Access to all articles, new health classes, discounts in our store, and more!
Relationship of Vitamin A and Oral Leukoplakia
Published in Archives of Dermatology, Vol. 88, pp. 607-612, November 1963.
* * *
From a review of the literature it appears that local trauma is a factor in the genesis of oral hyperkeratoses.
The major concern of systemic influence has been focused upon vitamin A metabolism. Existing evidence indicates that a vitamin A deficiency can produce oral hyperkeratosis and that therapeutic amounts of vitamin A exert an anti- or dekeratinizing effect on normal or hyperkeratotic oral mucosa.
An attempt was made in this investigation to study, under double-blind conditions (vitamin A versus placebo supplementation), the problem of leukoplakia in terms of its quantitative, qualitative, and histologic attributes.
Within the limits of this experiment, it appears that there is reduction in size, favorable qualitative change in the lesion, and histologic evidence of improvement in a substantial sample of leukoplakic cases subjected to vitamin A supplementation. No such changes were noted in the placebo group.
Within the limitations of this study, there is no clinical and biochemical (serum carotene) correlation.
The clinical and biochemical findings reported here are in accord with the limited studies reported by other investigators.
Review of the Literature
In the past, principal emphasis has been placed upon local irritation as a cause of oral leukoplakia. Recent evidence, however, indicates that vitamin A is playing a role in oral hyperkeratosis.
Vitamin A is necessary for the maintenance of epithelium.1 This includes not only our external covering but the lining of all internal body cavities.1,2 In the absence of vitamin A, cuboidal, columnar, and transitional epithelia tend to degenerate into a stratified, squamous, keratinized epithelium.1-3 This seems to take place through a series of events, including a decrease in the mitotic activity of epithelial cells2,4 with atrophy of the epithelium and a reparative mechanism resulting in keratinization.
The classical human skin lesions associated with avitaminosis A demonstrate hypoplasia and hyperkeratinization. When this occurs, the excessive keratin may cause plugging of the sebaceous glands and hair follicles, resulting in a roughened skin which is subject to chronic infection and eventual dermatosis.5,6 Also, a contributing role played by the B complex vitamins in skin hyperkeratoses has been demonstrated in experimental animals.7,8
Significant changes in the oral mucous membranes of rats and dogs have been observed in a state of vitamin A deficiency. These have been described as hyperplasia of the gingiva with a thickening of the keratin layer.9-11
A reduction in the keratin of normal monkey gingiva has been achieved through the administration of 10,000 USP units of vitamin A daily for 30 days.12 Similarly, in humans, the administration of a 150,000 unit buccal tablet of vitamin A daily for one month significantly reduced the number of keratin cells in normal epidermis in nine out of ten volunteers.13 The obvious antikeratinizing effect of systemic and topical vitamin A in these and other14-16 studies and the favorable response of vulvar leukoplakia17 and senile keratoses18 to this therapy suggested success in treating similar oral lesions.
A number of investigations into the effect of topical and topical-systemic vitamin A therapy on oral hyperkeratoses have been conducted.19-23 Troches containing 150,000 USP units of synthetic vitamin A were employed in divided daily doses totaling 150,000 to 900,000 USP units. In only a few instances was there any laboratory evidence of a vitamin A deficiency. Though these dosages may approach a toxic level if continued,24 no cases of hypervitaminosis A were reported as a result of one to nine months of therapy. Significant reversal of oral lesions, ranging in severity from epithelial hyperplasia to “leukoplakia,” occurred, and no incidence of recurrence was given.
Thus, existing evidence indicates that a vitamin A deficiency can produce oral hyperkeratosis9-11 and that excessive amounts of vitamin A can exert an anti- or dekeratinizing effect on normal or hyperkeratotic oral mucosa.12-23 Since the vast majority of individuals with such oral lesions present very adequate blood levels of vitamin A, there seems to be no correlation between hypovitaminosis A and hyperkeratosis.25 Several authors have suggested that a local tissue depletion may exist even in the presence of adequate blood levels of vitamin A.19,22 This may explain the lack of correlation between blood levels of vitamin A and carotene in the previously discussed reports.19-23 It is possible that local irritating factors (chemical, mechanical, and thermal) may create or intensify the depletion of vitamin A in oral tissues, since their elimination has, in many instances, resulted in a disappearance of a hyperkeratotic lesion. Thus, the degree of tissue saturation may be a needed diagnostic tool.2
Method of Investigation
The patients selected for this study presented a variety of oral lesions all of which showed some degree of keratosis. Patients were obtained from the University of Alabama School of Dentistry, University Hospital Tumor Clinic, and the Birmingham Veterans Administration Hospital. Both nonwhite and white patients of both sexes participated in the study. Table 1 shows the frequency of patient distribution in terms of age, sex, and race. Of the 40 subjects in the initial examination, only 37 returned for follow-up study.
Table 1.Patient Distribution in Terms of Age, Sex, and Race
Clinical Examination.–It is admitted that there is no perfect technique for judging the clinical state. However, within the limits possible, the lesion was graded by quantitative and qualitative means.
Lesion Size: The lesion was measured with a plastic rule calibrated in millimeters. The size and the flexibility of the rule permitted it to be reasonably adaptable within the oral cavity. The measured area was considered rectangular, and the size was recorded in square millimeters. Since most of the lesions were not truly rectangular, the size in area is, in many instances, slightly greater than the dimensions.
Quality of the Lesion: An attempt was made to classify the lesion according to color, texture, and height above the surrounding tissue. The clinical scoring ranged from one to nine with the lowest score being assigned the lesion least pathologic and the highest score where evidence of disease was greatest (Table 2).
Table 2.Clinical Evaluation of Leukoplakic Lesions
Histologic Evaluation.–An attempt was made to secure a biopsy in all cases. The biopsy reports were made by Dr. Leonard H. Robinson, Department of Oral Pathology, University of Alabama Medical Center, Birmingham, Ala. The biopsy findings were described in terms of the absence or presence of epithelial hyperplasia, hyperkeratosis, dyskeratosis, parakeratosis, acanthosis, inflammatory reaction and carcinoma in situ.
Biochemical Findings.–The subtle and most rapid changes in vitamin A metabolism are best reflected in the serum carotene concentration. In contrast, the serum vitamin A level changes more slowly.3,26 Since the period of observation was relatively short (21 days), it was deemed advisable to measure serum carotene rather than serum vitamin A.
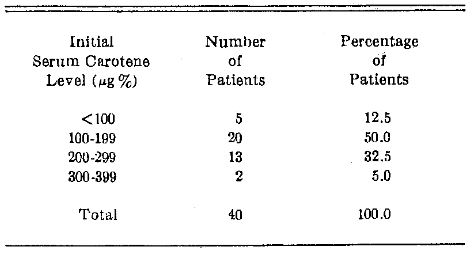
There is quite a wide range of values given by different authors for the physiologic serum carotene level. Normal limits are stated to be from 70µg to 300µg%.3,27-29 The mean carotene level of 12 presumably healthy individuals (without oral leukoplakia) seen in the Oral Medicine Clinic was 160µg%. The mean score for 40 patients (without leukoplakia but being studied for malabsorption syndrome) at the University Hospital was 165µg%. The mean value for the 40 leukoplakic patients in this series was 174µg%. Table 3 outlines the serum carotene levels for the leukoplakia group of subjects.
Table 3. Patient Distribution in Terms of Serum Carotene
Therapeutic Design.–For this study, subjects with keratotic lesions were selected from patients on recall lists with a history of known leukoplakia and from patients initially presenting themselves for treatment of a possible keratotic lesion. Some patients originally chosen and later found to have a malignant lesion were discounted.
At the initial appointment the size and quality of the lesion were determined according to the methods previously outlined. In addition, color photographs were taken, and a biopsy specimen was secured if possible. At the same time venous blood was drawn under nonfasting conditions to determine the serum carotene levels.28
The patients were subjected to one of two therapeutic regimens:(1) vitamin A (Aquasol, 50,000 IU per capsule), and (2) placebo (corn oil, volume per capsule same as Aquasol). At the initial visit the patients were instructed to take a total of eight capsules per day (400,000 IU), preferably two after each meal and two at bedtime. By the use of a table of random numbers, the patients were arranged into two groups. Technicians labeled the capsule boxes by number in order that neither the examiner nor the patient would know which preparation was being administered. The placebo and the vitamin A capsules were compounded to appear identical. The vitamin A group as well as the placebo group were both composed of 20 subjects. There were approximately 21 days between the initial and final appointments. At the second visit the patients were reexamined clinically, biopsies secured if possible, and the serum carotene levels once again determined. There was no knowledge of the previous scores when performing the second examination.
At the first visit 13 of the serum carotene determinations were performed twice by each of the two technicians. This was done to establish the experimental error. The reproducibility (intratechnician error) is made evident by a very significant (P<0.005) coefficient of correlation (r= +0.998) for each technician. For both technicians individually, there was no error in 65% of the cases. The mean error in micrograms per cent for technicians A and B was 2.3 and 2.7, respectively. There was no intertechnician (between technicians A and B) error in 46% of the cases, and the mean intertechnician error was 3.9µg%. The accuracy is heightened by a coefficient of correlation of +0.994 and a P<0.005. Thus, it appears that the experimental error is quite small at both the intra- and intertechnician levels.
Results
Two points deserve reiteration at this time. The leukoplakic lesions were all examined in terms of their size, quality, and histologic picture. Secondly, the only known variable was the introduction of vitamin A versus placebo supplementation. Accordingly, in this section an attempt will be made to answer three questions:
- What is the effect of supplementation upon leukoplakic size?
- What is the effect of supplementation upon the quality of the leukoplakic lesion?
- What is the effect of supplementation upon the histopathologic picture?
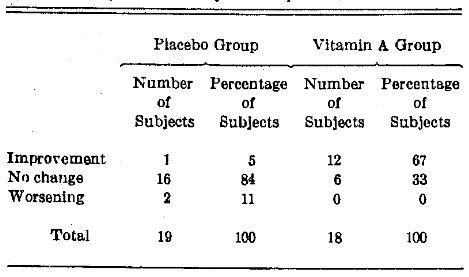
Lesion Size.–Table 4 outlines the percentage frequency distribution change in size of the leukoplakic lesion in the subjects provided with placebo versus vitamin A supplementation. It will be noted that the overwhelming number in the placebo group (84%) remained unchanged with respect to the size of the lesion. In contrast, the greatest fraction (67%) of the vitamin A supplemented group improved.
Table 4. Percentage Frequency Distribution Change in Size of Leukoplakic Lesion
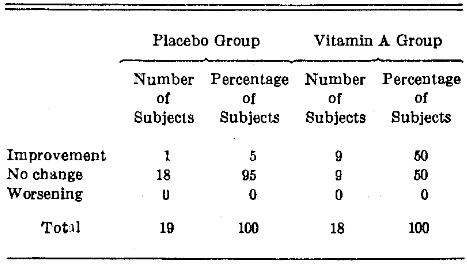
Quality of the Leukoplakic Lesion.–Table 5 summarizes the percentage frequency distribution change in the quality of the leukoplakic lesion after vitamin A versus placebo supplementation. It will be noted that the majority of the patients (95%) provided with the placebo remained unchanged. In contrast, one-half of those given vitamin A demonstrated qualitative improvement.
Table 5. Percentage Frequency Distribution Change in Quality of the Leukoplakic Lesion
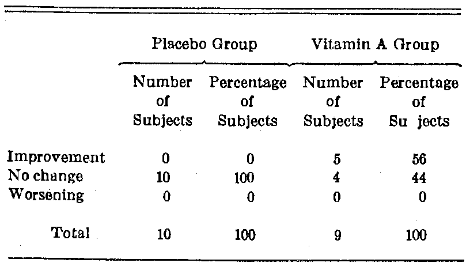
Histopathologic Results.–It is admittedly difficult to quantitate the histopathologic findings. However, admitting its crudity, the biopsy findings at the start and end of the study were classified in terms of the presence or absence of epithelial hyperplasia, hyperkeratosis, and dyskeratosis. It will be noted from Table 6 that no histopathologic change was noted in any of the individuals given placebo therapy. In contrast, 56% of the vitamin A subjects improved.
Table 6. Percentage Frequency Distribution Change in the Histology of the Leukoplakic Lesion
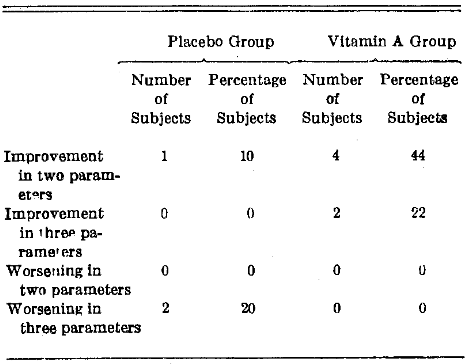
Combination Changes.–Table 7 summarizes the percentage frequency distribution of change on the basis of more than one parameter. Thus, only one subject (10%) given placebo capsules improved in two attributes. In contrast, 44% of the vitamin A group improved under these same circumstances. In all, six of the nine subjects studied showed improvement in two or all of the dimensions measured (size, quality, and histology of the lesion). On the other hand, only one in the placebo group improved, and two worsened.
Table 7. Percentage Frequency Distribution Change of Leukoplakic Lesion in Size, Quality, and Histopathologic Picture
Comment
On a percentage frequency distribution basis, the majority of the vitamin A supplemented group improved in one or more of the methods utilized to measure change. In contrast, the group given placebo medication remained essentially unchanged. Expressed in terms of percentage of improvement, the vitamin A supplemented group demonstrated a 21.4% reduction in lesion size, while the placebo group lesions increased in size by 2.4%. The qualitative improvement for the vitamin A and placebo groups, respectively, was 17.7% and 1.5%. Regression of the lesions, histopathologically, in the vitamin A treated group amounted to 55.5%; however, no evidence of histopathologic change was noted in the individuals receiving placebo supplementation. Not all the subjects given vitamin A improved, and, of those who did, all did not improve equally. It was, therefore, thought advisable to study the possible relationship between serum carotene levels and the clinical response.
In the six vitamin supplemented subjects whose leukoplakia decreased rapidly in size (from 533.3 ± 331.2 to 273.3 ± 275.1 sq mm), it is noteworthy that the serum carotene levels did not appreciably change (P=0.500).
A similar analysis of the six supplemented subjects with no change in size of the lesion also shows no significant change in the serum carotene level (P >0.400).
This lack of biochemical and clinical correlation has been noted by other investigators23,30 who also observed that some individuals with clinical improvement continued to maintain low serum carotene levels.
Two points, therefore, should be emphasized. Firstly, there is no clinical-biochemical parallelism in this study. Secondly, the observations in this study support the limited conclusion of other investigators concerning the beneficial effects of vitamin A upon oral hyperkeratosis.
Limitations of the Study
It is appropriate to add a brief report regarding the shortcomings in this study.
Patient Problems.–In any clinical study with human subjects, there is the chance the patients may or may not follow instructions. In addition, environmental changes during the 21-day period may have played a role. For example, dietary vitamin A consumption may change or infection could influence vitamin A absorption or utilization.
Examiner Problems.–There are no completely satisfactory quantitative and qualitative methods for the evaluation of the oral lesion.
Laboratory Problems.–Inter- and intratechnician error in performing the laboratory tests has already been reported. It was significant that, while the laboratory errors proved small, the technicians did differ, and a particular technician did not always arrive at the same result with the same sample. In addition, the methods of measuring the samples may not be as accurate as they could be to show a true reflection of vitamin A status.
Project Problems.–In addition, there are a number of points in the experimental design which should be mentioned.
Sample Size: It should be recalled that 37 subjects were seen in this study on the two visits. This, indeed, is not a large clinical sample. When subgroups were analyzed, the samples were not large enough to be adequately studied by statistical techniques. For this reason, primary attention was focused on the frequency distribution change rather than the probability values.
Age, Sex, and Race: These factors, though recorded, were not considered in this investigation. This is an admitted shortcoming. However, to subdivide further, according to age, sex, and race, would have produced even smaller groups. These variables may well be most significant.
Time and Dose Factor: It should be recalled the interval between visits was 21 days. It is very possible that some of the group did not actually need vitamin A therapy. With regard to those who required vitamin A therapy, there is no assurance that 21 days or the dosage given were enough.
References Cited:
- Nizel, A. E.: Nutrition in Clinical Dentistry, Philadelphia: W. B. Saunders Company, 1960, pp 154-162.
- Follis, R. H., Jr.: Deficiency Disease, Springfield, III: Charles C Thomas, Publisher, 1958.
- Sherman, B. S.: “The Effect of Vitamin A on Epithelial Mitosis in Vitro and in Vivo,” Invest. Derm. 37:469, 1961.
- Friedenwald, J. S.; Buschke, W. H.; and Morris, M. E.: “Mitotic Activity and Wound Healing in the Corneal Epithelium of Vitamin A Deficient Rats,” Nutr. 29:299, 1945.
- Frazier, C. N., and Hu, C. K.: “Cutaneous Lesions Associated With a Deficiency in Vitamin A in Man,” Intern. Med. 48:507, 1931.
- Frazier, C. N.; Hu, C. K.; and Chu, F. T.: “Variations in the Cutaneous Manifestations of Vitamin A Deficiency From Infancy to Puberty,” Derm. Syph. 48:1, 1943.
- Sullivan, M., and Evans, V. J.: “Nutritional Dermatoses in the Rat: XI. Vitamin A Deficiency Superimposed on Vitamin B Complex Deficiency,” Derm. Syph. 51:17, 1945.
- Kreshover, S. J.: “The Effect of Tobacco Smoke on Epithelial Tissues of Mice as Influenced by Vitamin B Deficiency and Gonadectomy,” Dent. Res. 31:487, 1952.
- King, J. D.: “Abnormalities in the Gingival and Subgingival Tissues Due to Diets Deficient in Vitamin A and Carotene,” Dent. J. 68:349, 1940.
- Mellanby, M.: “Diet and the Teeth: An Experimental Study,” Medical Research Council, Special Report Series No. 153, 1930, London, H. M. Stationery Office.
- Wolbach, S. B., and Howe, P. H.: “Tissue Changes Following Deprivation of Fat-Soluble A Vitamin,” Exp. Med. 42:753, 1925.
- Ziskin, D. E.; Rosenstein, S. N.; and Drucker, L.: “Interrelation of Larger Parenteral Doses of Vitamin A and Estrogen and Their Effect on the Oral Mucosa,” J. Orth. Oral Surg. 29:163, 1943.
- Hunter, R., and Pinkus, H.: “The Effect of Oral Vitamin A on the Number of Keratin Cells of Human Epidermis,” Invest. Derm. 37:459, 1961.
- Flesch, P.: “Studies on the Mode of Action of Vitamin A,” Invest. Derm. 21:421, 1953.
- Flesch, P., and Hunt, M.: “Local Depilatory Action of Some Unsaturated Compounds,” AMA Arch. Derm. Syph. 65:261, 1952.
- Sabella, J. D.; Bern, H. A.; and Kahn, R. H.: “Effects of Locally Applied Vitamin A and Estrogen on Rat Epidermis,” Soc. Exp. Biol. Med. 76:499, 1951.
- Hyams, M. N., and Bloom, O. H.: “Leukoplakia Vulvae: Its Etiology and Results of Treatment With Vitamin A,” J. Obstet. Gynec. 53:214, 1947.
- Savitt, L. E., and Obermayer, M. E.: “Treatment of Acne Vulgaris and Senile Keratoses With Vitamin A: Results of a Clinical Experiment,” Invest. Derm. 14:283, 1950.
- Zegarelli, E. V.; Kutscher, A. H.; and Silvers, H. F.: “Keratotic Lesions of the Oral Mucous Membranes Treated With High Dosage Topical-Systemic Vitamin A,” New York Dent. J. 25:244, 1959.
- Urbach, F.: “The Clinical Use of Vi-Don-A Buccal Tablets in the Treatment of Leukoplakia and Lichen Planus,” Ass. Milit. Derm. 6:17, 1957.
- Mulay, D. N., and Urbach, F.: “Local Therapy of Oral Leukoplakia With Vitamin A,” AMA Arch. Derm. Syph. 78:637, 1958.
- Monica, W. S.: “Topical Vitamin A Therapy in Oral Keratosis,” Times 89:1106, 1961.
- Smith, J. F.: “Clinical Evaluation of Massive Buccal Vitamin A Dosage in Oral Hyperkeratosis,” Oral Surg. Med. 15:282, 1962.
- Hillman, R.: “Hypervitaminosis A: Experimental Induction in the Human Subject,” J. Clin. Nutr. 4:603, 1956.
- Youmans, J. B.: “Vitamin A Nutrition and the Skin,” J. Clin. Nutr. 8:789, 1960.
- Davidson, S.; Meiklejohn, A. P.; and Passmore, R.: Human Nutrition and Dietetics, Baltimore: The Williams and Wilkins Company, 1959, pp 199-206.
- Cheraskin, E.: Diagnostic Stomatology, New York: Blakiston Division, McGraw-Hill Book Company, Inc., 1961.
- Natelson, S.: Microtechniques of Clinical Chemistry for the Routine Laboratory, ed 2, Springfield, IIl: Charles C Thomas, Publisher, 1961.
- Ross, G., and Parker, J. G.: “Serum Carotene Concentration in Normal Individuals and Its Clinical Interpretation,” New York J. Med. 62:3584-3589, 1962.
- Griesemer, C. N.; Frazier,.C. N.; and Blank, I. H.: “Nutritional Influences on Physiology of Skin; Metabolism of Vitamin A,” Medicine 32:293- 321, 1953.