Access to all articles, new health classes, discounts in our store, and more!
Absorbed Ions on the Colloidal Complex and Plant Nutrition
Published in Soil Science Society of America, Proceedings 1940, Vol. 5
* * *
Adsorption of ions and their exchange by colloids have done much to provide a clearer concept of the mechanism of those soil and plant root interactions, commonly spoken of as plant nutrition. There has always been a wide gap between the behavior of soil in the test tube by which its stocks of plant nutrients are measured, and its behavior under test against the plant root as computed in terms of crop yield and crop composition. Reasoning from the chemical behavior of the soil in the laboratory to the crop behavior in the field has corresponded to a jump across a tremendous abyss into which most reasoners eventually have found themselves plunged. With the clearer concept of the chemical behavior of the colloidal clay fraction of the soil as it may take, or may give, nutrient cations and anions, it is now possible to bring the clay composition and the plant growth together, and to observe with laboratory accuracy their chemical interactions. We are narrowing the abyss, not only by pushing the chemistry of the soil closer to the plant behavior, but are also using the plant metabolism as a biological reagent–possibly more delicate than chemical reagents–to give suggestions regarding the chemical nature and behavior of the colloidal clay fraction of the soil. Such suggestions are already numerous enough to warrant presentation of some at this time for critical examination and further experimental verification.
Colloidal Clay Versus Aqueous Solutions as Growth Medium
The soil solution has long been found inadequate as explanation for the relatively generous delivery of nutrients by the soil to the plant, particularly of the element phosphorus. Even the aqueous nutrient cultures may soon be discarded as media simulating plant behavior in the soil. Such solutions demand carefully controlled concentrations, osmotic relations, and other physico-chemical conditions that are quickly upset with only partial removal of the ions by the plant. The nutrient delivery per unit of solution volume is very low, and even then there is the danger of nutrient excess. Only the more experienced plant physiologist with continually renewed dilute solutions seems successful with this research tool. On the contrary, the colloidal clay as a nutrient medium is the haven of safety within which the most embryonic plant physiologist may sail about naively yet successfully. The colloidal clay offers its ease of suspension but yet a low solubility; its large supply–even to excess–of adsorbed nutrient ions but nearly constant physico-chemical conditions; and its capacity to remove from solution those injurious items bringing about what is commonly known by that cause-concealing term of “toxicity.” It permits a wide range in kind and amount of nutrient offerings to plants under experiment while other conditions so disconcerting by their fluctuations in nutrient solutions remain almost constant. At a pH of 5.0 in an aqueous solution, for example, the presence of 0.01 mg hydrogen per liter is an approach to the danger point, while in a 2% colloidal clay suspension at the same pH there would be 650 times as much hydrogen with no great danger. When the chemistry of the behavior of anions on colloidal clay is understood as well as that of the cations, then the colloidal clay medium will permit research in plant nutrition in the laboratory for interpretation in terms of field results with a satisfaction to which aqueous nutrient cultures can be no equal.
Simplified Concept of Nutrient Absorption by Plants
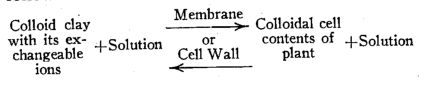
The ordinary equation of a chemical reaction at equilibrium may be helpful in formulating a concept of plant root and colloidal clay interactions. Suppose we consider it as a case of colloidal clay suspension with possibly some ions in solution as the left side, and the plant cell protoplasm, or a colloid, plus its aqueous accompaniment as the right side of the equation. Then in place of, or rather along with, the arrows between and pointing in opposite directions, we must interpose a membrane or the wall of the root hair. This may be represented as follows :
Imagine, further, the removal of the water to varying degrees or to the point of eliminating the solution phase on each side of the equation. Then we can write it as a case of soil colloid and plant colloid on opposite sides of the cell wall of the root hair. This brings us to the concept of two colloids in contact. We can believe them at equilibrium or as exchanging ions in either direction as regular chemical laws dictate, except for the modifications caused by the nature of the membrane and its changes in relation to the colloidal interactions or the plant metabolism products.
Unfortunately, we know very little about the chemical properties of the plant colloid in vivo. Chemical behavior of the plant cell contents, like the goose that laid the golden eggs, does not submit readily to internal observation. Thus, the conditions prevailing on the right side are not well known. In addition, the time factor, as a kind of fourth dimension, must be introduced. Displacement of equilibrium by the plant is a matter of a growing season of, say, 100 days and not an instantaneous performance. Thus, we can measure the accumulated displacement result on the right side of the equation, only after that interval of plant growth at which we choose to make analysis of the plants.
More fortunately, the colloidal clay and its properties, its behavior, and possible changes are known definitely enough to serve more nearly as the known side of the equation for solving the plant unknown side. The beidellite type of clay isolated from the claypan layer in the subsoil of the Putnam silt loam has been subjected to enough physical, chemical, mineralogical and other studies to establish its relative constancy in behavior as an anion,1 and its capacity for wide variation in kind of, and degrees of, saturation, by nutrient cations and anions. Thus, the relative concentrations of exchangeable ions are controlled by their degree of saturation on the clay. The total amount of ions is controlled by this character coupled with the amount of clay offered the plant. This simulates then the degree of ionization and the concentration of ions in the ordinary solutions.
By means of this simplified approach with considerable knowledge of the properties of the clay and its changes on the left side of the equation, we may observe, or measure, the plant growth behaviors, the incidence of plant disease, and the seed and plant compositions with their indications of the movements of both cations and anions from the colloidal clay to the plant, or in the reverse direction, or any other plant manifestations and clay changes, all as helps to interpret what has happened chemically on the right side. From such we may learn whether plant nutrition may not finally conform to the more commonly accepted laws of chemical behavior.
Calcium the Most Important of the Adsorbed Nutrients
Because several nutrient cations are required for plants, some preliminary trials were conducted in order to ascertain their relative importance. The choice of a legume as the plant for experimental service eliminated one nutrient, namely nitrogen, from the medium, because of its introduction into the plant from the atmosphere. The use of the soybean plant with a large seed of selected size and constant composition removed the necessity of providing on the clay medium those ions used in very small amounts but amply supplied through the seed for the early life period at least. Farmer experience of the common failures of legumes without limestone treatment pointed to calcium as the first nutrient warranting consideration as the limiting factor on the clay, particularly that from claypan soils.
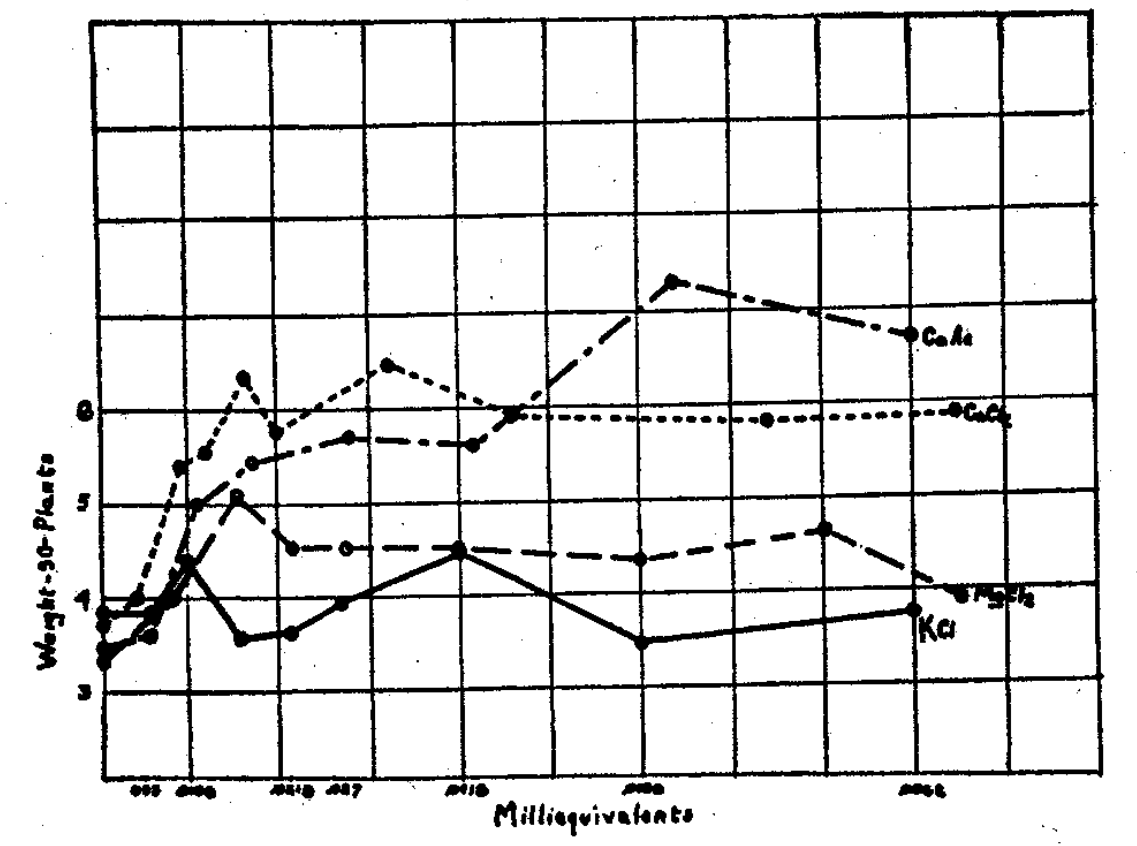
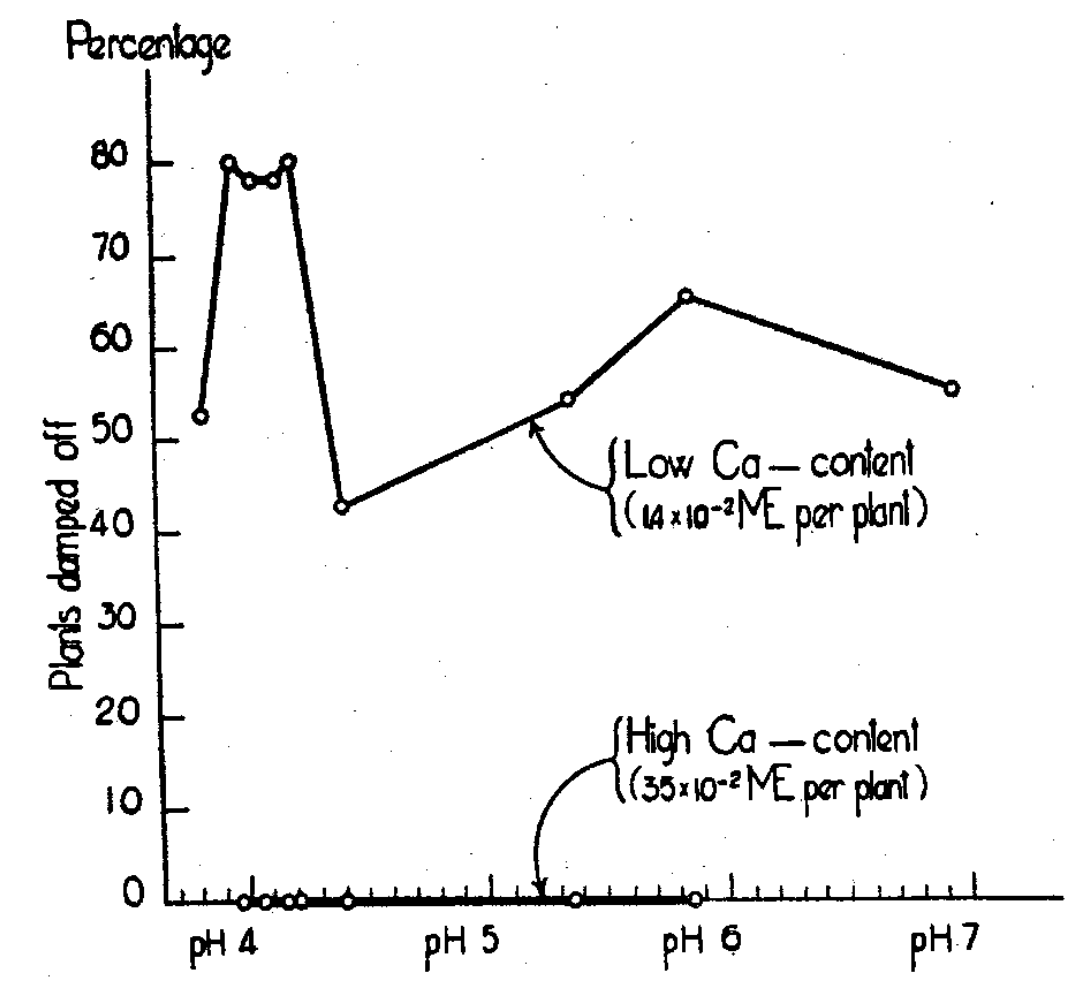
Aqueous nutrient solutions served to demonstrate calcium as the first requisite5 for growth of soybeans (Fig. 1). Much better growth occurred when potassium and magnesium were not supplied than when calcium was absent (Fig. 2). Incidence of disease with low calcium, and conversely healthy plants with high calcium, showed calcium requisite for growth (Fig. 3). Clay on which only calcium was adsorbed produced growth that improved with increased clay (Fig. 4). It was superior in giving growth over a wider range of calcium offered the plant than was possible by aqueous solutions (Fig. 5). These results suggested that calcium occupying so large a portion of the adsorbed and exchangeable store of cations on the clay becomes plant nutrient number one in importance, and even for the soybeans, a supposedly “acid-tolerant” legume.
Fig. 1–Calcium is the first requisite among cations required by plants. Its absence encouraged incidence of diseased plants.
Fig. 2–Magnesium and potassium failed to give growth equal to that by calcium.
Irregularities in the growth response by legume crops to limestone treatments threw doubt on the belief that the hydrogen ion concentration of the soil, or its pH, is the causal factor in legume crop failure. Some acid soils, failing to grow clover, were given limestone and showed no measurable change in pH after a year but yet produced clover successfully. This beneficial effect by the added calcium, when there was no change in pH, pointed to calcium deficiency in the soil rather than to an injury by the excessive hydrogen ion concentration as the problem of so-called “acid” soils. Electrodialyzed hydrogen clay on which the exchangeable hydrogen was neutralized by calcium hydroxide to varying degrees to give soils of different pH values, provided means of separating the effects by the hydrogen ion concentration from those by the amount of calcium.1,2 Controllable and variable amounts of calcium could be offered to the plant at any pH by varying the amount of the clay that had been titrated to any particular pH. Plant growth on such a series of clays in sands varied according to the degree of acidity, or pH, but was influenced far more by the amount of calcium offered to the plants (Fig. 6). Thus, in trying to relate plant growth to the pH of the soil, the facts indicated that it is related in reality to the approximate reciprocal of the hydrogen saturation and ionization, namely the calcium saturation.
Fig. 3–Plant disease shows relation to the calcium level rather than to pH.
Fig. 4–Increasing the clay to deliver more calcium at pH 4.4 (left to right) gives better growth.
Fig. 5–Adsorbed calcium on Permutit is as effective as ionic calcium.
(Increasing calcium, left to right.)
Nutrient Cations May Move From Plant to Soil
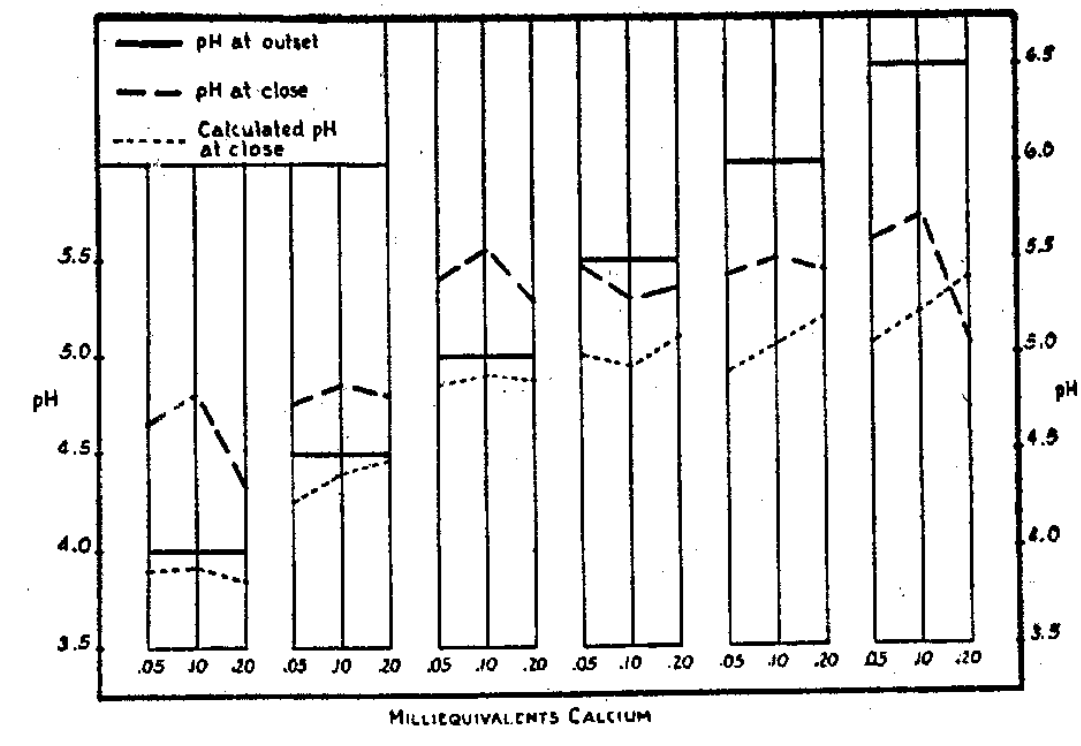
Changes in the pH of the colloidal clay medium as the result of the plant growth pointed to a displaced equilibrium, but a displacement toward both the right and the left. By using the analyses for calcium of the seed and clay at the outset and again of the final plants as a means of determining the direction of movement of the calcium, and then by calculating its amount left in the clay and the corresponding pH at the close of the growth of the series, it was discovered that the pH figures for the clay by determination were higher than those by calculation (Fig. 7). In other words, the clay was less acid than it should have been by the calculations based on the calcium removal and its assumed substitution by hydrogen. Increase in calcium in the crop over that in the seed established movement of calcium from the clay soil to the seed in every case, with growth parallel to the amount of calcium delivery. The fact that the pH was not lowered as calculated pointed to a return to the clay from the plant of some elements other than calcium, serving as bases or cations. to raise the pH of the clay. Whether anions of plant origin were also moving to add to the confusion may well be considered.
Fig. 6–Soybean growth according to different calcium levels at different degrees of acidity of colloidal clay.
Fig. 7–Changes in pH of clay in consequence of soybean growth.
Here was the first indication that exchange cations–possibly nutrients–may move from the plant to the soil as well as from the soil to the plant. Certainly, as shown by analysis, the element calcium did not go from seed back to the soil. Instead it moved into the plants. Its equilibrium was displaced by movement to the right. At the same time, some displacement toward the left occurred because of movement of other cations in that direction.
In the clays with pH figures below 5.5, the reduction in pH by calcium removal and hydrogen substitution (dotted lines in figure) corresponded to approximately 0.12 pH as average. In clays with pH values above 5.5, the change in pH through calcium removal was greater with increasing values of the initial clay, namely 0.45, 0.90, and 1.25 pH for clays at pH 5.5, 6.0, and 6.5, respectively. Equilibrium displacement by calcium removal from the clay was greater as the clay was more nearly saturated by calcium or as its pH figure was higher.
In spite of the calcium removal from the clay, a reaction which should have lowered the pH, measurements of this property of the clay reported the pH varying from those obtained by calculations by some rather consistent differences. These differences were not related to the pH level of the clay growing the crop. They were seemingly related inversely to the total calcium offered to the crop. Offerings of 0.05 M. E. of calcium resulted in differences which averaged 0.55 pH. For calcium offerings of 0.10 M.E., the change was but little less and for 0.20 M. E. of calcium allotted it was a shift toward alkalinity by only 0.25 pH.
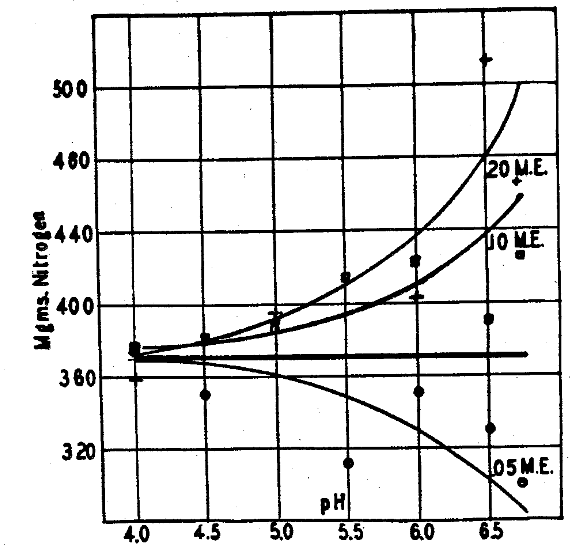
Nitrogen determinations of the seed and crop showed losses of this element at the pH figures below 5.5.2 Here may have been a cation that was going back to the clay in the form of ammonia to make it more alkaline. But since an increase in nitrogen in the system occurred at pH 5.5 and above for the offerings of 0.10 and 0.20 M. E. of calcium per plant (Fig. 8), there was nitrogen fixation or use of atmospheric nitrogen. This raises the question whether nitrogen fixation may be going on while losses of it to the soil are occurring at the same time. Since the pH shifts were so consistent for the calcium offerings at all six pH levels used, it seems doubtful if the plant losses of ammonium ion to affect this change could be so consistent when coming from such widely varying sources as seed only in some cases and from seed and atmospheric fixation in other cases. Doubtless this shift toward higher pH by cation movement from seed to the soil must be ascribed to cations other than the ammonium of seed origin, in this case using a calcium hydrogen clay delivering only calcium to the plant.
Fig. 8–Nitrogen fixation as correlated with calcium levels rather than with the degree of acidity. (Horizontal line represents nitrogen content of seed.)
Calcium Movement Related to Degree of Calcium Saturation of Clay
In order to list more accurately the significance of the degree of calcium saturation in the delivery of this element to the plant, clays were prepared with different degrees of calcium saturation, ranging from forty per cent to complete saturation. The balance of the exchange capacity of the clay was taken individually by hydrogen, giving variable acidity, by barium, magnesium, and potassium, all giving complete neutrality in the form of readily exchangeable ions; and finally by methylene blue, a non-exchangeable ion of a large and an organic mass. Such quantities of clay were added to sand as would supply equal amounts of calcium per plant.
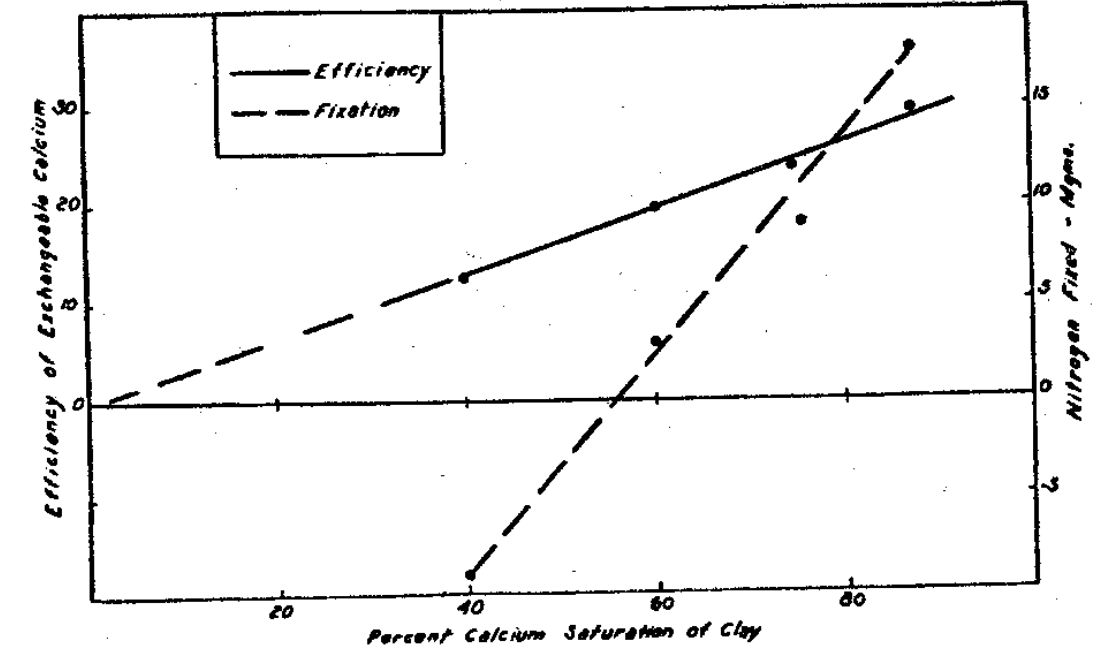
Plant growth followed the degree of clay saturation by the calcium,8 whether it was accompanied by hydrogen or by the other inorganic cations (Fig. 9). In all of the trials in this test, as well as in the others, growth was insignificant unless the seed content of calcium was doubled within the growth period, which was five weeks. Increased degrees of saturation delivered an increased percentage of the exchangeable calcium into the crop. These percentage figures representing the efficiency of the exchangeable calcium varied from 6 to 25 of the constant, exchangeable supply (Fig. 10). When a large complex organic ion, like methylene blue, accompanied the calcium, the variable degree of saturation was without effect and the growth followed the constant amount of exchangeable calcium (Fig. 11).
Fig. 9–Growth of soybeans as related to the degree of calcium saturation of the colloidal clay. (Left to right, 40, 60, 75, 87, 95%. Top to bottom, hydrogen, magnesium, and barium as the ions reciprocal to calcium.)
This situation is not easily explained on chemical bases, though it certainly excludes any effect by soil acidity. Seemingly as more calcium is placed on the clay molecule, those ions added at the more nearly complete saturation stage are more active in entrance into the plant, or they may be less forcibly held to the clay molecule. Much less of the same total calcium moves into the plant with the low degree of saturation on many clay molecules than with the higher degree of saturation on less clay molecules.3 Seemingly chemical equilibrium pressure is changed, particularly increased when less clay surface for root contact is the case in the higher saturation degree.
Such effects suggest that we should place the calcium into limited soil areas for more complete clay saturation rather than placing it throughout the root zone for only partial saturation, if the calcium is to have the most pronounced effects, and the application used most efficiently. In agricultural practice this would suggest drilling the limestone in the manner used for fertilizers.
Fig. 10–Efficiency of exchangeable calcium as related to the degree of calcium saturation and the nitrogen fixation by soybeans.
Fig. 11—Increasing saturation degree (left to right) by calcium is without effect on growth (lower row) when calcium is accompanied by the organic ion methylene blue in contrast to effects when accompanied by hydrogen with its varying acidity (upper row) or potassium with neutrality (center row).
Movements of Nutrients From Plant to Soil Under Calcium Deficiency
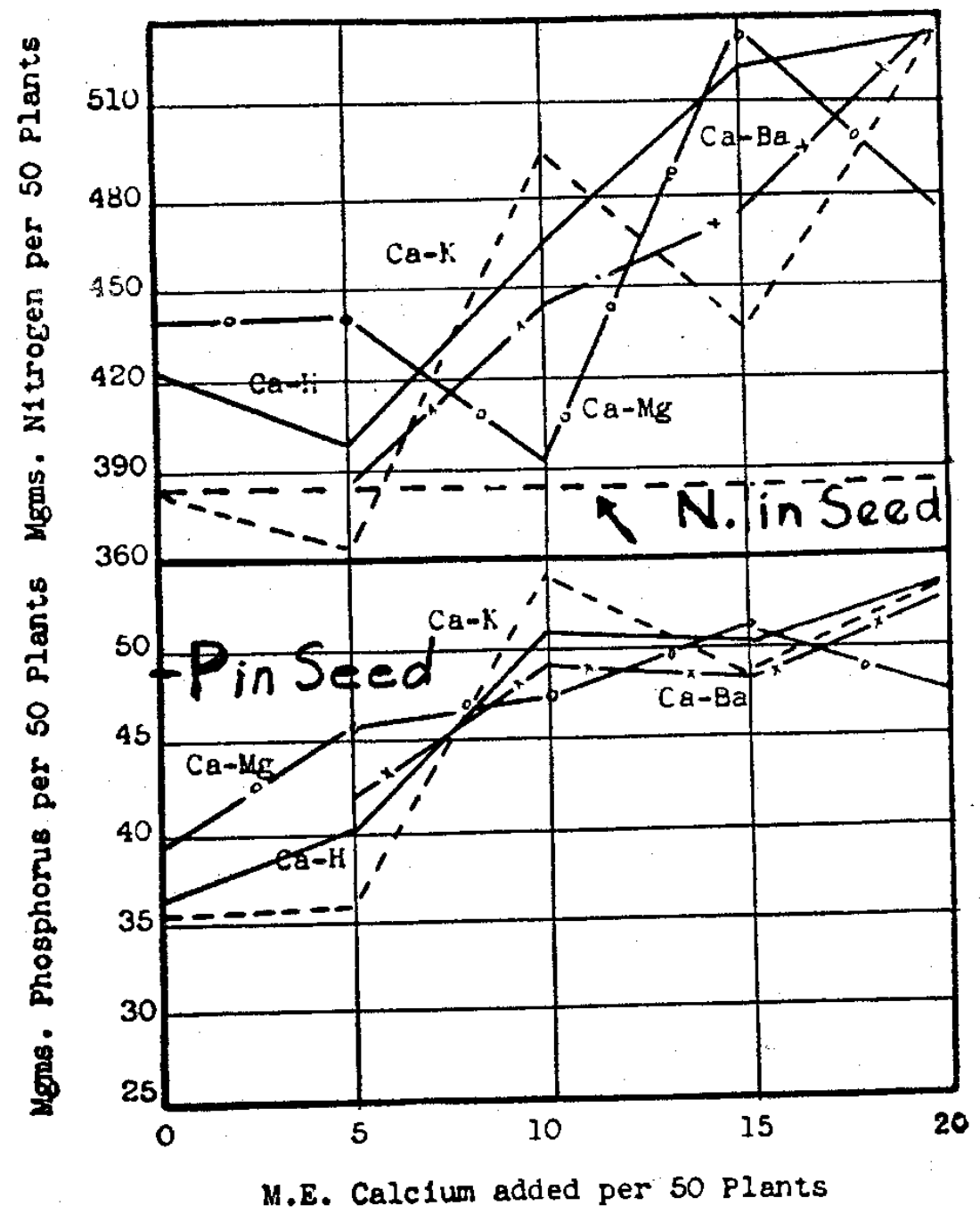
More complete chemical inventory of seed and clay at the outset was undertaken in order to determine the behavior of other nutrient ions beside calcium and nitrogen. The latter had been found seemingly moving from plant to soil, the former had always been going from soil to the plant when growth occurred. Trials were undertaken to determine the behavior of phosphorus in connection with different calcium levels9,6 and different amounts offered the plants. The element phosphorus, though not a cation, suggested its classification with nitrogen in this movement from plant to the soil. Unless larger offerings of calcium were given the plant, it failed to contain all the phosphorus originally in the seed (Fig. 12).
Fig. 12–Phosphorus and nitrogen contents of the soybean crop in relation to calcium levels. (Seed nitrogen=385 mgms, seed phosphorus = 47 mgms.)
Phosphorus and nitrogen, both constituents of protein, apparently are moved into the plant from its seed, rather than being lost from the seed back to the soil, only at high levels of delivery of calcium by the soil to the plant. Whatever the nutritional role of the calcium in the plants may be, it certainly raises the question whether it is not instrumental in metabolizing the nitrogen and phosphorus within the plant into insoluble protein to keep equilibrium displaced to the right, or whether it may not play some role in determining the nature and activity of the plant membrane interposed. The latter may seem such a function, according to the work of the late Professor True of Pennsylvania,12 though the former can scarcely be denied when calcium, nitrogen and phosphorus run so closely parallel in the plants growing near the lowest possible levels in these different trials. If calcium plays this role in membrane function, its significance in the early life of a plant is greater than that of merely adding calcium to the content of the seeds.
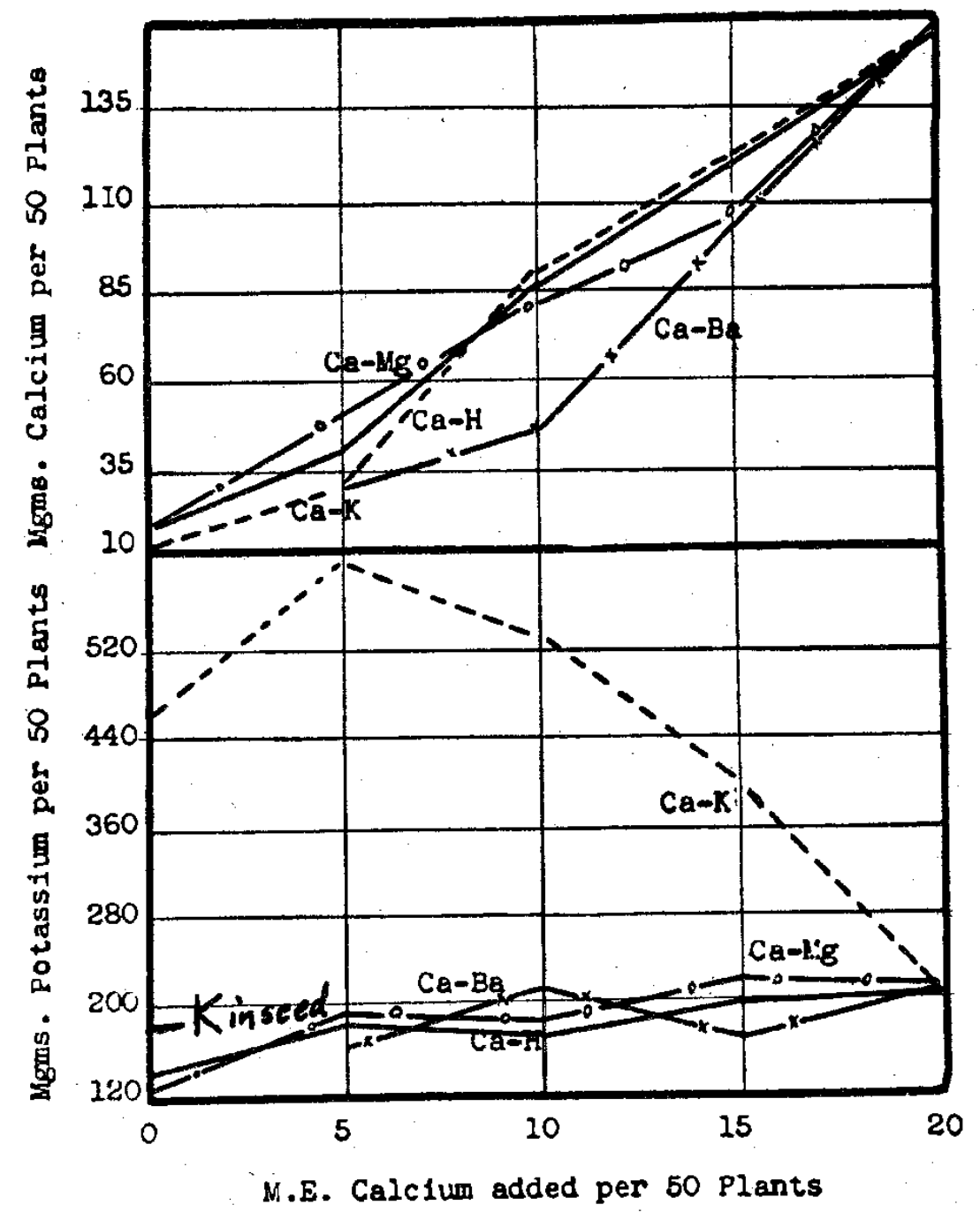
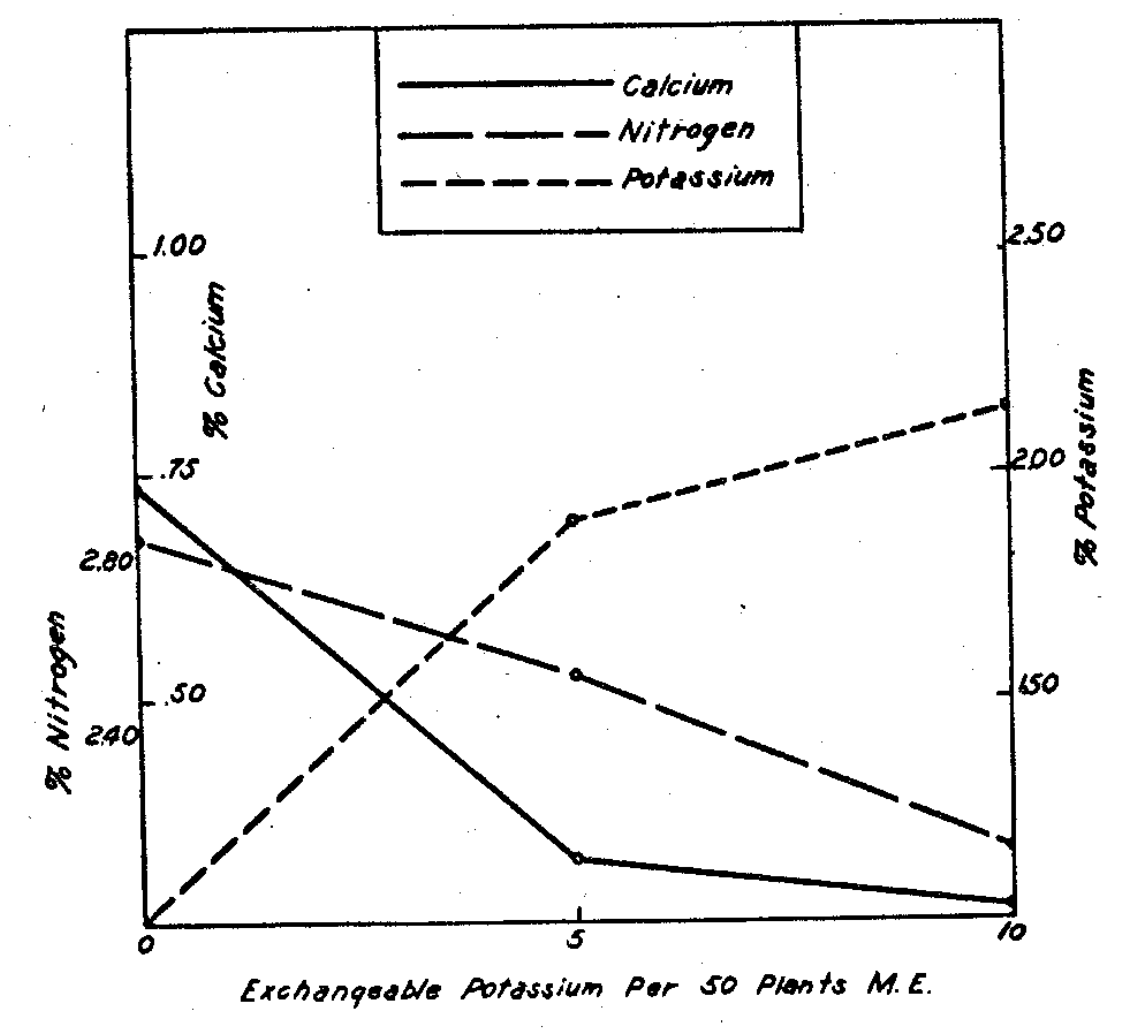
Fig. 13–Movement of seed potassium (lower graph) into the crop with increased calcium taken by the plants (upper graph). (Seed potassium= 171 mgms.)
In other tests9 it was revealed that potassium moves from the plant back to the soil (Fig. 13). Since this element appears in the seed in quantities larger than those of calcium or of phosphorus by roughly 15 and 3 times by weight, respectively, it is interesting to note that the magnitude of potassium movement to the left in our type equation has been as high as 50% of the seed content in some later experiments. It might be easy to imagine a “sour” soil serving as an acid extracting agent for taking potassium out of the plant, but the potassium was moving back to the soils when they were neutral, moderately saturated with calcium and containing no potassium. One crop which exhausted only part of the applied potassium brought potassium return to the soil when the second crop followed. This brings our viewpoint nearer to the equilibrium concept again and the belief that potassium must occur liberally on the clay, with calcium accompanying it if the potassium content of the crop is to increase over that in the seed.
The replacement of calcium in no small measure by potassium3 at certain calcium levels (Fig. 14) serves to bring up the calcium-potassium ratio and the significance of these two nutrients in determining the type of vegetation that dominates on the soil.4
Fig. 14–Reciprocal relations of calcium and potassium concentrations associated with declining nitrogen concentrations.
The full significance of calcium in these cases where nitrogen, phosphorus, and potassium have gone from the plant back to the soil unless calcium was liberally supplied is not yet explainable. It may not be as significant as first indications show, but it is significant that in no case has growth been possible unless calcium moved into the plant in its early life, while losses from the plant to the soil have occurred for nitrogen, phosphorus, and potassium. The quantities of these in the seeds may have some significance when we note calcium present in soybeans in a very small amount. The calcium, magnesium, phosphorus, potassium, and nitrogen occur in the seeds in the approximate ratio of 1 : 1 : 2 : 7 : 42 as molecular equivalents, respectively. Those in larger quantities may be more readily lost from the plant to the soil and yet permit plant growth. The shortage of calcium in the seed may be related to the need for delivery of it by the soil in the early plant life for plant growth.
When magnesium is considered in relation to its influences on soybean growth and nitrogen fixation7 it comes into importance quickly, but seemingly not directly. Improved plant manifestations, including nitrogen fixation, were not related to the amounts of magnesium taken by the plant but rather to the increased effectiveness by larger amounts of calcium from a constant source which went into the crop as the exchangeable magnesium on the colloidal clay was increased. Magnesium is apparently instrumental in bringing about greater effectiveness in the calcium use by plants in a manner much as calcium is apparently needed to make nitrogen, and phosphate more effective in the plant functions.
Another interesting relation of the calcium is that to manganese. Recent studies of the so-called “minor” element in plant nutrition point out that as more calcium carbonate is mixed throughout the soil, there is a reduction in the amount of manganese taken by such crops as bluegrass, redtop, lespedeza, and sweetclover. But when these same amounts of limestone are put into only the surface part of the soil to “feed” larger amounts of calcium into the plants, then the plants take more manganese from the soil. Here calcium carbonate seems to be playing a detrimental role to manganese delivery to the crop by its neutralizing effect throughout the soil, and a beneficial role to the same when it provides the plant with calcium as a nutrient in only a limited zone of the soil. Here are two distinct effects by calcium if this visualization of its role is correct.
Studies so far have seemingly emphasized the role of calcium as an adsorbed ion. Such emphasis has been one of dominating facts and overwhelming evidence. The calcium factor stands out clearly in soil development when the degree of this process is measured mainly in terms of calcium accumulation and calcium depletion. Vegetation in its ecological array also fits into the calcium picture.4 The plant composition with its requisite calcium for nitrogenous vegetation and the calcium depletion and potassium dominance for carbonaceous vegetation add importance to calcium for our understanding of the possibilities on our different soils. Then, too, the close linkage of phosphorus to calcium in its behavior must not be omitted. Phosphorus shows different chemical behavior when adsorbed on a calcium saturated beidellite clay than when on such carrying no exchangeable calcium.9,11 Microbial behaviors in colloidal clay medium suggest phosphorus-to-calcium linkage as these are adsorbed.10 Plant behavior suggests plant use of them in such combination6 and certainly in animal physiology, at least of the vertebrate class, these two elements cannot be separated very widely. These are some of the aspects of calcium that have given it emphasis, not because of its importance per se but because of its seemingly significance in relation to the behavior of the other ions and the entire physico-chemical structure of plants and soils.
To date the behavior of all the plant nutrient ions adsorbed on the colloidal clay complex can by no means be catalogued completely. However, a beginning has been made which has called attention first to calcium. One nutrient after the other can be brought into the picture in relation to calcium and then the different nutrients in relation to each other. The colloidal clay concept and its behavior as if in equilibrium with the plant colloid on the other side of a cell wall as membrane has opened fields of study in plant physiology and soil fertility that are bringing the soil and plants closer together with every research effort using this tool. These studies may point to a fuller significance when we learn of their reflections from plant to animal and human nutritional behaviors. Perhaps in time, even the mystical matters of plant growth will reveal that in the last analyses they can be catalogued as results of combinations of the more commonly accepted laws of simple chemical behavior.
References Cited:
- Albrecht, Wm. A.: “Inoculation of legumes as related to soil acidity.” Jour. Amer. Soc. Agron., 25 : 512-522, 1933.
- —— “Physiology of root nodule bacteria in relation to fertility levels of the soil.” Soil Sci. Soc. Proc., 2 :315-327, 1937.
- —— “Some soil factors in nitrogen fixation by legumes.” Intern. Soc. Soil Sci. Third Comm. Trans., A, 1939.
- —— “Calcium-potassium-phosphorus relation as a possible factor in ecological array of plants.” Jour. Amer. Soc. Agron., 32 : — —, 1940.
- ——, and Jenny, H.: “Available soil calcium in relation to ‘damping off’ of soybeans.” Bot. Gaz., 92 : 263-278, 1931.
- ——, and Klemme, A. W.: “Limestone mobilizes phosphate into Korean lespedeza.” Jour. Amer. Soc. Agron., 31 : 284-286, 1939.
- Graham, Ellis R.: “Magnesium as a factor in nitrogen fixation by soybeans.” Mo. Agr. Exp. Sta. Res. Bul., 288, 1938.
- Horner, Glenn M.: “Relation of the degree of base saturation of a colloidal clay by calcium to the growth, nodulation and composition of soybeans.” Mo. Agr. Exp. Sta. Res. Bul., 232, 1935.
- Hutchings, Theron B.: “Relation of phosphorus to growth, nodulation and composition of soybeans.” Mo. Agr. Exp. Sta. Res. Bul., 243, 1936.
- McCalla, Thomas M.: “Behavior of legume bacteria in relation to the exchangeable calcium and hydrogen ion concentration of the colloidal clay fraction of the soil.” Mo. Agr. Exp. Sta. Res. Bul. 256, 1937.
- Ravikovitch, S.: “Anion exchange. I. Adsorption of the phosphoric acid by soil.” Soil Sci., 38 : 219-239, 1934. “II. Liberation of the phosphoric acid adsorbed ions by soils.” Soil Sci., 38 : 279-290, 1934.
- True, R. H.: “The significance of calcium for higher plants.” Science, 55 : 1-6, 1922.