Access to all articles, new health classes, discounts in our store, and more!
The Effects of Intravenous EDTA Infusion on the Multichemical Profile
Published in American Clinical Laboratory, Vol. 7, No. 7, pp. 42-43, October 1988.
* * *
The intravenous infusion of EDTA (ethylene diamine tetraacetic acid) is a therapeutic option sometimes used for the treatment of toxicological conditions (e.g., lead intoxication)1,2 and other, somewhat controversial, treatments (e.g., occlusive vascular disease).
There are certain concerns about the potential health hazards of intravenous EDTA treatment.3 Some suggest that intravenous EDTA treatment invites a “nonspecific metabolic upheaval,” while others believe that the EDTA treatment may cause iatrogenic renal problems. Another concern deals with the potential demineralization of bone as a result of intravenous EDTA treatment.
Several of these concerns have been investigated previously. When used properly, EDTA treatment does not appear to compromise renal function. As a matter of fact, renal function appears to improve in some patients after the EDTA treatment.4-8 In addition, preliminary evidence suggests that demineralization is not evident in patients after EDTA treatment.9
This paper deals with the more general question of a nonspecific metabolic upheaval as the result of intravenous EDTA treatment as monitored by a multichemical profile of 23 tests.
Method
Twenty-eight people were randomly drawn from 127 volunteers who responded to an advertisement requesting the participation of people in the experiment and who fit the following criteria: 1) Are over the age of 40; 2) are ambulatory; 3) demonstrate a five-fold increase in the urinary lead as a result of a 3-g EDTA challenge; 4) have a fasting serum creatinine level of less than 1.5 mg/dL; and 5) have completed an extensive history and screening workup by a physician. All participants signed an informed consent form.
The therapeutic treatment consisted of a series of 20 EDTA intravenous infusions administered at approximately weekly intervals (where possible) over a period of 20 weeks. The maximum time span for a few subjects extended to 38 weeks. The EDTA infusion consisted of 3.0 g EDTA (Keylate®, Editate sodium, The Key Co.), 15.0 g ascorbic acid buffered in sodium bicarbonate (Bronson Pharmaceuticals), 800 mg magnesium chloride, 40.0 mg procaine, and 1000 units of heparin delivered in 500 mL of sterile, deionized water. The solution was intravenously infused over a period of 3-5 hours. Additionally, each subject was given three Insurance FormulaTM (Bronson Pharmaceuticals) vitamin and mineral supplement tablets per day.
The multi chemical profile was drawn while fasting, before treatment, after 10 infusion treatments, and after 20 infusion treatments. The serum was separated immediately and sent to a local private reference laboratory for processing.
Results
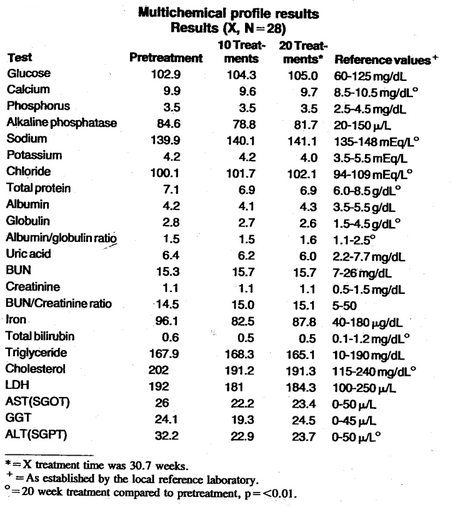
Table 1 lists the parameters and results of the multi chemical profile before treatment, after 10 treatments, and after 20 weekly treatments. Also included are the suggested reference ranges for each parameter as established by the reference laboratory performing the tests. All the parameters measured before treatment were within the reference values established for each procedure. Also, as shown, there were no clinically significant changes in any parameter after 10 or 20 treatments. Statistical treatment of the data revealed significant changes (p =<0.01) in calcium, sodium, chloride, total protein, globulin, albumin/globulin ratio, total bilirubin, and cholesterol in the 20 treatment data as compared to. the pretreatment data. However, all parameters remained within the normal accepted ranges; therefore, these changes were probably not of clinical importance.
Table 1–Multichemical profile results. Results (X, N = 28)
Discussion
There appears to be no evidence of a nonspecific metabolic upheaval after 20 EDTA intravenous infusions. Creatinine and blood urea nitrogen levels were not significantly changed. This is similar to previous work that showed EDTA treatment does not compromise renal function.6-8 Calcium and phosphorus levels did not significantly change over the period of 20 treatments. Although there were no radiographic studies of bones performed in this study, it would appear from the calcium and phosphorus results that there was no excessive mobilization of these compounds from the bone. However, the patients in this treatment group were on supplemental vitamin and mineral therapy. Total iron levels were not significantly changed. All liver function tests (ALT, AST, LDH, T. bilirubin, GGT, alkaline phosphatase) were normal. Cholesterol was reduced by 10.7 mg/dL after 20 treatments. McDonagh et al, showed similar results with cholesterol and HDL. 9-13
The results of the 23-multi chemical tests after 20 treatments of intravenous infusion with EDTA in this experiment would tend to show that there is no nonspecific metabolic upheaval. It would also appear that there is no renal compromise based on the BUN and creatinine results.
References Cited:
- Wyngaarden, J.B., Cecil’s Textbook of Medicine (W.B. Saunders Co., Philadelphia, PA, 1985) pp. 598-599, 2307-2309.
- Isselbacher, K.J. et al., Harrison’s Principles of Internal Medicine (McGraw-Hill, New York, NY, 1980), p. 967.
- Jones, R.J., “Chelation therapy,” JAMA 250 (5), 672 (1983).
- Cranton, E.M. and Frackelton, J.P., “Current status of EDTA chelation therapy in occlusive arterial disease,” J. Holistic Med. 4 (1), 24-33 (1982).
- Riordan, H.D. et al., “Another look at renal function and the EDTA treatment process,” Orthomol. Med. 2(3), 185-188 (1987).
- McDonagh, E.W., Rudolph, C.J., and Cheraskin, E., “The effect of EDTA chelation therapy plus multivitamin-trace mineral supplementation upon renal function: A study in serum creatinine,” J. Holistic Med. 4(2), 146-151 (1982).
- McDonagh, E.W., Rudolph, C.J., and Cheraskin, E., “The effect of EDTA chelation therapy plus supportive multivitamin-trace mineral supplementation upon renal function: A study in blood urea nitrogen,” J. Holistic Med. 5 (2), 163-171 (1983).
- Sehnert, K., Clague, A.F., and Cheraskin, E. “The improvement in renal function following EDTA chelation and multivitamin-trace mineral therapy: A study in creatinine clearance,” Med. Hypotheses 15 (11), 301-304 (1984).
- Riordan, H.D. et al., “A safe treatment for demonstrated chronic lead burden” (submitted for publication).
- McDonagh, E.W., Rudolph, C.J., and Cheraskin, E., “Serum cholesterol and the aging process,” Med. Hypotheses. 7 (6), 685-694 (1981).
- McDonagh, E.W., Rudolph, C.J., and Cheraskin, E., “Homeostatic effect of EDTA with supportive multivitamin-trace mineral supplementation upon High Density Lipoproteins (HDL),” J. Osteopathic Phys. and Surg. Calif. 8(2) (1982).
- McDonagh, E.W., Rudolph, C.J., and Cheraskin, E., “The influence of EDTA salts plus multivitamin-trace mineral therapy upon total serum cholesterol/high-density lipoprotein cholesterol,” Med. Hypotheses 9(6), 643-646 (1982).
- McDonagh, E.W., Rudolph, C.J., and Cheraskin, E., “The effect of intravenous disodium ethylenediaminetetraacetic plus supportive multivitamin/trace-mineral supplementation upon fasting serum calcium,” Med. Hypotheses II (4), 431-438 (1983).