Access to all articles, new health classes, discounts in our store, and more!
Fertilize the Soil, Then the Crop
Published in Commercial Fertilizer Yearbook, July 1947
* * *
As the researches in soil science, plant physiology, and other sciences related to plant production make progress, we are coming to a clearer understanding of the behavior of the root of the plant in relation to its supplies of nutrients and their activities in the soil. As the different sciences contribute new facts relevant to the reactions between the soil and the plant, those who direct the manufacture and utilization of fertilizers may well keep alert to all new methods by which these commercial chemical materials applied to the soil can be made to serve for food production more effectively.
It may be in good order, therefore, to attempt the formulation of a clearer mental picture of the reactions between the soil as a chemically dynamic body and the plant root as an active biochemical one. We need to understand the chemical and physical forces by which the nutrients move from the soil into the plant–or in the opposite direction–to give us plant production which is the basis of all life. Though we may grow plants commercially in water cultures, most plants still grow by means of the fertility coming from the soil. We therefore fertilize the soil to bolster it as the basis of all life.
Nutrients serving only as solutions in soil water for plant use as such is no longer a tenable concept.
That the nutrients in the soil are taken only as there is a water solution of them going into the plant to carry them along, is an ancient and exotic idea transplanted too suddenly from the laboratory to the field. That a plant nutrient must be readily soluble in water is no longer a requisite for its service in the plant, even though this concept may have held on since the day of the lively discussions by such well known scholars of the soil as Dr. Milton Whitney of the United States Department of Agriculture, and Dr. C. G. Hopkins of the University of Illinois. It is not necessary to believe, as some once did, that ionic nutrient elements must be swept into the plant root by a current of water flowing in that direction. Nevertheless, this older view rendered services until there came the colloidal concept of adsorption of nutrients on the clay, and their activities there under their own dynamics, irrespective of the movement of the water.
One of these two pioneers in soil science claimed, in substance, that soil minerals make a saturated solution in water in the soil. Were all of the nutrients of a 2-ton crop of clover, for example, in solution in the soil water at the start of the crop in the spring, they certainly would not be expected to stay there while the heavy rains are going down through the soil to leach them out. We know that we cannot leach much out of the soil, for even the very “hard” water in our wells contains too little to support the growth of household plants in it. Had the soil minerals been very soluble during the long geological ages, would there be even much left as soil at this late stage of time, and in places where we have enough annual rainfall to grow extensive bulk of vegetation? If nutrients are left in the soil in such amounts and forms from which a 2-ton crop of forage can take 150 pounds of them as elements in a 90-day growing season, it seems necessary to discard the belief that they are present there in a simple–and of necessity very dilute–water solution. Conditions like those which we find in the soil demand the belief that the plant nutrients are possibly first taken out of their dilute solutions and concentrated somewhere in the soil to be held for quick delivery to the roots on their searching advance by growth through the soil.
Under such reasoning, then, soluble fertilizers are not rendering their services merely because they go into solution in the soil water, and certainly not because they are retained in that form. If such were true for phosphates, for example, why would we say that “they stay in the surface soil where they are put”? Would we not expect them to be leached down and out of the soil? If it were as simple as solution in water would there have been occasion for putting fertilizers directly with the seed? Would they not be distributed through the moist soil by their own ready solubility and diffusion, or be moved in the percolating water?
If fertilizer activities were so simple there would have been no occasion to have considered putting them into granular forms. Nor would we find the better crop response when a granular fertilizer is mixed through the soil than when, as an infinitely fine powder, the same amount is completely blended into the soil. Nor would a small application of the mill-run, 10-mesh limestone, for example, carry over its effects in the soil much longer than those from 100-mesh, as we know it does.7 Small amounts so well mixed through are too low in concentration and too completely held against movement into the root. Granules represent centers of high concentration and ready migration from there.
This emphasis on making fertilizers that are soluble in water has demonstrated, of course, that they are more effective for quick crop returns. But these more rapid returns have not come because fertilizers stayed in the soil in water soluble form. These returns were not the result of the plant’s taking them along with the water, evaporating this from its leaves and thereby having the salt residue from this distilling process left as nutrients in their tissues. This fanciful picture may not have been your idea of how water-soluble, or so-called “available” fertilizers function to more speedy advantage for crop production. It has, however, been the mental concept of some in the past.
High solubility of fertilizer spells rapid adsorption by the soil colloid rather than by the plant.
We now have a better understanding of the clays as minerals of known chemical composition, molecular construction, and specific chemical behaviors in relation to many plant nutrients. This points out clearly that water-soluble fertilizers are more effective because they react rapidly with the colloidal clay fraction of the soil. They react rapidly also with the organic matter, or the so-called “humus” which, like the clay, is another colloidal performer. Both of these soil components give speed to any chemical reaction because of their extensive and active surfaces which hold also considerable water. These two colloids have a large capacity to take out of dilute solution and combine or hold on themselves many plant nutrient in exchangeable form. If the colloidal clay and the humus did not have these capacities of absorption and exchange these nutrients would not be held against loss as leaching; nor would they be concentrated on one or both of these components of the soil in sufficient quantity to supply the plant during the growing season. More clay in the soil means that the soil can hold more exchangeable fertility and thereby has the capacity to produce more crop. The more hasty growth of a late planted crop on the “gumbo” soil after the spring overflow is possible because of this extra fertility on the clay. More clay means speedy and plentiful delivery to the crop rooms of the fertility adsorbed on it. Adsorbed nutrients on the clay and humus cannot be washed off from there by pure water. They can be removed only as some other ions, whether nutrients or non-nutrients, are exchanged for them.
Water-soluble fertilizers are more effective because they go into solution, yes, but more because in that form their contents of nutrient ions are adsorbed on the clay; are held there against loss by leaching; and are exchangeable to the root as it comes along and offers some other ion with corresponding electrical charge to replace them. Hydrogen is, for example, the most common one offered. It is the water solubility of the fertilizers that spells “availability” to the plants above the soil and to the microbes within the soil through absorption and exchange on the colloids rather than through direct use by the plant and microbe as water solution. These services rendered to the plants by soluble fertilizers–possibly also by nutrients not so soluble in water–are dependent then on their reaction with the colloidal part of the soil and not wholly on their degree of solubility in water. In this one respect then, namely, solubility, we fertilize the soil more than we fertilize the crop.
Soluble salts, as hindrance to seed germination, prohibit heavier applications of fertilizers.
The very fact that fertilizers are such readily soluble salts has been mitigating against more extensive use–and therefore larger sales–of them as long as they were applied in direct contact with the seed. Damage to seed germination by fertilizers is not news to many farmers who became generous in amounts of fertilizers applied in the row. The numerous trials of a specific placement of fertilizers at the side of, or below, the seed in the soil are merely a part of the effort to use more fertilizer but yet escape the danger of injury to the germinating seed, or the growing seedling, in contact with the salt that has been dissolved in the soil water but not yet adsorbed out of that solution by the soil colloids. The success of such placements through more complete absorption by the soil colloid of the fertilizer helps us to understand why fertilizers on sandy soils with little colloid may be more dangerous in these respects than on those of heavier texture.
Isn’t the placement of fertilizers at some distance from the seed more effective merely because it is a means of keeping the seed out of the salty soil area; of allowing time for the soil to absorb the fertilizer and thereby take it out of solution before the plant roots arrive; and of permitting the roots to act under their own stimulus of growing into the particularly suitable concentration of the adsorbed fertilizer? Such a concept of the soil conditions suggests that fertilizer placement is giving chance for the roots to avoid the danger of salt injury and to grow into the soil areas of suitable degrees of its concentration in the adsorbed forms on the clay rather than compelling them to be closely surrounded by fertilizer salts. Making fertilizers so highly water-soluble has worked against their own more extensive use so long as they were applied directly with the seed. The concept of fertilizing the crop, rather than the soil, has, as it were, held back the advance of the sound business of wider use of fertilizers for greater crop production. Now that we are thinking more about fertilizing the soil, they are being used more extensively both in new areas formerly not fertilized and in heavier applications for bigger and better crop yields.
Fig. 1. adsorbed NUTRIENTS ARE MORE EFFECTIVE THAN THOSE IN SOLUTION. Nutrients adsorbed on colloidal clay in dilute suspension and poured over zonolite (right) grew better marigolds than the same nutrients in water cultures handled similarly (left). Photo by Missouri Agr. Exp. Station.
Now that we are plowing the fertilizers down, much larger amounts per acre are being safely applied. There is no recognized damage to seed germination under these conditions. There is less damage later in the plant’s growing season. Corn can be given more nitrogen for use at the later time in its growth or the time when it commonly “fires.” This trouble, so often ascribed to drought, can be eliminated. Fertilizers in this case are put down not only with the immediate crop, but also with the succeeding crops as well in mind. Such fertilizer practice is moving–in thought at least–toward fertilizing the soil more than the crop and toward using fertilizers for maintaining the fertility of the soil rather than only for stimulating the early growth of the crop to which it is directly applied.
Calcium suggests its protective effects.
Fortunately, not all fertilizers have been “Frankensteins” to the same degree as some when used in heavier applications with the seed. Ordinary superphosphate in contact with the seed is less disastrous than salts of nitrogen and potash. Various explanations have been offered for the damage by these salts. We have not been giving consideration to the hypothesis that calcium in the gypsum of the superphosphate may possibly be providing a protective action.2 But when Professor Rodney H. True of the University of Pennsylvania, demonstrated the loss of cations, i. e., the positive ions, by the plant root to a dilute solution of potassium salts into which it was immersed, but an uptake of both potassium and calcium from this same solution of potassium given some calcium as supplement,9 he was giving possible explanation of these differences in dangers from different fertilizers to the germination of the seed and to the emergence of the plant.6
Additional possible support to this hypothesis about the protective effects of calcium may be gotten from the fact that in numerous trials of soybeans growing on colloidal clay, it has always been requisite that calcium should move into the crop in order to get plant growth. But growth has been possible even if too little calcium went from the clay into the root to prevent nitrogen, or phosphorus, or potassium, from going back from the root of the plant to the colloidal clay.1 Isn’t it possible that calcium, and possibly some other nutrients, may be playing a role in the root conditions that move the nutrients into the plant from their adsorbed stage on the clay, rather than allowing them to go in the opposite direction as the case of the injury to germination and the injury to even the growing crop may possibly be? Here may be reason, in part at least, why the so-called “neutral” fertilizers, made so by additions of calcium and magnesium, are superior to those not so treated and therefore considered to be “acid” fertilizers. Here may be reason why fertilizers are so much more effective on limed land than on that unlimed.
Adsorbed ions move under their own power.
We can now believe that it is not necessary that water flow into the roots to carry nutrients there. We can now believe, too, that the nutrient ions move from regions of higher concentrations of adsorbed forms to those relatively lower, much as gases diffuse in the air; or soluble salts diffuse through water; or as water itself moves through the soil under capillary forces. Root surface areas of most any crop are not large enough to give it sufficient nutrients if it were to take only those adsorbed on the clay face or that surface in immediate contact with the roots. Since crops under experiments took amounts of nutrients much larger than those on the surface of one molecular clay layer,5 it became necessary to test whether the exchangeable ions adsorbed on the clay could move from soil areas of higher to those of lower concentration while no water was flowing.
By using a sand mixed with colloidal clay carrying adsorbed calcium and by placing this mixture into a pan on one side of a removable metal partition, there could be put some more sand with clay carrying adsorbed hydrogen or acidity on the other side. It was possible then to bring these two mixtures into intimate contact by dropping the pan after the partition had been removed. Tests at time intervals of the sand-clay mixtures on the two sides of their line of juncture showed that the hydrogen was moving from the acid-clay into the calcium clay, while the calcium was going in the opposite direction, both at the rates of more than an inch per month.5 These mixtures were kept covered and lost no water. So, without water movement, these insoluble but exchangeable ions held on the clay were moving from higher to lower concentrations. They were adjusting themselves toward their equilibrium as adsorbed ions even when no water was moving.
Fig. 2. FERTILIZERS PUT WITH THE SEEDING MAY DAMAGE THE CROP. Increasing amounts of ammonium sulfate put into the soil with the seeds prohibited emergence of the young plants (upper photo), while such was not the case where superphosphate was used (lower photo). (Figures denote pounds per acre if in five-foot rows.) Photo by courtesy John Bushnell, Ohio State Agr. Exp. Station.
In the light of such facts, fertilizer action in the soil is a case of having the nutrients present on the clay and in the exchangeable forms at concentrations high enough to move significant quantities of their ions toward the lower concentrations within the root while it is growing through the soil. It is certainly not logical to visualize the nutrients being carried into the plant by a flow of water that might in any sense wash them out of the soil and into the roots. It is the concentration of the nutrient ions adsorbed on the clay, as given it by whatever kind of fertilizer there is–whether readily soluble or Iess soluble–and not necessarily the water solubility in laboratory tests that we need to visualize as important in any fertilizer service by way of the soil to the plant. This suggests obtaining a higher effectiveness from fertilizers put into the soil as granules to give a few scattered areas of high saturation, than from a uniform blending of it throughout the entire soil mass.
Soil acidity as well as calcium makes fertilizers more efficient.
The confusion in our understanding of soil acidity has brought confused thinking about “acid” and “neutral” fertilizers. Since we now believe that soil acidity is mainly a deficiency of soil fertility, this acid soil condition which is recognized so readily by simple soil tests, represents in reality a better market for more fertilizer as well as for lime. Then, too, if soil acidity is in reality a benefit rather than a detriment, as we understood it is,4 acid soil conditions suggest that calcium and magnesium are serving as plant nutrients even when put into the fertilizers for the purpose of making them neutral in reaction. These two nutrient elements are serving for a “synergistic” effect, as Professor True called it, when they are making natural fertilizers that are better crop producers than are those fertilizers left in an acid reaction.
Nutrient ions coming from fertilizers put on clays carrying some acidity will move more efficiently into the crop than those on clays distinctly neutral. More efficient use will result, therefore, if the fertilizer carries calcium, and if this element is adsorbed on the clay fraction which retains some hydrogen, or some degree of acidity, at the same time. It is the fertilizer’s content of plant nutrients, including the calcium, and not the reaction or the degree of acidity expressed as pH of its ash contents, that comes into consideration in what has been considered a question of fertilizer reaction.3 It is only the better understanding of the chemical reactions between the fertilizers and the clay of the soil, and between the clay and the plant roots, that will clear up the confusion prevalent regarding the acidity of the soil and that of fertilizers.
Acidity of soil mobilizes nutrients from mineral reserves.
That the acidity of the soil should have been considered such a serious danger to the crop production seems strange when we realize that most of our food production has been on soils that are acid. Then, too, it has been the acid soils that made fertilizer sales and their use in agriculture the big business and the return on the investment that they are. If sulfuric acid in the manufacture of fertilizers makes the phosphorus of bone or rock more available for plant use, can we not likewise picture the acid of the soil, i. e. the hydrogen adsorbed on the clay, breaking down the rock to mobilize the nutrients in the mineral reserves of the soil? Through the same manner, we can see that the calcium applied as lime-rock fragments becomes all the more active and the more available for use in nourishing the plant.7 Has the total fertility delivered by naturally productive soils these many years been no larger than the total exchangeable supply on the clay as we measure it in the exchange capacity at any one moment? Could that supply have been maintaining the productivity during all this time without restocking the clay many times over from some reserve supply? When we use fertilizers in the soluble form to stock the clay with exchangeable nutrients, are we not providing there only the more temporary supplies? What are fertilizer practices contributing toward the maintenance of a more permanent and possibly more wisely balanced soil fertility? These are challenging questions to those who hope that the properly designed soil treatments may not only cure soil troubles temporarily, but may give benefits reaching beyond the few post-application years. They are the more challenging, but also the more hopeful as we think first of fertilizing the soil and then of fertilizing the crops.
When we consider that our soils have been producing crops these many years, surely it cannot have been only the supply of nutrients held in exchangeable form on the clay that was serving. An examination of the output of fertility by a soil like the Putnam silt loam (Sanborn Experiment Field), for example, during its cropping history points out clearly that the nutrient supply in the clay cannot have been the only source of nourishment for the crop. There must have been some activity mobilizing the reserve rock or mineral nutrients to restock the clay and to hold up its supply at some level suggesting equilibrium with the restoring forces. This soil has an exchange capacity of 15 milligram equivalents per 100 grams of soil. Of this capacity, even today scarcely 4.00 equivalents are taken by hydrogen or acidity. This clay still has itself relatively well stocked with nutrient cations that include calcium to the extent of almost 50 per cent of the exchange capacity. If the regular cropping of this soil at the rate of 2 tons of clover hay, 25 bushels of wheat, 40 bushels of oats, and 50 bushels of corn with all crops removed in this 4-year rotation had been getting only its calcium, potassium, and magnesium by trading hydrogen to the clay for them, then the hydrogen accumulated on the clay to date would represent the nourishment of these crops during less than seven rotations or a cropping duration of less than 28 years. Yet these soils have been cropped under record for more than twice that long. If such crop yields continue for such periods of time as we know they have in the Cornbelt; and if plants can get their nutrients only by trading hydrogen or acidity into the clay; surely then, if there has been no more acidity accumulated in these soils while we are not adding fertility there must have been a neutralization of it suggesting that this is brought about through the breakdown of the soil minerals representing reserve fertility.
That the acid clay can play a role in this respect has been demonstrated by Dr. E. R. Graham,7 when he separated the colloidal clay fraction from the Putnam subsoil, and, after converting it by electrodialysis to an acid clay completely saturated with hydrogen, mixed this with the carefully washed silt fractions of different soils. Observations revealed that some silt minerals were reacting with the clay acid to neutralize it while others were relatively inactive. This effectiveness was correspondingly less as the silt minerals represented soils in an arrangement from lower to higher annual rainfalls across the central portion of the United States west to east.8 When the mixtures of silt and clay were planted to crop growth, the silts were more productive as they were originally from less weathered soils and as they consisted more of minerals other-than-quartz.
Here is a clear suggestion that we may well think that the minerals of the silt fraction of some soils are being broken down by the acid clay as a weathering agent for them. The rate at which this reaction occurs for the different minerals and the ratio of the clay to the silt fractions of the soil determine how rapidly the clay, once exhausted by one crop growth, can be restocked with nutrient supplies for the next one. Here is a dynamic force in fertility renewal that has not been fully recognized as a variable in connection with fertilizer practices in one section of the country as compared with those needed in another. It is also suggesting that when values of fertilizer materials are determined wholly on the basis of increased crop returns, this criterion may represent undue credit to this soil treatment because fertility is also delivered by the soil’s mineral reserve and is not measurable as stock on the clay by soil tests. When natural production has been going forward these many years by means of nutrients taken from the clay in total amounts which no clay could carry in exchangeable form as a single stock, there is reason to believe that the reserve minerals may be significant contributors. The nutrient reserves in the silt minerals of the soil and their mobilization by the dynamics of the acid clay and acid humus as colloids deserve recognition in soil management. This seems wise even before we accept the introduction of water-soluble, concentrated, salts of nutrients into the soil for adsorption on the clay as the main concept of the dynamics through which plants are nourished by the soil.
High content of organic matter in soils often spells security against bad fertilizer practices.
The generous supply of organic matter and probably the reserve minerals, especially the limestone, in our soils have been absorbers of the shock to our soils by bad fertilizer practices. Since organic matter is the source of energy for the microbes, their increased activity resulting from the added fertilizers may do much to restore the soil from its chemical upset and to lessen the shock on the crop by this soil treatment. This absorption of the chemical shock is bought at the price of some of the organic matter that is rapidly depleted unless some fertilizers are used to grow crops for its restoration in the soil. Isn’t it probable that the plowing under of fertilizers may be partial prevention against the so-called “burning out of organic matter by fertilizers” if they are put below the humus-rich surface layer and in the deeper, more moist horizon away from some of the microbial competition with the plant roots for them?
Fig. 3. MINERAL RESERVES IN SILT ARE WEATHERED OUT BY ACID CLAY. Better soybeans resulted as the substrate growing them consisted of the silts taken from soils farther west and mixed with acid clay (free of nutrients) to test the mobilization of the nutrients from the silt minerals by this acid fraction of the soil. Photo by Missouri Agr. Exp. Station.
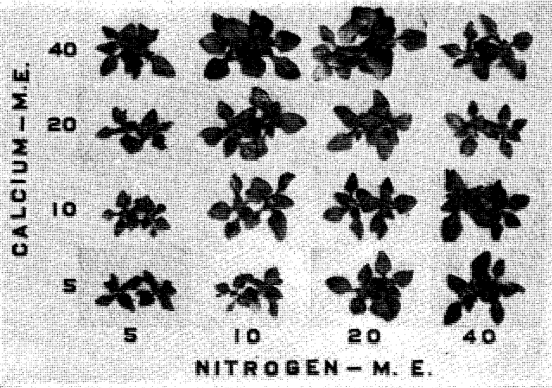
Fig. 4. FERTILIZER USE MUST CONSIDER THE SOIL AS WELL AS THE CROP AND THE FERTILIZER. Increasing the amount of nitrogen applied (left to right) gave
(a) little extra growth when the exchangeable calcium of the soil was high (upper row),
(b) extra growth when the exchangeable calcium of the soil was medium (middle row),
(c) some but less effect when the exchangeable calcium of the soil was low. Photo by Missouri Agr. Exp. Station.
Organic matter and mineral reserves in the soil may have been doing more than we realize in preventing troubles in our applying large amounts of a highly soluble single nutrient. Phosphorus has been used in wide variations in the amounts applied. Nitrogen classifies under the same kind of practices. Much remains to be learned before we can apply fertilizers in combinations representing balanced amounts for the particular soil and crop. Much of the confusion about rates of application of fertilizers results from failure to know what the soil has to offer in its decomposed organic matter, and in its weatherable minerals as well as on its clay in exchangeable form. Balanced fertilizer on the soil in the fullest sense of those terms cannot be a ready realization when we are not only juggling the three common elements, nitrogen, phosphorus, and potassium to be applied, but when the soil itself is highly variable in its delivery of nutrients from the lime applied, the decaying organic matter, and the mineral reserves. The more nearly correct answer can be given only by several years of experience in growing the crops under soil treatments. If the organic matter in the soil could be kept at a high level and if the fertilizer materials applied were less soluble, our juggling act would not be so trying. There is good reason for using a part of the fertilizer to grow organic matter into the soil so that it may be insurance against the possible dangers lurking in our incomplete knowledge at this time about balancing fertilizers more wisely for each soil.
Concepts to date of reactions within the soil.
Fortunately, we are making rapid progress in our understanding of the dynamics of the soil. We are gearing fertilizers into them more effectively. The concepts developed to date of the reactions between soluble fertilizers and the soil, and between the soil and the plant roots visualize these behaviors about as follows: (1) Water soluble nutrients, like the nitrogen, phosphorus, and potassium of fertilizers, are not taken up by the plant as water solutions. They react, rather, with the soil to become adsorbed on the colloidal clay and humus. High solubility of fertilizers means more rapid reaction with these colloids. It is often not rapid enough in drier soils, however, to be without salt injury to germinating seeds if they and the fertilizer are put simultaneously into the same soil area. (2) Nutrients adsorbed on the clay are taken by the plant root mainly as it exchanges hydrogen or acidity for them. (3) Calcium probably plays an important role in the root conditions by which nutrients are taken, or moved, from the clay into the root rather than in the reserve direction. (4) These beneficial effects by calcium are seemingly greater if some hydrogen or acidity accompanies it. (5) Consequently, soil acidity is beneficial not only in making the calcium more efficient but also in mobilizing other nutrients into the exchangeable forms, including those in the silt mineral reserves. (6) The complexity and numbers of the chemical reactions and interactions within the soil emphasize the insufficiency of considering only the inorganic aspects of them, and suggests that the soil organic matter may represent benefits not yet well understood.
Fertilizing the soil as a help toward its better feeding of the crop is still much of an art. But recent contributions from soil science and plant physiology have been numerous enough to encourage the belief that we are fitting fertilizers more accurately into the conditions dictated by the soil. We are, thereby, moving toward better plant nutrition through an embryo science, at least, of fertilizer use. Much is still ahead as we continue to learn first to fertilize the soil and then to fertilize the crop.
Fig. 5. FERTILIZER USE MUST CONSIDER THE SOIL AS WELL AS THE CROP AND THE FERTILIZER. Increasing the amount of phosphorus applied (left to right) gave
(a) Pronounced extra growth when the exchangeable calcium and the nitrogen were high (upper row),
(b) some but less effect when the exchangeable calcium was high and the nitrogen was medium (middle row)
(c) no significant effect when the exchangeable calcium and the nitrogen were low (lower row). Photo by Missouri Agr. Exp. Sta.
Fig. 6. SELECTION BY INSECTS SUGGESTS THAT THEIR INJURY MAY BE DUE TO IMPROPERLY BALANCED FERTILITY. Injury by thrips to spinach plants occurred initially only on plants given lower amounts of nitrogen. More calcium made limited amounts of nitrogen more efficient protection against injury by these insects. Photo by Missouri Agr. Exp. Sta.
References Cited:
- Albrecht, Wm. A., and Smith, N. C.: “Saturation Degree of Soil and Nutrient Delivery to the Crop.” Jour. Amer. Soc. Agr., (1940) 148-153.
- Albrecht, Wm. A.: “Calcium as a Factor in Seed Germination.” Jour. Amer. Soc. Agr., 33 (1941) 153-155.
- Albrecht, Wm. A.: “Calcium-Bearing Versus Neutral Fertilizers.” Commercial Fertilizer Year Book, 1941.
- Albrecht, Wm. A. and Schroeder, R. A.: “Plant Nutrition and the Hydrogen lon. I. Plant nutrients used most effectively in the presence of a significant concentration of hydrogen ions.” Soil Sci., 53 (1942) 313-327.
- Albrecht, Wm. A., Graham, E. R., and Shepard, H. R.: “Surface Relationships of Roots and Colloidal Clay in Plant Nutrition.” Amer. Jour. of Botany, 29 (1942) 210-213.
- Albrecht, Wm. A. “Plant Nutrition and the Hydrogen Ion. Relative effectiveness of coarsely ground and finely pulverized limestone.” Soil Science, 61 (1946) 265-271.
- Graham, E. R.: “Acid Clay–An Agent in Chemical Weathering.” The Jour. of Geology, XLIX (1941) 392-401.
- Graham, E. R.: “Soil Development and Plant Nutrition. I. Nutrient delivery by the sand and silt separates.” Soil Science Soc. of Amer. Proc., 6 (1941) 259-262.
- True, Rodney H.: “The Function of Calcium in the Nutrition of Seedlings.” Jour. Amer. Soc. of Agron., 13 (1921) 91-107.