus and in all of twelve additional filtrates of chick-mash cultures of five strains of streptococci from poliomyelitis.
Distribution of Microdiplococci
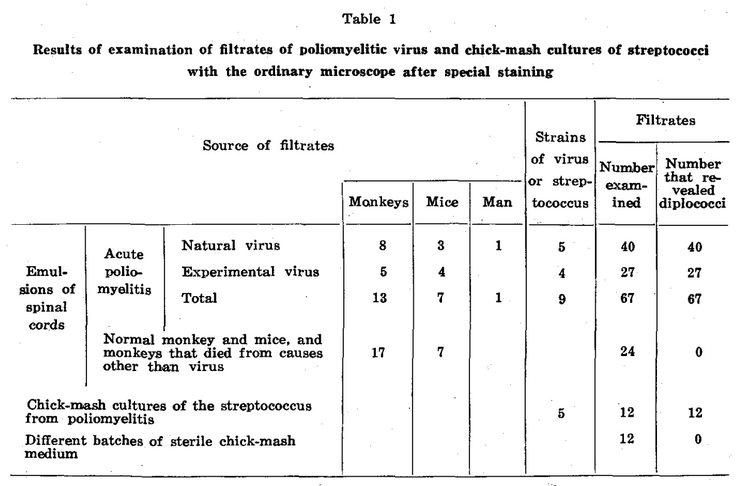
The number of microdiplococci found in the different stained films of filtrates of virus and chick-mash cultures of the streptococcus varied between wide limits, from ten to several hundred. The number found was greatest in filtrates representing emulsions prepared from spinal cords immediately after death, and in glycerolated virus the number was roughly inversely proportional to the duration of glycerolation. However, unmistakable forms were found in filtrates prepared from poliomyelitic spinal cords after they had been in 50 per cent glycerol in the refrigerator for eighteen months. The finding of at least ten typical microdiplococci diffusely stained and with smooth margins was considered essential for proof of the presence of organisms in any specimen. Spherical forms found in test filtrates, often in association with diplococci and streptococcal forms (figs. 1 b’, c’ and b”; 2 b’, i’ and e”), while no doubt often organisms, were not considered sufficiently different from similar forms sometimes found as precipitate in specimens of control filtrates, and hence were not counted. The source of test and control material, from which filtrates were made and examined, and the results obtained are summarized in table 1.
The Capsule of the Streptococcus
If this special staining method is applied to cultures of the streptococcus of ordinary size a capsule is demonstrable surrounding the streptococcus (fig. 3b’, c’, e’ and f’) as isolated in dextrose-brain broth. This capsule is not stainable by Gram’s method, although it is occasionally suggested by a colorless halo (fig. 3d’). Single organisms and chains of organisms stained deeply purple of about the size of organisms stained by Gram’s method (fig. 3a’ and d’), are seen in the center of (fig. 3c’ and f’), or are obscured by a wide rim of brownish-pink stained capsular substance (fig. 3b’ and e’).
Control Technics
In this, as in our previous study, controls were used to make certain that the organisms found were from the material under study. Different batches of staining and other solutions were checked for organisms. The slides of different makes were new and thoroughly cleaned. Blotting after staining always was done on new and clean filter paper. The glassware with which the materials came in contact was scrupulously cleaned. Films of control filtrates and other material were made and stained on the same slide with filtrates of virus and with filtrates of chick-mash cultures of the streptococcus.
In order to check still further the results obtained, additional filtrates were made of emulsions of tissues known to, and those known not to, contain virus. The tissues were from an ether-killed monkey that showed typical flaccid paralysis and other symptoms of poliomyelitis eight days after inoculation of a virus strain derived from a streptococcus in the eighth monkey passage. Filtrates of 3 to 5 per cent emulsions in solution of sodium chloride were made immediately after the death of the animal from the spinal cord, brain, sciatic nerve, liver, spleen, kidney, myocardium, skeletal muscles and stool. Portions of the emulsion of the spinal cord were passed respectively through new Berkefeld V, N and W and Seitz filters and Swinney filter disks. Stained films from each of these filtrates revealed characteristic microdiplococci in numbers roughly inversely proportional to the density of the filter. Each of the emulsions from the other tissues was passed through Berkefeld N and Swinney filters. Characteristic organisms were found in relatively large numbers in filtrates of the liver, and in small numbers in filtrates of the brain, spleen and sciatic nerve. Microdiplococci were not found in filtrates of the kidney, myocardium, skeletal muscles or stool. Cultures in dextrose-brain broth of the emulsions of the spinal cord, spleen and sciatic nerve yielded the streptococcus, whereas corresponding cultures in dextrose broth and on blood agar were negative. All cultures of filtrates and of the emulsions from liver, kidney, muscle, heart and brain remained sterile.
Autolysis of the Microdiplococci in Filtrates
Films were made repeatedly, on storage at room or incubator temperature, of filtrates that showed unusually large numbers of microdiplococci immediately. There was no perceptible diminution of stainable forms in twenty-four to thirty-six hours, usually moderate diminution in forty-eight to seventy-two hours, and almost complete disappearance in four to five days. Centrifugal force sufficient to pack the cultivable streptococci to the bottom of centrifuge tubes did not diminish perceptibly the number of organisms in the supernatant of the filtrates.
Cultures from Emulsions and Filtrates
The microdiplococci are very different in their growth requirements from the streptococci. Cultures of emulsions in dextrose-brain broth of the different strains of viruses represented in this study usually yielded the streptococcus, while cultures of only four of twenty-two filtrates yielded pure growths of the streptococcus in this medium. In no instance did parallel cultures of emulsions and filtrates in dextrose broth and on blood agar yield streptococci. Pools of filtrates of emulsions from mice and monkeys that revealed microdiplococci produced poliomyelitis in the mouse or monkey, after intracerebral inoculation.
Morphology of the Microdiplococci
The number, size, shape and density of the microdiplococci were closely similar in the different filtrates regardless of whether they were from emulsions of the spinal cord of a person who had died of epidemic poliomyelitis, from emulsions of natural virus from monkeys and mice, from emulsions of experimental “streptococcic” poliomyelitic virus from monkeys and mice or from old cultures of the streptococcus in chick-mash medium. Some of the organisms found with the electron microscope resembled the photomicrographs of vaccine virus and of infectious ectromelia of the mouse by von Borries, Ruska and Ruska.8 The demonstration by Barnard9 of organisms in the virus of foot and mouth disease and of vesicular stomatitis with ultraviolet light at x 3,250 should also be mentioned.10
The variations in size, shape and grouping and apparent breaking of the large microdiplococci into minute forms (fig. 1e) in filtrates of virus and in filtrates of chick-mash cultures of the streptococcus were similar to variations in size, shape, grouping and the breaking of large diplococci into small ones, found in stained films of the globoid bodies described by Flexner and Noguchi,11 in the cultures of the streptococcus isolated in 191612 and since in our studies, depending on the medium in which they were grown,6 in stained films of nasopharyngeal swabbings and spinal fluid13, 14 and in stained sections of the spinal cord and tonsils in poliomyelitis.11,15,16
Conclusions
The presence of microdiplococci in filtrates of poliomyelitic virus as revealed by the electron microscope and, after special staining by the ordinary light microscope is reported. The results previously reported have been corroborated and extended. Microdiplococci have now been found in unstained films of filtrates of natural and experimentally produced poliomyelitic virus under very high magnification with the Rife polarized light microscope (x 8,000) and the electron microscope (x 12,000), and in specially stained films of filtrates under lower magnification (x 1,000) with the ordinary light microscope. The microdiplococci are considered to be forms of the virus and to be related to or associated with the streptococcus which we have isolated consistently in studies of poliomyelitis.
References Cited:
- Rosenow, E. C.: Observation on filter-passing forms of Eberthella typhi (Bacillus typhosus) and of the streptococcus from poliomyelitis. Proc. Staff Meet., Mayo Clin. 7: 408-413 (July 13) 1932.
- Rosenow, E. C.: A method of staining microorganisms and their capsular substance, and its application to streptococci and to filtrates of the viruses and spinal fluids in poliomyelitis and encephalitis. Proc. Staff Meet., Mayo Clin. 10: 115-121 (Feb. 20) 1935.
- Hillier, J. and Vance, A. W.: Recent developments in the RCA electron microscope. Proc. Inst. Radio Engineers 29: 167-176, no. 4, 1941.
- Rhea, H. E.:· The new electron microscope. Science. n.s. 93: 357-358 (Apr. 11) 1941.
- Rosenow, E. C.: The relation of streptococci to the viruses of poliomyelitis and encephalitis: preliminary report. Proc. Staff Meet., Mayo Clin, 10: 410-414 (June 26) 1935.
- Rosenow, E. C. and Towne, E. B.: Bacteriological observations in experimental poliomyelitis of monkeys. J.M. Research 36: 175-186 (Mar.) 1917.
- Rosenow, E. C..: Isolation of bacteria from virus and phage by a serial dilution method. Arch. Path. 26: 70-76 (July) 1938.
- von Borries, B., Ruska, E. and Ruska, H.: Bakterien und Virus in iibermikroskopischer Aufnahme (mit einer Einführung in die Technic des tlbermikroskops). Klin. Wchnschr. 17: 921-925 (July 2) 1938.
- Barnard, J. E.: Foot-and-mouth disease and vesicular stomatitis; a comparative microscopical study. Proc. Roy. Soc., London s. B. 124: 107-113 (Nov. 19) 1937.
- After submitting this paper for publication my attention was directed to the demonstration by Coles of small, pleomorphic organisms in stained films of affected tissues and filtrates of poliomyelitic, encephalitic and other viruses. The references are: Coles, A. C.: A microscopical inquiry into the aetiology of poliomyelitis with a note on chronic encephalitis lethargica. Edinburgh M. J. 46: 627-638 (Oct.) 1939, and, The size and visibility of the filterable or virus bodies. Brit. M. J. 2: 507-509 (Oct. 11) 1941.
- Flexner, Simon and Noguchi, Hideyo: Experiments on the cultivation of the microorganism causing epidemic Poliomyelitis. J. Exper. Med. 18: 461-485 (Oct.) 1913.
- Rosenow, E. C. and Wheeler, G. W.: The etiology of epidemic poliomyelitis. J. Infect. Dis. 22: 281-312, 1918.
- Rosenow, E. C.: Streptococci in the spinal fluid in acute epidemic poliomyelitis: Preliminary report. J. A. M. A. 91: 1594-1595 (Nov. 24) 1928; Proc. Staff Meet., Mayo Clin. 3: 330- 331 (Nov. 14) 1928.
- Rosenow, E. C.: Streptococci in the spinal fluid of experimental poliomyelitis in monkeys. J. Bact. 19: 27-28 (Jan.) 1930.
- Hektoen, Ludvig, Mathers, George and Jackson, Leila: Microscopic demonstration of cocci in the central nervous system in epidemic poliomyelitis. J. Infect. Dis. 22: 89-94, 1918.
- Rosenow, E. C.: Streptococci in the lesions of experimental poliomyelitis in monkeys. J. Bact. 19: 27 (Jan.) 1930; Proc. Soc. Exper. Biol. & Med. 27: 444-445 (Feb.) 1930.