Access to all articles, new health classes, discounts in our store, and more!
Vegetable Crops in Relation to Soil Fertility: III. Oxalate Content and Nitrogen Fertilization
Published in Food Research, Vol. 11, No. 1, pp. 54-60, 1946
* * *
Green leafy vegetables may be one of our richest sources of dietary calcium. Although these crops generally make adequate growth over a wide range of soil differences, the fertility level may presage a wide range in their mineral composition. Of late, the relative availability or digestibility of organo-calcium and other mineral-organic complexes in many of these vegetables has been the subject of numerous investigations, such as those of Tisdall and Drake (1938), Spiers (1939), and Kohman (1939). A summary of this research, which has involved feeding tests conducted on both man and smaller animals, shows rather conclusively that many common leafy vegetables, including spinach, Swiss chard, beet greens, and New Zealand spinach, may be worthless as contributors of calcium and magnesium and in addition, if consumed in moderate quantities, may render unavailable a large percentage of the calcium normally furnished in the diet by other foods. This undesirable feature is attributed to the large amounts of oxalic acid which probably combine not only with calcium in the plant but also with the magnesium to form insoluble oxalates, Pierce and Appleman (1943), making these two essential elements from the soil valueless so far as human nutrition is concerned.
Since these oxalate-containing foods now play a prominent, though perhaps not a desirable, role in the human diet, an analysis of plants for the chemically equivalent relationships between their oxalate contents on the one hand, and their concentrations of calcium and magnesium on the other, was deemed essential. Moreover, the wide fluctuations in oxalic acid known to occur in a given crop of the same variety, as indicated by Kohman (1939), prompted a further inquiry into possible soil factors, causal of this variation, other than those of calcium and pH already outlined by Schroeder and Albrecht (1942). The following investigation represents an analysis and study of the oxalate and mineral contents of Swiss chard (variety Lucullus), which was subjected to 16 levels of soil nitrogen and calcium as combinations of four different amounts of each.
Experimental Procedure
Satisfactory control in providing the desired fertility gradients was achieved by using the subsoil of Putnam silt loam as a carrier for exchangeable soil nutrients. This material, with its high content of colloidal acid clay, has the semblance of natural soil and permits the uptake of nutrients by the plant through contact exchange.
A series of soil treatments was prepared by supplying calcium and nitrogen levels each of 5, 10, 20, and 40 milliequivalents (me.) with all possible (16) combinations of two nutrients. Accordingly, for each level of calcium there were four levels of nitrogen, and for each level of nitrogen there were four of calcium. The calcium and nitrogen were provided by adding variable quantities of calcium acetate and ammonium nitrate, respectively, to the correspondingly required clay aliquots, replacing the adsorbed hydrogen thereon with cationic nutrients. Other materials supplied in constant amounts consisted of 20 me. each of potassium and phosphorus, and six me. each of magnesium and sulfate. The resulting clay samples, almost completely saturated with various ratios of exchangeable calcium and nitrogen, were, by using moisture, blended with pure white quartz sand. Each treatment was replicated 10 times, the plants being grown in one-gallon glazed crocks in which one plant for each container was retained till harvest time. The nutrients and the clay supplied for each plant with the resulting pH are presented (Table 1).
Table 1–Nutrients Added to Clay to Provide Variable Calcium and Nitrogen Levels
| Treatment | Milliequivalents per plant | Clay per plant, gm. | Resulting pH | |||||
| Ca | N | P | K | Mg | S | |||
| Ca Group I1 | ||||||||
| 1
2 3 4 |
40
40 40 40 |
40
20 10 5 |
20
20 20 20 |
20
20 20 20 |
6
6 6 6 |
6
6 6 6 |
716.6
633.3 591.6 570.8 |
6.5
6.8 6.8 7.0 |
| Ca Group II | ||||||||
| 5
6 7 8 |
20
20 20 20 |
40
20 10 5 |
20
20 20 20 |
20
20 20 20 |
6
6 6 6 |
6
6 6 6 |
550.0
466.6 425.0 404.1 |
6.4
6.6 6.6 6.8 |
| Ca Group III | ||||||||
| 9
10 11 12 |
10
10 10 10 |
40
20 10 5 |
20
20 20 20 |
20
20 20 20 |
6
6 6 6 |
6
6 6 6 |
466.6
383.3 341.6 320.8 |
6.4
6.5 6.6 6.6 |
| Ca Group IV | ||||||||
| 13
14 15 16 |
5
5 5 5 |
40
20 10 5 |
20
20 20 20 |
20
20 20 20 |
6
6 6 6 |
6
6 6 6 |
425.0
341.6 300.0 279.1 |
6.6
6.4 6.5 6.5 |
1 In a like manner four N groups may be arranged, each containing four levels of Ca.
The experiment was conducted under greenhouse conditions during the late fall and early winter 1943-44. All plants were harvested after a growing period of 90 days; at which time they were judged to be in the most desirable eating condition. The tops were cut, weighed, dried at 65°C. (149°F.), and the total dry matter recorded. After being shredded in a Wiley mill and finely ground in a Merker mill, the dried material was suitable for analyses. The chemical determinations consisted of oxalate, total nitrogen, calcium, magnesium, and phosphorus. All analyses were performed in accordance with regular laboratory methods recognized as official or standard procedures.
Results and Discussion
The total yields as fresh weights and the results of the chemical analyses in terms of per cent concentrations in the dried material are assembled (Table 2). The treatments are first arranged into calcium groups, of which the four different nitrogen levels are averaged to show the influence of each level of calcium. Secondly, the treatments are regrouped according to the nitrogen applied, and the mean values of the calcium treatments taken so that the average for each nitrogen level could be recorded.
The most striking features evident from the data presented seem to be the following: (1) the magnitude of yield increase was about equal when either nitrogen or calcium was applied in additive quantities; (2) regardless of the calcium supply, the percentage of oxalate in the crop generally increased as more nitrogen was provided; (3) the percentage of total nitrogen in the crop was directly correlated with that of the oxalate and with the nitrogen supplied in the substrate; (4) the percentages of plant calcium were consistently greater as the calcium in the soil was increased, while the magnesium at first varied directly with the increments of both calcium and nitrogen and then showed a decided drop when the highest level of either was reached; and (5) there were marked differences, owing to soil treatment, in the magnesium and phosphorus percentages although the quantities of these nutrients applied to the soil were constant for all groups.
These results are of special significance in that they demonstrate remarkable variability in the important dietary components within the same crop, as a result of growing it on soil media differing in fertility. Differences in fertility not only gave variances in the quantities of the mineral elements moved from the clay-sand mixture into these plants, but it is likely that much of the fluctuation in the concentrations of the oxalate–a synthetic product of the plant’s metabolism–may be accounted for by variability in the fertility of the soil.
Table 2–Yield, Oxalate Content, and Mineral Composition of Swiss Chard When Grown Under Variable Levels of Nitrogen and Calcium.
| Treatment | Variable me. applied | Total yields in fresh weight per 10 plants | Concentrations in per cent of dry weight | |||||
| N | Ca | Oxalate | Total nitrogen | Calcium | Magnesium | Phosphorus | ||
| Ca Group I | ||||||||
| 1
2 3 4 Average |
40
20 10 5 …. |
40
40 40 40 …. |
634.6
486.4 233.3 179.8 383.5 |
6.63
5.58 4.70 3.93 5.21 |
4.29
3.16 2.58 2.40 3.10 |
1.032
0.888 0.886 0.748 0.889 |
.477
.416 .534 .320 .437 |
1.33
1.30 1.87 2.67 1.79 |
| Ca Group II | ||||||||
| 5
6 7 8 Average |
40
20 10 5 …. |
20
20 20 20 …. |
487.3
693.5 243.4 133.8 389.5 |
6.47
5.57 4.79 4.32 5.29 |
4.61
3.92 2.63 2.06 3.31 |
0.784
0.676 0.720 0.776 0.739 |
.734
.864 .678 .542 .705 |
1.04
1.10 1.05 1.66 1.21 |
| Ca Group III | ||||||||
| 9
10 11 12 Average |
40
20 10 5 …. |
10
10 10 10 …. |
391.6
333.8 215.2 116.6 264.3 |
5.63
6.94 4.38 3.78 5.18 |
4.57
3.97 2.53 2.43 3.38 |
0.600
0.776 0.824 0.746 0.737 |
.440
.720 .552 .368 .520 |
1.06
0.88 1.41 2.04 1.35 |
| Ca Group IV | ||||||||
| 13
14 15 16 Average |
40
20 10 5 …. |
5
5 5 5 ….. |
32.0
212.7 171.5 104.6 130.2 |
6.50
7.34 5.00 3.96 5.70 |
4.15
3.91 2.77 2.25 3.27 |
0.488
0.820 0.660 0.552 0.630 |
.380
.646 .284 .390 .425 |
1.81
0.85 1.11 1.57 1.34 |
| N Group I | ||||||||
| 1
5 9 13 Average |
40
40 40 40 .… |
40
20 10 5 …. |
634.6
487.3 391.6 32.0 386.4 |
6.63
6.47 5.63 6.50 6.31 |
4.29
4.61 4.57 4.15 4.41 |
1.032
0.784 0.600 0.488 0.726 |
.477
.734 .440 .380 .508 |
1.33
1.04 1.06 1.81 1.31 |
| N Group II | ||||||||
| 2
6 10 14 Average |
20
20 20 20 …. |
40
20 10 5 …. |
486.4
693.5 333.8 212.7 431.6 |
5.58
5.57 6.94 7.34 6.36 |
3.16
3.92 3.97 3.91 3.74 |
0.888
0.676 0.776 0.820 0.790 |
.416
.864 .720 .646 .662 |
1.30
1.10 0.88 0.85 1.03 |
| N Group III | ||||||||
| 3
7 11 15 Average |
10
10 10 10 …. |
40
20 10 5 …. |
233.3
243.4 215.2 171.5 215.9 |
4.70
4.79 4.38 5.00 4.72 |
2.58
2.63 2.53 2.77 2.63 |
0.886
0.720 0.824 0.660 0.773 |
.534
.678 .552 .284 .512 |
1.87
1.05 1.41 1.11 1.36 |
| N Group IV | ||||||||
| 4
8 12 16 Average |
5
5 5 5 …. |
40
20 10 5 …. |
179.8
133.8 116.6 104.6 133.7 |
3.93
4.32 3.78 3.96 4.00 |
2.40
2.06 2.43 2.25 2.29 |
0.748
0.776 0.746 0.552 0.706 |
.320
.542 .368 .390 .405 |
2.67
1.66 2.04 1.57 1.99 |
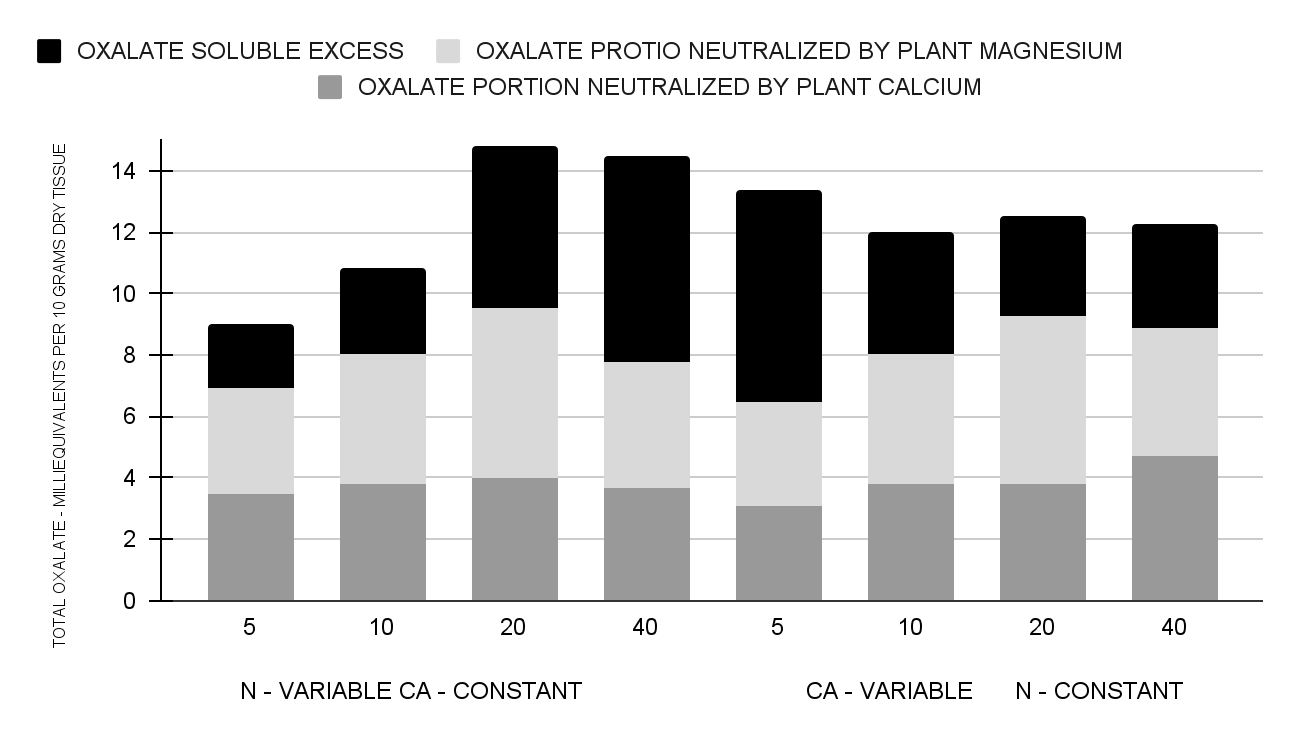
Oxalate in chemical combination with calcium forms a very insoluble compound. This is likely true also of its union with magnesium. Since some of the leafy vegetable crops have been reported as containing more than sufficient oxalate to precipitate and put the entire supply of these two mineral nutrients into inert forms, so far as ordinary digestive processes are concerned, it seemed essential in these studies to examine to what degree the oxalate was stoichiometrically balanced by the calcium and magnesium in Swiss chard. These relations are best shown by Fig. 1, wherein the probable fate of the total plant oxalate is portrayed.
Fig. 1. Probable disposition of oxalate in Swiss chard when grown under variable levels of nitrogen and calcium in the soil.
Several facts are evident. Stoichiometrically, the oxalate content exceeded by several times that of calcium at all fertility levels. In every instance the total oxalate was in excess of the plant calcium and magnesium combined. This left a sizeable surplus of soluble oxalates for possible combination with calcium derived from other foods in the diet. It is also to be noted that the quantity of oxalate in excess of the plant calcium alone, or of the summation of the calcium and magnesium, was directly correlated with the nitrogen offered in the soil. This excess was particularly marked when the calcium supply was low. The excess soluble oxalates diminished as the nitrogen supply decreased. This was true regardless of the calcium level.
Though the above relations between plant oxalates and the degree of nitrogen fertilization present a new departure in plant nutrition with respect to the nutritive quality of some leafy vegetables, the basic concept that nitrogen fertilizers may profoundly influence the organic acid composition of other crops was first emphasized by Ruhland and Wetzel (1926). This effect is accentuated if nitrate rather than ammonium nitrogen is used, according to Clark (1936); Wadleigh and Shive (1939); Vickery, Pucher, Wakeman, and Leavenworth (1940); and Blackman and Templeman (1940). Pepkowitz, Gilbert, and Shive (1944) suggest a direct association of oxalic acid formation with nitrate reduction. In the present investigation, ammonium nitrate was used as the nitrogen fertilizer. If nitrate-nitrogen alone had been employed, the results might have proven even more conspicuous.
These data, interpreted in conjunction with those of Schroeder and Albrecht (1942), suggest that soil-fertility practices in the production of Swiss chard and spinach may offer a certain degree of control over the quantities of oxalate produced. One would expect the least disturbance from oxalate when the crops are grown on an acid soil high in exchangeable calcium and low in readily available nitrogen. Wittwer, Schroeder, and Albrecht (1945) have demonstrated that heavy applications of nitrogen as a fertilizer salt seem to lessen their mineral as well as vitamin C contributions in the human diet. With reference to food qualities, it is unfortunate in the production of these vegetables that the yield response is so favorable toward increased nitrogen fertilization, as shown by Schroeder and Wittwer (1944).
Where dietary preferences permit, spinach, Swiss chard, beet greens, and New Zealand spinach may be replaced by other green leafy vegetables known to be free from the harmful effects of oxalate. Such crops include kale, mustard greens, collards, and turnip greens. The initial supply of all basic minerals and vitamins in this latter group of vegetables generally exceeds that of the high oxalate plants, and, containing an insignificant amount of oxalate, they contribute calcium and magnesium in forms that are readily assimilated by the digestive system.
Summary
By a study of the oxalate-calcium and oxalate-magnesium relations in Swiss chard, grown under 16 levels of variable calcium and nitrogen in colloidal clay cultures, it was shown that the concentrations of oxalate in the crops increased as more nitrogen was provided in the substrate. This increase seemed to be independent of the calcium supply.
When the chemically equivalent quantities of oxalate with respect to its possible union with the plant’s calcium and magnesium were considered, it was demonstrated that for all fertility levels the total oxalate production was far in excess of its equivalent of the plant’s calcium, or even of calcium and magnesium combined. These surpluses of oxalate, beyond precipitation by calcium and magnesium, were progressively of greater magnitude as the nitrogen supply was increased and the calcium decreased.
References Cited:
- Blackman, G. E., and Templeman, W. G.: “The interaction of light intensity and nitrogen supply in the growth and metabolism of grasses and clover (Trifolium repens). IV. The relation of light intensity and nitrogen supply to protein metabolism of the leaves of grasses.” Ann. Botany, 4, 533-587. 1940.
- Clark, H. E.: “Effect of ammonium and of nitrate nitrogen on the composition of the tomato plant.” Plant Physiol., 11, 5-24. 1936.
- Kohman, E. F.: “Oxalic acid in foods and its behavior and fate in the diet.” J. Nutrition, 18, 233-246. 1939.
- Pepkowitz, L. P., Gilsert, S. G., and Shive, J. W.: “The importance of oxygen in the nutrient substrate for plants-organic acid.” Soil Sci., 58, 295-303. 1944.
- Pepkowitz, E. C., and Appleman, C. O.: “Role of ether soluble organic acids in the cation-anion balance in plants.” Plant Physiol., 18, 224-238. 1943.
- Ruhland, W., and Wetzel, K.: “Zur Physiologie der organischen Säuren in grünen Pflanzen. 1. Wechselbeziehung im Stickstoff–und Säurestoffwechsel von Begonia semperflorens.” Planta, 1, 558-564. 1926.
- Schroeder, R. A., and Albrecht, Wm. A.: “Plant nutrition and the hydrogen ion. III. Soil calcium and the oxalate content of spinach.” Bull. Torrey Botan. Club, 69, 561-568. 1942.
- —- and Wittwer, S. H.: “Vegetable crops in relation to soil fertility. I. Yields of lettuce and spinach as influenced by variable calcium and nitrogen.” Proc. Am. Soc. Hort. Sci., 44, 469-472. 1944.
- Spiers, Mary: “The utilization of the calcium in various greens.” J. Nutrition, 17, 557-564. 1939.
- Tisdall, F. F., and Drake, T. G. H.: “The utilization of calcium.” J. Nutrition, 16, 613-620. 1938.
- Vickery, H. B., Pucher, G. W., Wakeman, A. J., and Leavenworth, C. S.: “Chemical investigations of the tobacco plant. VIII. The effect upon the composition of the tobacco plant of the form in which nitrogen is supplied.” Connecticut Agr. Expt. Sta. Bull., 442. 1940.
- Wadleigh, C. H., and Shive, J. W.: “Organic acid content of corn plants as influenced by pH of substrate and form of nitrogen supplied.” Am. J. Botany, 26, 244-248. 1939.
- Wittwer, S. H., Schroeder, R. A., and Albrecht, Wm. A.: “Vegetable crops in relation to soil fertility. II. Vitamin C and nitrogen fertilizers.” Soil Sci., 59, 329-336. 1945.