Access to all articles, new health classes, discounts in our store, and more!
Local and Systemic Influences in Periodontal Disease: II Effect of Prophylaxis and Natural Versus Synthetic Vitamin C Upon Gingivitis
Published in The Journal of Periodontology, Vol. 35, pp. 58-67, May-June 1964.
* * *
There is growing evidence that periodontal disease is the result of the interplay of the local environment and the host.1-6 The purpose of this study is to analyze the relative effects of natural versus synthetic vitamin C with and without bioflavonoids and with and without scaling upon the gingival state.
Review of the Literature
For this study the review will be limited to one oral factor (calculus) since it was the local variable which was altered and three systemic factors (natural vitamin C concentrate versus synthetic vitamin C with and without bioflavonoids) since these were varied..
Of all of the known local factors, calculus is considered the most important of the gingival irritants.7-11 Whether calculus actually initiates the inflammation or whether gingival inflammation caused by other irritants is necessary before calculus can be formed remains to be established.9 Whatever the sequence of events, the irritating effect of calculus as a result of the combined mechanical, chemical and/or microbial factors, contributes to the development of gingival inflammation. As the calculus increases in size, the inflammatory reaction spreads and gingival recession eventually occurs. Progressively the deeper periodontal tissues become involved with pocket formation.
Generally speaking, lower animals subjected to vitamin C deficient diets show detrimental changes in the gingiva, periodontal membrane and alveolar bone.12-16
Human studies have likewise revealed that there is a correlation between ascorbic acid status and the appearance of the gingiva.17-21 The manifestations of vitamin C deprivation are held to proceed in a definite order affecting in sequence the interdental papilla, marginal and the alveolar gingiva.22-23
Increasing attention is being paid to capillary integrity. The bioflavonoids have been found to be one of the essential factors in the maintenance of capillary resistance. Some few investigators have reported upon the successful use of the water-soluble bioflavonoid preparations in the treatment of the periodontal diseases.24-26
Very limited reports indicate the possible superiority of natural versus synthetic vitamin C.27 No known stomatologic studies are available.
Method of Investigation
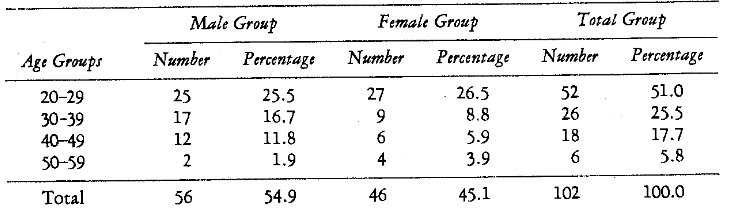
One hundred and two subjects participated in this study. The selection was as random as possible from volunteers at the University of Alabama Medical and Extension Centers and from the Birmingham Fire Department. Table 1 shows the patient distribution in terms of age and sex.
Table 1–Patient Age and Sex Distribution
All subjects reported to the Department of Oral Medicine following a 12-hour fast. Upon admission, a fasting venous blood sample was drawn. The oral examination was conducted with the patient comfortably seated in a dental chair. The mouth was illuminated with a dental spotlight. In all cases, the examination started in the maxillary right canine region and progressed across to the left cuspid. The same pattern was followed in the mandible. One examiner (G. M. A.) made all of the observations.
Calculus Scores: Although the quantity of calculus undoubtedly has a bearing upon the degree of pathosis, it was felt that the location of these deposits was of major importance. Hence, the evaluation was based upon whether there was an absence of calculus or whether supragingival, subgingival, or both supra- and subgingival calculus was present on each tooth examined. The inspection was done with an explorer and mirror. Compressed air was used to retract the gingiva for better detection of subgingival deposits. The legend for scoring calculus is shown (Table 2). In the individual with all 12 anterior teeth, 12 calculus scores were recorded. The individual scores were then added and divided by the number of teeth to obtain a mean value for each side.
Table 2–Calculus Scoring System
Gingival Scores: The soft tissues surrounding the 12 anterior teeth were evaluated by scoring the labial and lingual marginal gingiva on each tooth and the labial and lingual aspects of the gingival papillae between each tooth. Thus, in a subject with 12 anterior teeth, 48 measurements were obtained. Table 3 shows the gingival scoring system. From the individual measurements, mean right and left values were derived. Finally, a color photograph of the anterior teeth and gingiva was taken.
Table 3–Gingival Evaluation
Measurement of Vitamin C State: The method of Mindlin and Butler28 was utilized for the determination of plasma ascorbic acid levels. The test was always done twice from equal portions of the same blood sample. These duplicate determinations served as a check on the accuracy of the procedure (r = +0.988, P < 0.001).
For the determination of tissue ascorbic acid status, the lingual vitamin C test was employed.29-30 This is a simple procedure which involves the timing (in seconds) of the decolorization of one minim of N/300 dichlorophenolindolphenol dye deposited upon the dorsum of the dried tongue. In order to evaluate the reproducibility of the test, the procedure was always performed twice. The correlation between the first and second lingual time readings was found to be highly significant (r = +0.989, P < 0.001).
Bioflavonoid state was not ascertained since there are no known laboratory tests.
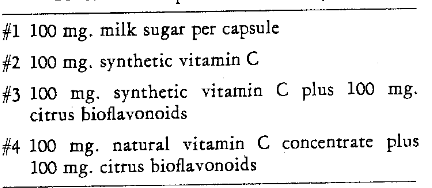
By means of a table of random numbers the patients were arranged into two groups: (1) those to be scaled on the right side, and (2) those to receive prophylaxis on the left. Also, the patients were randomized to receive one of the systemic regimes. Thus, in terms of systemic therapy, four groups were developed. Table 4 shows the composition of the capsules which were administered. All capsules looked exactly alike. Thus, neither the patient nor the examiner were aware at any time during the experiment which preparation was employed.
Table 4–Codes and Composition of the Capsules
At the first visit, each patient received thorough scaling of the teeth on one side of the mouth. Jaquette and McCall scalers were employed to remove the calcareous deposits. Moistened flour of pumice was applied with a revolving rubber cup in a contra-angle handpiece to polish the accessible crown and root surfaces. Finally, dental tape and pumice were utilized to polish the interproximal surfaces of the teeth.
At the completion of the first visit each patient was instructed to take by mouth three capsules per day for three weeks. Thus, 25 subjects received a placebo, 25 were given 300 mgm. synthetic vitamin C daily, 25 were administered 300 mgm. synthetic vitamin C plus 300 mgm. citrus bioflavonoids per day, and the remaining 27 were supplemented with 300 mgm. of a natural vitamin C concentrate with 300 mgm. bioflavonoids each day.
Approximately 21 days later each of the patients returned. All participants were reexamined clinically and biochemically without reference to the earlier records or the nature of the supplementation.
Results
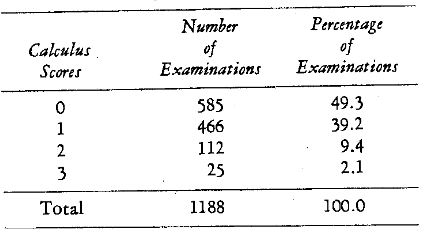
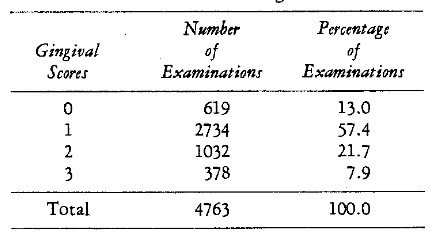
Pretherapy Findings: Table 5 summarizes the initial calculus scores. It is clear that about one-half of the examined areas are without calculus. The initial gingival scores are also shown (Table 6). About 13 per cent of the gingival areas showed no signs of pathosis. The remaining 87 per cent ranged from slight pathosis (57.4 per cent) to severe gingivitis (7.9 per cent). Table 7 outlines the patient distribution in terms of the initial plasma ascorbic acid levels. It was found that the scores ranged from 0 to 1.38 mgm. per cent. The distribution of the initial lingual times is included (Table 8). The values ranged from 12 to 60 seconds.
Table 5–Calculus Distribution (Initial Scores)
Table 6–Distribution of Initial Gingival Scores
Table 7–Patient Distribution in Terms of Initial Plasma Ascorbic Acid
Table 8–Patient Distribution in Terms of Initial Lingual Time
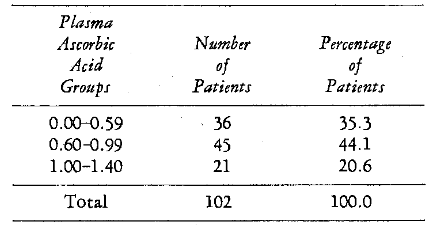
The relationship between calculus and gingivitis is pictorially portrayed (Figure 1). It will be noted that the subjects with low calculus scores also have low mean gingivitis values. Conversely, those individuals with high calculus ratings are associated with high mean gingival readings. A significant correlation was found between calculus and gingivitis (r = +0.568, P < 0.001).
Fig. 1. Pretherapy relationship of a local factor (calculus) to periodontal pathosis (gingivitis).
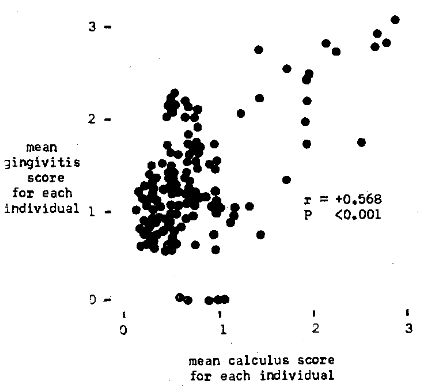
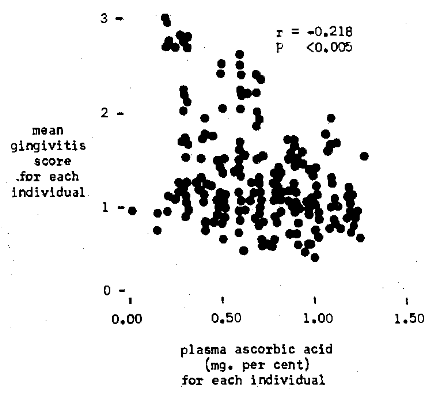
Figure 2 illustrates graphically the correlation of plasma ascorbic acid to gingivitis. The chart shows that those subjects with the highest mean gingivitis scores are associated with the lowest vitamin C levels. On the other hand, those persons with the lowest mean gingival values are in parallel with the highest plasma vitamin C levels. The relationship between plasma ascorbic acid and gingivitis was found significant (r = -0.218, P < 0.005). The correlation between the lingual times and gingivitis is also shown (Figure 3). Subjects with high mean gingivitis ratings are associated with long lingual times; individuals with low gingival values have short lingual times. A statistically significant correlation was obtained (r = +0.200, P < 0.005).
Fig. 2. Pretherapy relationship of a systemic factor (plasma ascorbic acid) to periodontal pathosis (gingivitis).
Fig. 3. Pretherapy relationship of a systemic factor (lingual time) to periodontal pathosis (gingivitis)
The relationship of both calculus and vitamin C state (plasma vitamin C levels) is included (Table 9). The participants with the lower calculus (0.0-0.5) and higher vitamin C levels (0.60-1.40) have the lowest gingival scores (1.0 ± 0.2). Those individuals with the higher calculus values (> 0.5) and poorer vitamin C levels (0.00-0.59) are associated with the greatest gingivitis ratings (1.8 ± 0.6). As one proceeds from left to right (irrespective of plasma ascorbic acid levels) the gingivitis increases with increase in calculus values. As one moves from top to bottom in either column (irrespective of calculus state) the gingivitis increases with a decrease in the vitamin C level.
Table 9–Pretherapy Relationship of the Mean Calculus Scores and Ascorbic Acid Levels (mg. per cent) to the Subjects’ Mean Gingivitis Scores
Though interesting, the preceding relationships do not necessarily demonstrate cause-and-effect. These correlations simply indicate that variables co-exist. To try and pinpoint possible cause and effect, it was felt advisable to study the gingival pattern after both local (prophylaxis) and systemic therapy (synthetic and natural vitamin C concentrate with and without bioflavonoids).
Posttherapy Findings: The nature of the experimental design provided the opportunity to study the changes in the gingivae after both local (prophylaxis) and/or systemic (synthetic and natural vitamin C concentrate with and without bioflavonoids) therapy.
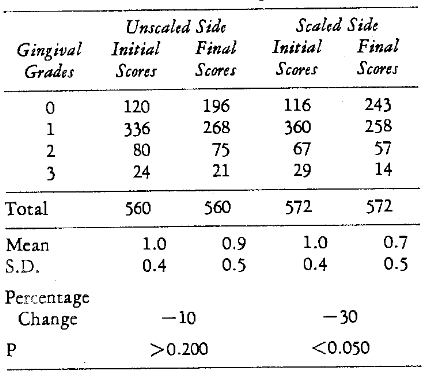
Effect of Scaling: In each subject one half of the teeth were scaled at the first visit. Thus, in the placebo group, it was possible to examine the effect of the removal of calculus upon the periodontium on one side versus the untouched (nonscaled) other side of the gingiva. Table 10 shows the original and mean initial and final gingival scores for the scaled and nonscaled sides in those persons without systemic treatment (placebo). There appears to be a 30 per cent and significant (P < 0.050) improvement in mean gingivitis score with scaling. The nonscaled side showed an insignificant (P > 0.200) slight reduction (10 per cent).
Table 10–Gingivitis Change after Prophylaxis (Placebo Group)
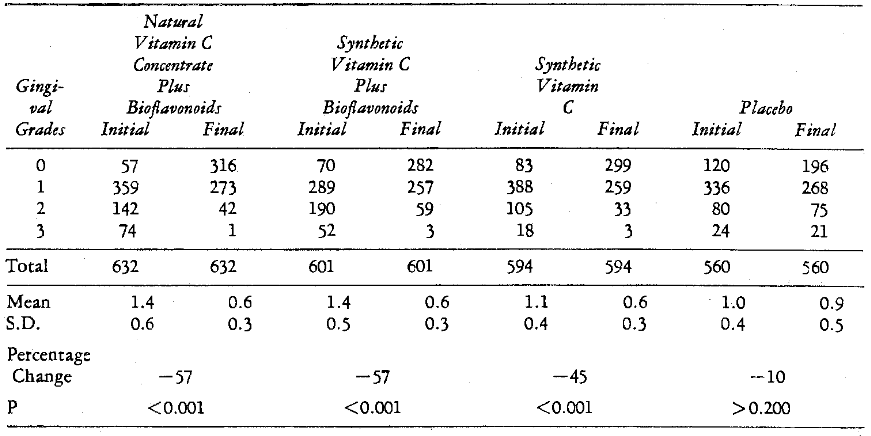
Effect of Systemic Therapy: It was also possible to evaluate the effect of altering the host without local treatment (no scaling). Table 11 includes the initial and final original and mean gingival ratings of those subjects receiving systemic therapy versus placebo supplementation. There appears to be a 57 per cent improvement in the mean gingivitis scores (P < 0.001) in both the groups subjected to natural vitamin C concentrate and synthetic vitamin C with bioflavonoids. The synthetic vitamin C group without bioflavonoids followed with 45 per cent significant reduction (P < 0.001). Finally, the 10 per cent decrease in the placebo group is not significant (P > 0.200).
Table 11–Gingivitis Change after Systemic Therapy (Unscaled Side)
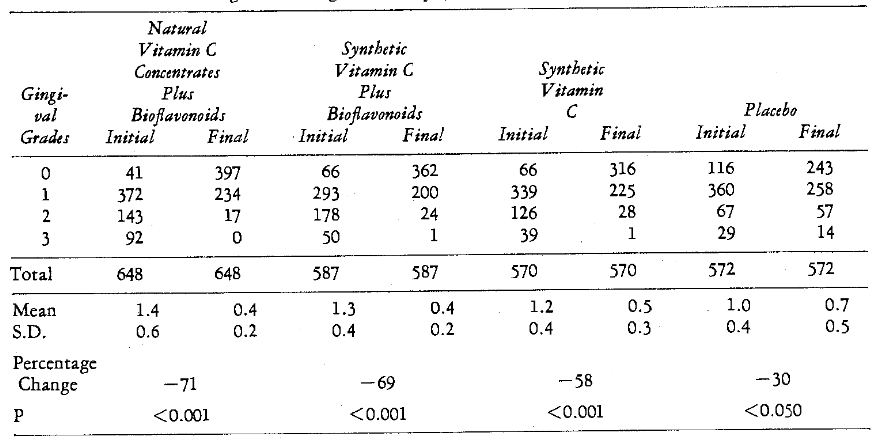
Effect of Local and Systemic Therapy: Table 12 illustrates that the greatest improvement on a mean basis (71 per cent) occurred in those persons subjected to both prophylaxis and natural vitamin C concentrate with bioflavonoids. In order followed those with scaling and synthetic vitamin C with bioflavonoids (69 per cent). The next greatest improvement was shown with prophylaxis and synthetic vitamin C without bioflavonoids. Finally, the patients with only local therapy (placebo group) had a decrease of 30 per cent. On a statistical basis, all groups given both local and systemic treatment had more significant percentage improvement (P < 0.001) than those given only local therapy (P < 0.050).
Table 12–Gingivitis Change after Prophylaxis and Systemic Therapy
Discussion
It should be recalled that two possible factors were studied in terms of their relationship to gingival pathosis. These variables were calculus, representing the local problem, and vitamin C state on the systemic side. The higher correlation (r = +0.568, P < 0.001) was found between calculus and gingivitis. Then followed the correlation of plasma ascorbic acid to gingivitis (r = -0.218, P < 0.005). An equally significant relationship was obtained between the lingual time and gingivitis (r = +0.200, P < 0.005). It is well to underscore the fact that, at the initial visit, the correlation of calculus and gingivitis is greater than that of vitamin C and gingival states.
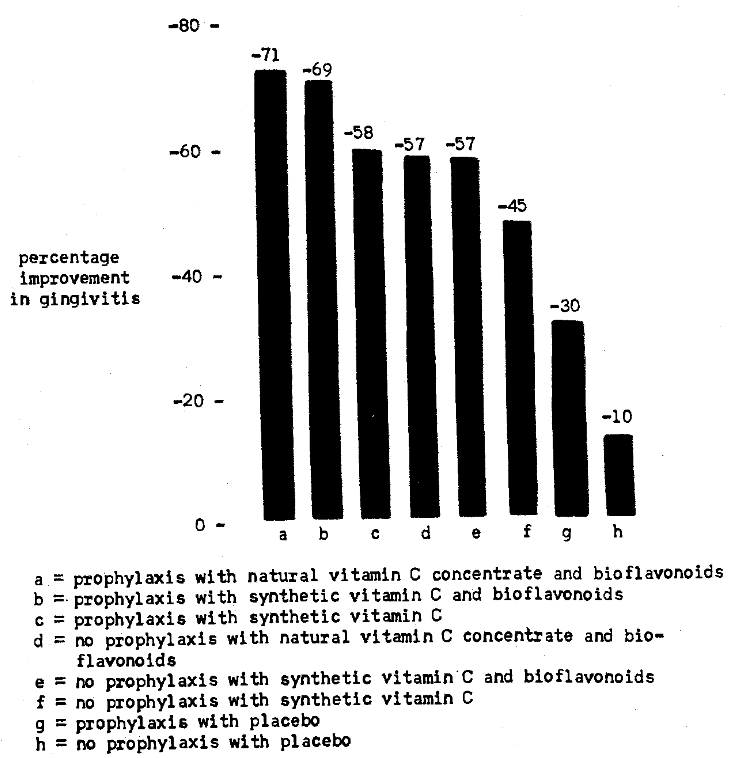
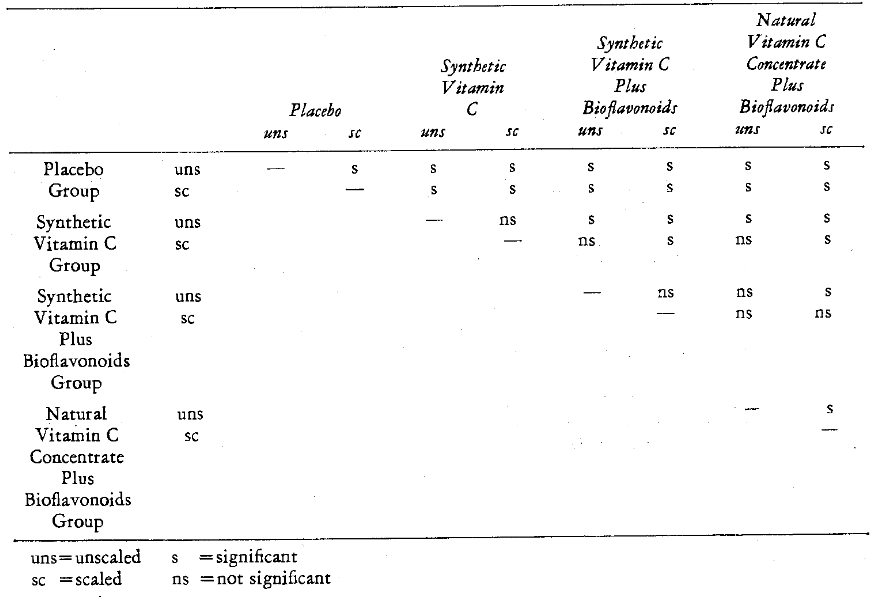
Figure 4 graphically depicts the percentage change in gingival state with the eight different forms of therapy. Table 13 summarizes the intergroup statistical significance. It is noteworthy that all nonscaled groups were statistically significantly different except for the nonscaled natural versus synthetic vitamin C with bioflavonoids (57 per cent). Table 13 also illustrates that all scaled groups were significantly different except for the gingivitis reductions (71 and 69 per cent) in those subjects treated with natural versus synthetic vitamin C with bioflavonoids (Table 12). It is also clearly shown that only two of the four groups were significantly different (Tables 11 and 12) in terms of scaling versus nonscaling. Specifically, natural vitamin C concentrate with bioflavonoids with (71 per cent) and without scaling (57 per cent) were significantly different. Also, the placebo groups with (30 per cent) and without prophylaxis (10 per cent) were statistically different. It appears that both synthetic and natural vitamin C concentrate exerts a beneficial effect in reducing gingivitis and particularly so along with bioflavonoids and prophylaxis.
Fig. 4. Posttherapy percentage gingivitis change.
Table 13–Comparison of the Statistical Significance of the Therapeutic Effectiveness on Gingivitis of the Various Subgroups
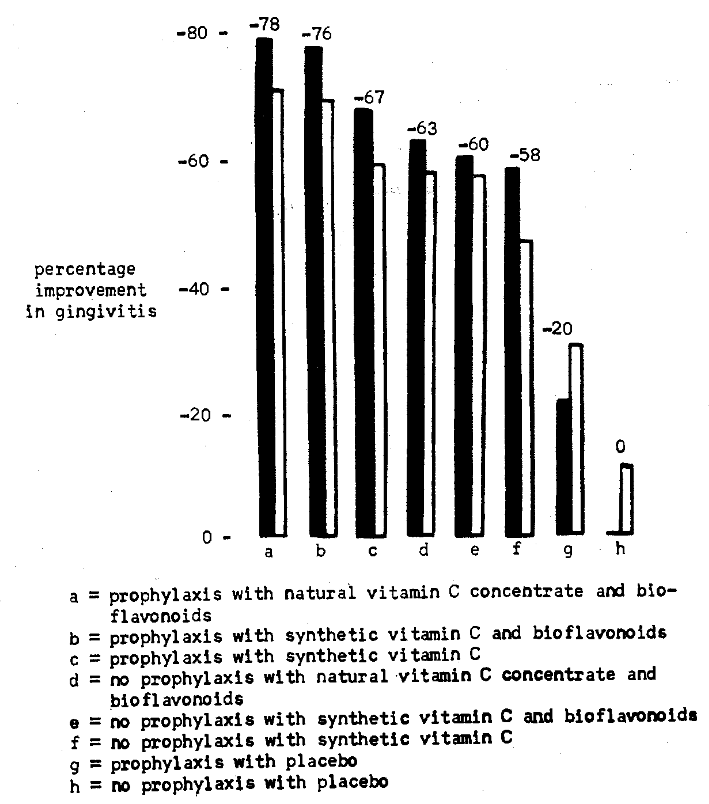
It should be recalled that vitamin therapy was administered on a random basis. Obviously, by this technique, some of the vitamin-supplemented subjects actually had satisfactory vitamin C levels at the start and therefore did not require vitamin therapy. Hence, it was thought proper to restudy the gingival patterns in the subjects with the lowest plasma ascorbic acid levels and highest lingual times. For this phase, the ten patients in each group with the poorest scores were selected. Figure 5 outlines the findings for the entire groups (white columns) and those with the poorer vitamin C findings (black columns).
Fig. 5. Comparison of the percentage improvement in gingivitis of each group listed below (white column) with the ten subjects from each group with the poorest vitamin C levels (black columns).
Three points deserve particular attention. Firstly, the overall pattern for the 10 subjects follows the pattern for the entire group. Secondly, in each group both locally and systemically treated, greater improvement occurred in the selected group than in the entire sample. Finally, those 10 persons given only local therapy showed less change than the entire group. This may possibly be due to the fact that these relatively sicker persons required more than prophylaxis. Also, the relatively sicker (placebo treated with no scaling) showed zero change in contrast to the 10 per cent mean improvement for the entire group.
The question arises as to whether the biochemical patterns followed the clinical change. No significant change in plasma ascorbic acid levels and lingual times followed placebo therapy (P = 0.500 and > 0.100 respectively). On the other hand, there were significant increases in the plasma vitamin C levels following natural vitamin C concentrate with bioflavonoids (P < 0.005), and synthetic vitamin C with and without bioflavonoids (P < 0.001). Also, all vitamin-treated groups showed significant reductions in lingual time (P < 0.001). Finally, all three supplemented groups significantly differed from the placebo-treated subjects. All of these findings parallel the clinical patterns. However, there were no other intergroup biochemical differences. This is at variance with the clinical changes. This difference is probably due to the fact that neither plasma ascorbic acid nor lingual vitamin C testing reflects bioflavonoid change.
Summary and Conclusions
This study was intended to evaluate the relative effects of correction of a local factor (calculus) and systemic factors (natural vitamin C concentrate versus synthetic vitamin C with and without bioflavonoids) upon periodontal pathosis (gingivitis).
An attempt was first made to correlate gingival state with calculus and ascorbic acid status. The greater correlation was found between calcareous deposits and gingivitis. Though these relationships were all statistically significant, one cannot ascribe from these data any cause and effect.
To attempt possible cause-and-effect relationships, the latter phase of the study involved the appraisal of gingival state after the subjects received one of eight different therapeutic regimes. The treatments involved placebo versus synthetic vitamin C with and without bioflavonoids versus natural vitamin C concentrate with bioflavonoids with and without scaling of one half of the mouth.
Re-examination three weeks later showed a significant reduction in gingivitis in those groups given local and/or systemic therapy. It appears, within the limits of this study, that the combined treatments yielded the best results.
References Cited:
- Osgood, H. A. “Systemic aspects of periodontal disease.” J.A.D.A. 15:1, 144-148, January 1928.
- Chilton, N. W. “Nutritional aspects of periodontal therapeutics.” J.A.D.A. 43:11, 583-592, November 1951.
- Radusch, D. F. “Periodontics and diet control.” J. Canad. D. Assn. 17:7, 378-384, July 1951.
- Glickman, I. “Interrelation of local and systemic factors in periodontal disease. Bone factor concept.” J.A.D.A. 45:4, 422-429, October 1952.
- Miller, S. C. and Roth, H. “The present state of knowledge of scorbutic gingivitis.” New York D. J. 23:5, 208-212, May 1957.
- Stahl, S. S., Miller, S. C. and Goldsmith, E. D. “The effects of vertical occlusal trauma on the periodontium of protein deprived young adult rats.” J. Periodont. 28:2, 87-97, April 1957.
- Beube, F. E. Periodontology. 1953. New York, The Macmillan Company. First edition. pp. 37-63.
- Coolidge, E. D. and Hine, M. K. Periodontology. 1958. Philadelphia, Lea & Febiger. Third Edition. pp. 149-152.
- Glickman, I. Clinical periodontology. 1958. Philadelphia, W. B. Saunders Company. Second Edition. pp. 311-323.
- Goldman, H. M., Schluger, S., Cohen, D. W., Chaikin, B. and Fox, I. An introduction to periodontia. 1959. St. Louis, C. V. Mosby Company. pp. 39-63.
- Sorrin, S. The practice of periodontia. 1960. New York, McGraw-Hill Book Company. pp. 1-78.
- Boyle, P. E. “Effects of various dietary deficiencies on the periodontal tissues of the guinea pig and of man.” J.A.D.A. 28:11, 1788-1793, November 1941.
- Hunt, A. M. and Paynter, K. J. “The effects of ascorbic acid deficiency on the teeth and periodontal tissues of the guinea pig.” J. D. Res. 38:2, 232-243, March-April 1959.
- Chapman, O. D. and Harris, A. E. “Oral lesions associated with dietary deficiencies in monkeys.” J. Infect. Dis. 69:1, 7-17, 1941.
- Glickman, I. “The periodontal tissues of the guinea pig in vitamin C deficiency.” J. D. Res. 27:1, 9-23, February 1948.
- Waerhaug, J. “Role of ascorbic acid in periodontal tissues.” J. D. Res. 39:6, 1089, November-December 1960. (abstract)
- Stuhl, F. “Vitamin C subnutrition in gingivostomatitis.” Lancet 1:21, 640-642, May 22, 1943.
- Roth, H. “Vitamins as an adjunct in the treatment of periodontal disease.” J.A.D.A. 32:1, 60-66, January 1955.
- Walsh, R. E. “Pregnancy gingivitis.” Penn. D. J. 51:2, 7-8, December 1947.
- Martin, M. P., Bridgeforth, E., McGanity, W. J. and Darby, W. J. “The Vanderbilt cooperative study of maternal and infant nutrition. X. Ascorbic acid.” J. Nutr. 62:2, 201-224, June 10, 1947.
- Tillman, H. H. “Oral and systemic changes in acute adult scurvy.” Oral Surg., Oral Med. and Oral Path. 14:7, 877-881, July 1961.
- Harris, R. and Hutchinson, G. “Effect of vitamin C on a scorbutic type of gingival lesions.” Oral Surg., Oral Med. and Oral Path. 8:4, 392-399, April 1955.
- Dreizen, S., Stone, R. E. and Spies, T. D. “Oral manifestation of nutritional disorders.” D. Clin. N. America 429-440, July 1958.
- Wellensiek, E. K. “Therapeutic evaluation of bioflavonoids in periodontal disease.” Texas D. J. 74:6, 289-292, June 1956.
- Roth, H., Greene, H. and Kirsch, S. “Preliminary progress report of a clinical study of the effect of C.V.P. on gingival hemorrhage.” Oral Surg., Oral Med. and Oral Path. 10:6, 590-600, June 1957.
- Carvel, I. and Halperin, V. “Therapeutic effect of water soluble bioflavonoids in gingival inflammatory conditions.” Oral Surg., Oral Med. and Oral Path. 14:7, 847-8 5 5, July 1961.
- Todhunter, E. N., Robins, R. C., Ivey, G. and Brewer, W. “A comparison of the utilization by guinea pigs of equivalent amounts of ascorbic acid (vitamin C) in lemon juice and in the crystalline form.” J. Nutr. 19:2, 113-120, February 1940.
- Mindlin, R. L. and Butler, A. M. “The determination of ascorbic acid in plasma: a macromethod and a micromethod.” J. Biol. Chem. 122:3, 673-686, February 1938.
- Ringsdorf, W. M., Jr. and Cheraskin, E. “A rapid and simple lingual ascorbic acid test.” GP 25:6, 106-108, June 1962.
- Ringsdorf, W. M., Jr. and Cheraskin, E. “A lingual vitamin C test for periodontic diagnosis.” Odont. Revy 14:1, 23-31, 1963.