Access to all articles, new health classes, discounts in our store, and more!
The Psychotherapeutic Implications of Cytotoxic Testing
Published in the Journal of Orthomolecular Psychiatry, Vol. 14, No. 2, 128-135, 1985.
* * *
Abstract
This is the first attempt to measure the “emotional state” of patients undergoing dietary change following cytotoxic testing by means of a respected measuring instrument for feelings and mood, the M-R section of the Cornell Medical Index Health Questionnaire. The average patient demonstrated poor emotional health prior to testing. After approximately 60 days of dieto-therapy, nearly 77 percent of symptomatic patients showed improvement. with a symptomatology over-all reduced approximately 44 percent. The improvement ranged from a low of 37 percent (inadequacy) to a high of 63 percent (depression). It is hoped these observations will catalyze interest by others in pursuing the potential of cytotoxic testing and subsequent therapy.
Introduction
One of the most rapidly developing, and controversial, fields in medicine is the diagnosis and treatment of allergies. Food allergies are particularly troublesome because they do not seem to altogether fit the models and classifications established for inhalant and contact allergies. Some would dispute that most adverse reactions to food are “allergies”, in the atopic, Class I sense at all, and hence the term food sensitivities will be used here to encompass a larger, less wei! understood phenomenon.
It is now recognized that food sensitivities can affect virtually every system of the body, including the eyes, ears, gastrointestinal, cardiovascular, respiratory, urinary/genital, integumentary and musculoskeletal systems. The nervous system is no exception. Davidson1 enumerated the nervous and mental disorders that have been associated with food sensitivities: emotional immaturity, antisocial behavior, depression, anxiety, organic brain syndrome, temporary deafness, nerve palsies, neuralgias, urinary incontinence, hemiplegia, convulsions, syncope, vertigo, ataxia, tinnitus, myoclonus. numbness, impotence, diplopia, blurred vision, micropsia and macropsia. Others have described similar symptoms secondary to food sensitivities.2-9
Much has been discussed about the limitations of the various diagnostic methods currently used to identify foods which contribute to sensitivity reactions. Physicians and patients alike await a procedure that is both practical and effective. One possibility is the cytotoxic test, an in vitro blood test that can analyze hundreds of foods or other materials with no more time, pain or inconvenience to patient or physician than that required to draw a single blood sample. At one time accepted as “reasonably firmly established, both as to scientific fact and important clinical significance”,10 the validity of the test is now subject to dispute.
The purpose of this study is to attempt to measure changes in the status of various physical and psychologic systems of a patient who has completed a period of dietary improvement based upon the results of the cytotoxic test for food sensitivities. This is believed to be the first such attempt using an accepted and respected measuring instrument, the Cornell Medical Index Health Questionnaire (CMI). This paper will report our findings relative to the psychologic parameters of the CMI.
Review of the Literature
The cytotoxic test was introduced in 1956 by Arthur P. Black, M.D.11 Dr. Black observed that when allergens were added to the plasma of a patient with suspected allergies, polymorphonuclear leukocytes showed progressive cytotoxic effects and advanced destruction. Control samples did not show these reactions. By eliminating foods which demonstrated these cytotoxic reactions from patients’ diets he was able to provide patients with relief from their allergy symptoms. A subsequent study was unable to confirm these results, finding 70% of the tests impossible to interpret and poor correlation between test results and patient histories, though few of the allergens tested were foods.12 An animal study confirmed, however, that significantly more cytologic changes in blood, such as leukocyte and platelet aggregation, leukocyte vacuolization, and distorted and fragmented cells, occur when an antigen is added to blood that has been sensitized to that antigen than in normal, non-sensitive blood.13
In the 1960’s and 70’s the test procedure was refined significantly by Dr. and Mrs. William T. K. Bryan.14-18 Improved methods of preparing the sample and the antigens were developed and more precise criteria specified for analyzing the morphologic changes in leukocytes, platelets and RBCs. The Bryans also determined that the test was clinically useful.
The test continues to be criticized,19-23 however, on the basis of 4 studies.24-27 Lieberman et al.24 in a test of 45 patients, found little reproducibility of results when samples for the same patient were read more than once. The level of experience of the technicians conducting the test is not stated. Lehman27 noted variation in results when tests on 10 patients were repeated up to 5 times over a two month period, though no statistical analysis was done to determine the significance of this variability. However, Lehman also examined reproducibility of results on identical samples. Split samples were blind-tested for 11 patients. Exact duplicate results were seen in 79.9% of the tests and a total of 98% varied, at most, by one degree of reactivity, evidencing extremely high reproducibility.
Other researchers have also found significant reproducibility of the cytotoxic test: Ulett & Perry28,29 completed from three to eight sets of tests on each of 10 patients and stated results were very consistent from test-to-test, both for the individual food items and the degree of response. In Benson and Arkins’ critical report25 it was noted that each test was read separately six times and a significant degree of reproducibility was demonstrated. Conners30 performed double-blind tests at least twice on 95 subjects and achieved a correlation coefficient of .85, where .70 would be satisfactory. Holopainen et al.31 had samples from 49 subjects read double-blind by two technicians and found 78% of the results were identical and a total of 94% had at most a slight divergence in the degree of reactivity.
Another criticism of the test is that its results do not correspond to other clinical indications of allergy. King26 compared the results of cytotoxic tests on 300 patients to the results of intracutaneous provocative testing, a technique that has also been criticized,19,21,22 and found no correlation. These results are open to question, however, in that as conducted the cytotoxic tests produced 68% positive reactions, as opposed to positive results well below 30% noted by other researchers,9,17,27,32 and the objective intracutaneous tests showed positive reactions in 92% of all foods tested.
Lieberman et al.24 concluded the cytotoxic test had poor concordance with clinical indications on the assumption that foods not known to the patient to produce symptoms and non-reactive with skin tests were false-positives. Benson and Arkins25 criticized the test on the basis of “false-positives” for 14 subjects based on patient histories.
The difficulty with comparing cytotoxic test results to clinical indications is the absence of a benchmark. Unverified patient perceptions of symptom-provoking foods seem of limited value for this purpose. Test results can only be classified as correct or false if some independent, objective and correct measure is available, and none exists for food sensitivities. This was illustrated in a study by Ruokonen et al.32 Ninety patients were tested for allergies using skin tests, RAST, tests for nasal and blood eosinophilic cells, total lgE, tests for IgA, IgG and IgM, precipitating antibodies, the Mantoux test, allergy history and cytotoxic testing. None of these procedures correlated well with any of the others. Of all the tests, however, the cytotoxic test correlated best with the allergy history.
Though there is no simple or foolproof method to evaluate the efficacy of cytotoxic lesting, there is nonetheless some evidence of the clinical effects of cytotoxic testing and the appropriate follow-up. A number of studies have shown that foods identified as reactive by the cytotoxic test will cause symptoms upon consumption. Conners30 conducted double-blind tests of ten patients with small capsules of freeze-dried foods. Nine out of the ten showed a positive allergic response to foods which had tested positive in the cytotoxic test, but none showed response to foods tested negative. Fifty-five test patients and ten control patients were selected for oral challenges in a study by Trevino.33 Each subject received a cytotoxic test and one reactive food was chosen for Ihe test group and a non-reactive food was chosen for the control group. Fifty-three (96%) of the test group developed symptomatology, and none of the control group developed any symptomatology. Duhon34 correlated cytotoxic tests with provocative testing and found only 7 conflicts in 666 tests, or almost 99% agreement.
Ulett and Perry9,28,29 demonstrated that, in addition to manifesting allergy symptoms, subjects consuming foods to which they are cytotoxically reactive have a corresponding increase in white blood cell count, increase in pulse rate and desynchronization of EEG. Subjects consuming foods that tested negatively in the cytotoxic test experienced none of these changes.
Observers have also noted significant decreases in food sensitivity symptoms when foods determined to be reactive in cytotoxic tests are removed from patients’ diets. The Bryans, in a study of 80 patients using their procedure, found 85% showed improvement in symptoms and only 5% displayed no improvement.17 In a survey of 50 patients in an otolaryngologic practice, 47 (94%) received satisfactory relief from symptoms ranging from rhinitis to fatigue.35 In another practice, 118 patients responding to a questionnaire after cytotoxic testing and follow-up indicated relief of symptoms such as stuffy nose (63.5% of 50 patients), headache (76% of 40 patients), and earache (91% of 15 patients). Of all those responding, 39% reported relief from fatigue and 27% from mental depression.36 Duncan37 employed skin scratch tests from 1965-66 with 308 patients, 8% of whom achieved good results sustained over a year. From 1971-72, 377 patients were treated by skin titration and food provocation methods and 27.6% obtained useful relief. From 1973-75, 680 patients were investigated using cytotoxic testing. Of these, 60% reported very good relief and 26.3% described useful relief, for a total of 86.3%.
While the CMI has not previously been used to evaluate patient progress after cytotoxic testing, it has been widely used in other contexts. Its strengths and weaknesses as a measure of emotional state have been outlined by the inventors38-41 and others.42-47 Suffice it to say that, while it is obviously not perfect, it does provide a reasonable estimate of feelings and moods. It is used here as a more sophisticated measurement of changes in patient status than is offered by physician or patient expressions of improvement or its absence.
Method of Investigation
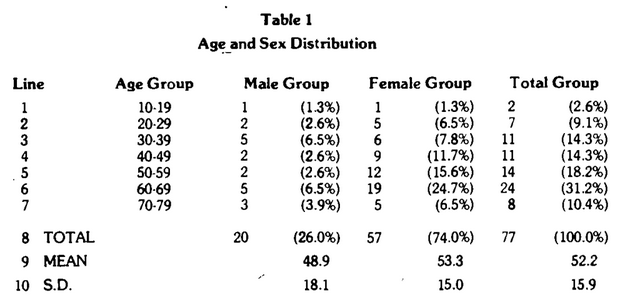
Subjects of this study were individuals receiving cytotoxic testing and follow-up nutritional counselling at a commercial cytotoxic testing laboratory. Age and sex distributions are presented in Table I. Prior to the blood draw each client was asked to complete the appropriate CMl questionnaire for their sex. After a 12-hour fast, blood was drawn and tested against 146 foods and food additives.
The protocol of the cytotoxic test as applied for this study was as follows:
- Citrated blood was centrifuged at 2500 RPM for ten minutes and the plasma added to 0.5 ml of .6% NaCI. The buffy coat was transferred to a Wintrobe Sedimentation tube, centrifuged at 2500 RPM for ten minutes and returned to the plasma.
- Using a precision-measurement multi-pipette (Flow Laboratories, Virginia) the plasma was pipetted into commercially prepared microtiter-type plates containing precision-measured amounts of filtered, diluted antigens (Physicians Cyto Laboratories, Florida). Test plates were incubated in a humidified container without coverglass.
- Samples were examined using video-enhanced microscopy. Results were not graded, but were simply classified as non-reactive or reactive, based upon 25% or more of leukocytes demonstrating vacuoles, cell destruction (ghosting) or lack of pseudo-padding, or, secondarily, platelet aggregation or disintegration or crenation or abnormal rouleaux formation in RBCs. A blind scoring system was used to eliminate technician bias.
Clients were given copies of the results of the test and counseled to avoid reactive foods for a period of eight weeks. A cooking class for clients provided assistance in food substitution and menu planning. Clients were requested to return after eight weeks for assistance in re-introducing reactive foods into their diets. Individual counseling was available for clients needing help in adhering to their nutritional plan. Clients who returned after eight weeks were requested to complete another CMI Questionnaire. Seventy-seven subjects completed the second questionnaire.
Utilizing the questionnaire according to the instructions set forth by the inventors,38 it is possible to derive two major groups of information concerning emotions and moods. First, the entirety of Sections M through R may be totaled as a measure of overall emotional “health”. Generally, according to the inventors, a significant emotional problem should be suspected when more than three or four questions are answered in the affirmative. The importance of this particular marker will become apparent later in this report. Second, each section deals with a separate emotional component: sensitivity, anxiety, inadequacy, anger and tension, and each of these subsets may be evaluated.
Because we were concerned with the change in these components, it was decided to exclude from our tabulations those questions not subject to change, i.e. those involving historical symptoms or family history. Questions 164, 167, 169, 170, 171 and 173 were excluded, leaving 12 questions for inadequacy, 6 questions for depression, 4 questions for anxiety, 5 questions for sensitivity, 9 questions for anger, 9 questions for tension and 45 for all sections (M-R).
Results
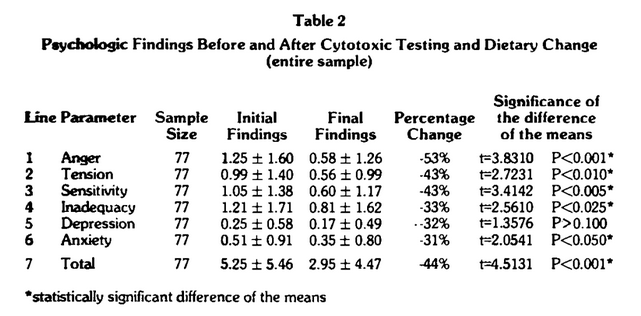
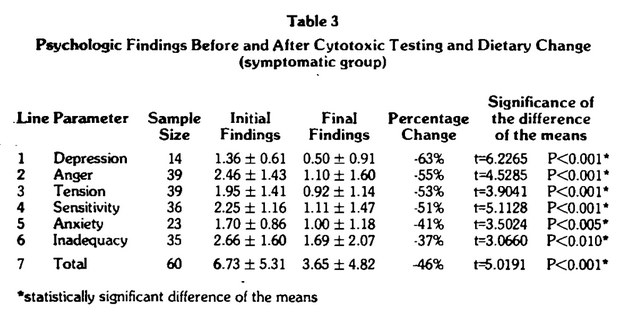
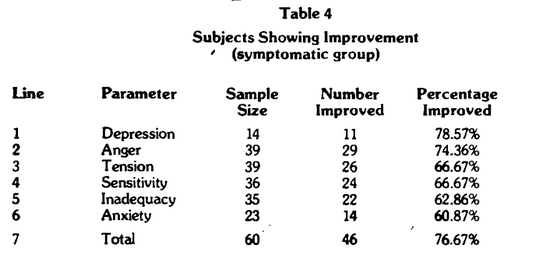
Table 2 is an overall summary of the affirmative responses for the M-R section of the CMI. Four points warrant special consideration. First, the average patient reported 5.25 emotional complaints. Mention was made earlier that three or four suggests “significant” disease. Hence, on a purely mean basis, this group must be viewed as being in very poor emotional “health.” Second, following cytotoxic testing and dietary change, in approximately 60 days, the mean number of findings decreased to 2.95. Under these conditions, therefore, there was a statistically significant improvement (t=4.5131, P<0.001) in feelings and mood of an order of 44 percent. Third, of the 77 subjects, 60 reported positive findings. Recalculations for this symptomatic group (Table 3) showed higher values before (6.73) and after (3.65) and a statistically significant (t=5.0191, P<0.001) improvement of 46 percent. Finally, 46 of the 60 symptomatic subjects (77%) showed improvement (Table 4).
It was previously mentioned that the M-R section of the questionnaire is so designed as to allow analysis of data subsets: anger, tension, sensitivity, depression, anxiety and inadequacy (Table 2). On a mean percentage basis, all emotional parameters improved, ranging from a low of 31 percent (line 6) for anxiety to a high of 53 percent (line 1) for anger. However, it should be noted that only five of the six proved to be statistically significant. For depression (line 5), the mean improvement of 32 per cent was not significant at the 5 percent confidence level. In this category there were a great number of asymptomatic subjects; out of the total sample of 77 subjects only 14 reported depression findings.
The data was recalculated including the symptomatic subjects only (Table 3). All mean percentage improvements are higher, ranging from a maximum of 63 percent for depression (line 1) to a minimum of 37 percent for inadequacy (line 6). The rank order in terms of mean percentage improvements changed as depression moved from a rank of 5 (Table 2, line 5) to the top rating (Table 3, line 1). It is especially important to note that, in the symptomatic group, all categories are statistically significant. The percentage of subjects experiencing improvement ranged from 61% for anxiety (Table 4, Iine 6) to 79% for depression (Table 4, line 1).
Discussion
The information derived from this preliminary study and reported here is clearly new. As far as we can ascertain, there is not one published report on the results stemming from cytotoxic testing and therapy as it relates to emotional state as judged by any of the many available psychometric techniques. The data presented here suggests that dietary management based on the cytotoxic test, as imperfect as it may be, can be very useful in reducing the common symptoms of emotional illness in the population at large.
Many questions remain to be answered concerning the cytotoxic test. Insights into the way it works and the extent of its usefulness will continue to be gained as we learn more of the mechanisms of food allergies and sensitivities. It may develop that some of the day-to-day variation in cytotoxic test results is due to more frequent and rapid change in the body’s reactions to foods than previously realized, as opposed to inconsistency of the test itself. It is also hoped and expected that the recent technological improvement in the procedure will continue, increasing its precision and accuracy. The increasing evidence of the impact of food sensitivities on health and the limitations of other testing methodologies warrants continued investigation of the potential of cytotoxic testing and subsequent therapy.
References Cited:
- Davidson, H. M.: “Allergy of the nervous system,” Quart. Rev. Allerg. Applied Immun. 6:157, 1952,
- Alvarez, W.: Nervousness, Indigestion and Pain. 1943.
- McGovern, J. and Knight, J.: Allergy and Human Emotions, 1967, Charles C. Thomas/Springfield, III.
- Speer, F.: Allergy of the Nervous System, 1970, Charles C. Thomas/Springfield, Ill.
- Wunderlich, R.: Allergy, Brains and Children Coping, 1973. Johnny Reeds, Inc./St. Petersburch, FI.
- Campbell, M. B.: “Neurological and psychiatric aspects of allergy.” Otolaryngol. Clin. North Am. 7(3):805-824, 1974.
- Speer, F.: “The Allergic Tension-Fatigue Syndrome.” Pediatr. Clin. North Am. 1:1029-1037, 1954.
- Philpott, W. H: “Allergy and ecology in orthomolecular psychiatry.” In Dickey, L. D. (ed.), Clinical Ecology, 1976, Charles C. Thomas/Springfield, Ill.
- Ulett, G. A.: “Food allergy–cytotoxic testing and the central nervous system.” Psychiatr. J. Univ. Ottawa 5(2):100-108, June, 1980.
- Scientific Board of the California Medical Association: “Important advances in clinical medicine.” Western Journal of Med. 120(1):62-64, Jan. 1974.
- Black, A. P.: “A new diagnostic method in allergic disease.” Pediatrics 17(5):716-724, May, 1956.
- Chambers, V. V., Hudson, B. H. and Glaser, J.: “A study of the reactions of human polymorphonuclear leukocytes to various allergens.” J. Allergy 29(2):93-102, March, 1958.
- Hartman, J. D. and Hoch, W. S.: “Changes in blood leucocytes resulting from an antigen-antibody reaction.” Am. J. Physiology 183:214-220, Oct./Dec., 1955.
- Bryan, W. T. K. and Bryan, M. P.: “The application of in vitro cytotoxic reactions to clinical diagnosis of food allergy.” Laryngoscope 70:810-824, June, 1960.
- Bryan, W. T. K. and Bryan, M. P.: “Cytotoxic reactions in the diagnosis of food allergy.” Laryngoscope 79(8):1453-1472, Aug. 1969.
- Bryan, W. T. K. and Bryan, M. P.: “Clinical examples of resolution of some idiopathic and other chronic disease by careful allergic management,” Laryngoscope 82:1231-1238, 1972.
- Bryan, W. T. K. and Bryan, M. P.: “Cytotoxic reactions in the diagnosis of food allergy.” Otolaryng. CIin. N. Amer. 4(3):523-533 Oct. 1971.
- Bryan, W. T. K. and Bryan, M. P.: “Cytotoxic reactions in the diagnosis of food allergy,” in Dickey, L. D. (ed), Clinical Ecology (1976), Charles C. Thomas/Springfield, III.
- Golbert, T. M.: “A review of controversial diagnostic and therapeutic techniques employed in allergy.” J. Allergy Clin. Immun. 56(3):170-190, Sept.,1975.
- American Academy of Allergy: “Position statements–controversial techniques.” J. Allergy Clin. Immun. 67(5):333-338, May, 1981.
- Grieco, M. H.: “Controversial practices in allergy.” J.A.M.A. 247(22):3106-3111, June 11, 1982.
- Van Metre, T. E.: “Critique of controversial and unproven procedures for diagnosis and therapy of allergic disorders.” Pediatric Clin. N. Am. 30(5): 807-817, Oct. 1983.
- Terr, A. I.: “Editorial–The Cytotoxic Test.” Western J. of Med. 139(5): 702-703, Nov. 1983.
- Lieberman, P., Crawford, L., Bjelland, J., Connell, B. and Rice, M.: “Controlled study of the cytotoxic food test,” J.A.M.A. 231(7): 728-730, Feb. 17, 1975.
- Benson, T. E. and Arkins, J. A.: “Cytotoxic testing for food allergy: evaluation of reproducibility and correlation,” J. Allergy Clin. Immunol. 58(4): 471-476, Oct., 1976.
- King, W. P.: “Testing for food allergy: a statistical comparison of cytotoxic and intracutaneous tests.” Laryngoscope 88(1): 1649-1659, 1978.
- Lehman, C. W.: “The leukocytic food allergy test: a study of its reliability and reproducibility. Effect of diet and sublingual food drops on this test.” Ann. Allergy 45(3): 150-158, Sept., 1980.
- Ulett, G. A. and Perry, S. G.: “Cytotoxic testing and leukocyte increase as an index to food sensitivity.” Ann. Allergy 33(1): 23-32, July, 1974.
- Ulett, G. A. and Perry, S. G.: “Cytotoxic testing end leukocyte increase as an index to food sensitivity. II. Coffee and Tobacco,” Ann. Allergy 34(3): 150-160, March, 1975.
- Conners, C. K.: Food Additives and Hyperactive Children, 1980, Plenum/N.Y.
- Holopainen, E., Palva, T. ,Stenberg, P., Backman, A., Lehti, H. and Ruokonen, J.: “Cytotoxic leukocyte reaction.” Acta Otolaryngol. 89:222-226, 1980.
- Ruokonen, J., Holopainen, E., Palva, T., and Backman, A.: “Secretory Otitis Media and allergy.” Allergy 36(1):59-68, 1981.
- Trevino, R.J.: “Immunologic mechanisms in the production of food sensitivities.” Laryngoscope 91(11):1913-1936, Nov. 1981.
- Duhon, S. C.: “Cytotoxic tests in food allergy.” Gulf Coast Allergy Study Group, Feb. 1968. Dallas, Texas, cited in Bryan, W. T. K. and Bryan, M. P., “Cytotoxic reactions in the diagnosis of food allergy,” Otolaryng. Clin. N. Amer. 4(3):523-533, Oct., 1971.
- Updegraff, T. R.: “Food allergy and cytotoxic tests.” Ear, Nose & Throat J. 56(11): 48-64, Nov., 1977.
- Boyles, J. H.: “The validity of using the cytotoxic food test in clinical allergy.” Ear, Nose & Throat J. 56(4):35, April, 1977.
- Duncan, R. B.: “Otolaryngeal allergy in Wellington.” N. Z. Med. J. 85:45-49, Jan. 26,1977.
- Brodman, K., Erdmann, A. J., and Wolff, H. G.: The Cornell Medical Index Health Questionnaire Manual. 1949, Cornell Univ. Medical College, Ithaca.
- Brodman, K., Erdmann, A. J., Lorge, I. and Wolff, H. G.: “The Cornell Medical Index: an adjunct to medical interview.” J.A.M.A. 140(6): 530-534, June, 1949.
- Brodman, K., Erdmann, A. J., Lorge, I., Gerhenson, C. P. and Wolff, H. G.: “The Cornell Medical Index Health Questionnaire. III. Evaluation of emotional disturbance.” J. Clin. Psychol. 8(2),119-124, April, 1952.
- Brodman, K., Erdmann, A. J., Lorge, I., Deutxchberger, J. and Wolff, H. G.: “The Cornell Medical Index Health Questionnaire. VII. The prediction of psychosomatic and psychiatric disabilities in Army training.” Am. J. Psychiat. 111(1):37-40, July, 1954.
- Knox, S. J.: “An evaluation of CMI responses in the identification of psychiatric illness associated with mitral surgery,” J. Psychosom. Res. 7(1): 35-39, July, 1963.
- Arthur, R. J., Gunderson, E. K. E., and Richardson, J. W.: “The Cornell Medical Index as a mental health survey instrument in the naval population.” Military Med. 131(7):605-610, July, 1967.
- Gibson, H. B., Hanson, R. and West, D. T.: “A questionnaire measure of neuroticism using a shortened scale derived from the Cornell Medical Index.” Br. J. Soc. Clin. Psychol. 6(2), 129-136, June, 1967.
- Verghese, A.: “Relationships between the Eysenck Personality Inventory N score, the Cornell Medical Index M-R Score and the psychogalvanic response.” Br. J. Psychiat. 116(530): 27-32, January, 1970.
- Seymour, G.E.: “The structure and predictive ability of the Cornell Medical Index for a normal sample.” J. Psychosom. Res. 20(5):469-478, 1976.
- Costa, P. T. and McCrae, R. R.: “Psychiatric symptom dimensions in the Cornell Medical Index among normal adult males.” J. Clin. Psychol. 33(4):941-946, October, 1977.