Access to all articles, new health classes, discounts in our store, and more!
The Control of Bleeding
Published in The Journal of the American Dental Association, Vol. 58, pp. 17-28, April 1959.
* * *
Bleeding is the result of extravascular, vascular and intravascular defects. Most commonly, bleeding is the result of a defect in the vessel wall. Hemorrhage can be controlled by local and systemic measures. Local measures include: local pressure; rest; coagulation-promoting agents; thermocautery; application of ice; local vasoconstrictors; chemical escharotic agents; antimicrobial agents and sutures. Systemic measures include: rest; whole blood, serum and plasma; plasma fractions; platelets; ACTH and cortisone; estrogens; ascorbic acid; flavones; vitamin K; toluidine blue and protamine; the adrenochromes; antimicrobial agents, and splenectomy. The diagnosis of a bleeding disorder requires a history, clinical examination and laboratory studies.
A practical comprehension of the control of bleeding requires: (1) a clear-cut definition of hemorrhage, (2) a sound appreciation of the normal mechanism of hemostasis, and (3) knowledge of the many and varied hemostatic agents and measures.
Hemorrhage becomes clinically apparent when blood escapes from a blood vessel. The big stomatologic problem is the hemorrhage which follows an injury which, under normal circumstances, would not result in undue bleeding. This sequence of events is commonly observed after the extraction of a tooth, during or after a prophylaxis or gingivectomy, with toothbrushing, or it may seemingly be unrelated to any form of intraoral insult.
Mechanism of Hemostasis
It should be pointed out at the start that the physiologic control of bleeding is a complex, dynamic process. Only for descriptive purposes will it be dissected.
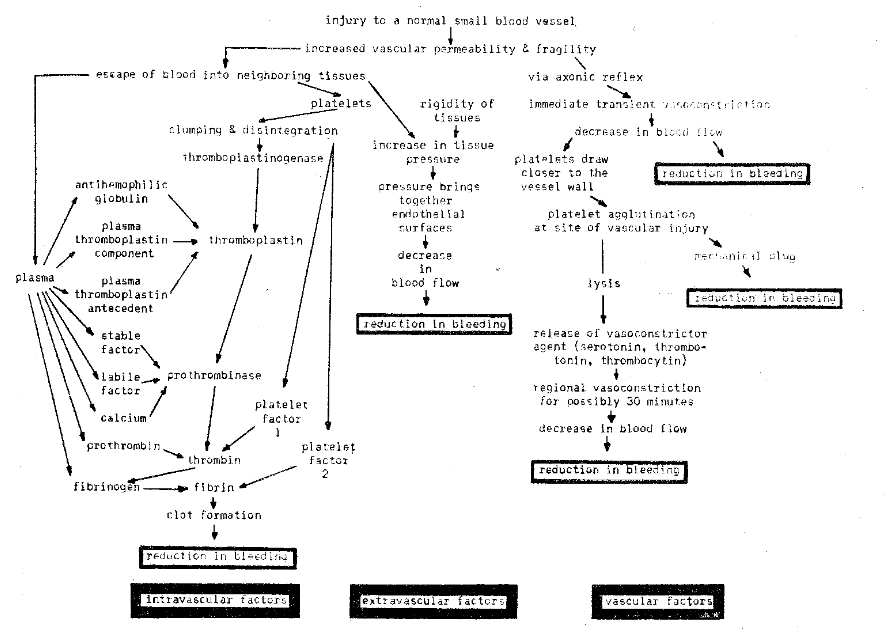
First, an injury to a small vessel leads to increased permeability and fragility (Fig. 1). Almost immediately, there is reflex smooth muscle contraction.1 With a reduction in the caliber of the damaged vessel, blood flow diminishes and may even cease completely. Obviously, should such vasoconstriction not take place, there is the possibility of excess bleeding.
Fig. 1–Diagrammatic explanation of the hemostatic mechanism. These may be grouped as intravascular (left), extravascular (middle), and vascular (right) factors.
After vascular trauma, blood escapes from within the vessel and enters the neighboring, perivascular tissues (Fig. 1). This then means that there are now platelets, other formed elements and plasma in the extravascular compartment. The escaping platelets stick together2 and liberate a platelet vasoconstrictor agent which has been variously designated as thrombocytin, thrombotonin and serotonin.3 The action of the platelets at this point is twofold: (1) they become adherent to the edges of the vascular wound and act as a mechanical plug, and (2) the vasoconstrictor substance tends to constrict the regional vessels even more, thus further reducing blood flow and acting in a hemostatic fashion.4
At the same time, the escaping blood creates an extravascular force. This pressure, plus that which normally prevails in the tissues, produces an even greater pressure against the vessel wall (Fig. 1). Such pressure tends to compress further the blood vessels, leading to an added reduction in the caliber of the vascular lumen, even greater slowing of blood flow, and a further diminution of hemorrhage. Obviously, this particular phase of hemostasis is not as important after the extraction of a tooth as it is in other types of oral bleeding.
The escaping blood, it should be recalled, contains platelets and plasma. Therefore, the interplay of the thrombocytes and various plasma fractions represents the next step in hemostasis (Fig. 1). It is clear that there is, within plasma, antihemophilic globulin (AHG),5 plasma thromboplastin component (PTG),6 a possible plasma thromboplastin antecedent (FTA),7 a labile8 and a separate stable factor,9 calcium, prothrombin and fibrinogen. The chart (Fig. 1) discloses the vital role of the platelets and the formation of thromboplastinogenase. Through this mechanism, thromboplastin is made available. The thromboplastin, plus the labile and stable factors and calcium, produces the enzyme prothrombinase which, in turn, is activated by platelet factor 1 and frees thrombin from prothrombin.10 Finally, the interaction of thrombin and fibrinogen and the accelerating effect of platelet factor 2 yield fibrin.11 The result is clot formation.
The sequence of events (not shown in the chart) is then clot retraction, possibly some degree of clot lysis, organization of the clot and vascular recanalization.
Disturbances in the Hemostatic Mechanism
It should be emphasized that the hemostatic process just outlined is indeed simplified. Much more is known than is here described and even more of the phenomenon requires clarification. However, it becomes abundantly clear from a composite of these oversimplified charts that the control of bleeding rests with the synchronized interplay of three factors: (1) extravascular, (2) vascular, and (3) intravascular (Fig. 1).
Extravascular Factors: The elasticity of the perivascular tissues serves two purposes: (1) it protects the vessels from damage, and (2) it resists or opposes the escape of blood from a traumatized vessel. Broadly speaking, bleeding due solely or almost exclusively to extravascular pathosis is encountered in two different types of situations:(1) when vessels course close to the surface of the skin or mucosa, and (2) when there is a loss of elasticity.
Apropos to the first situation, that is, vascular superficiality, it is of interest that a blood vessel which courses close to the periphery lacks the support of neighboring tissues on its surface side. This is especially true when the blood vessel is directed parallel to the surface. This point applies particularly to the oral mucosa because of its structural design and the customary traurna. With regard to the second point, it is well to point out that in the aged and in those with serious debilitating disorders, there is often considerable loss of subcutaneous and submucosal elasticity. This combination of extravascular circumstances may, in part, explain the ease with which bleeding occurs in some patients.12
Vascular Factors: There are other situations in which the extravascular tissues are normal and the intravascular blood fractions are present in adequate amounts. Still, bleeding is encountered.13 Generally, the difficulty can be traced to pathosis in the vessel wall itself.14 Weakness of the capillary and metarteriolar wall is the common denominator. Precisely why there should be such a fragile and permeable vascular barrier is not always clear. However, among the known causes are: (1) heredity, (2) infection, (3) nutrition, (4) hypersensitivity, and (5) hormones.
It seems clear that there can be a familial trait of vascular weakness. This possibility is well supported by the findings in hereditary hemorrhagic telangiectasia.15 Case reports with oral bleeding are available16 but no carefully tested studies are known to demonstrate the relationship of hereditary capillary weakness and oral bleeding.
Certainly, infection produces toxins which may exert a deleterious effect on the vascular endothelium. This mechanism is well demonstrated in the streptococcal invasion of scarlet fever, meningococcemia, and, for that matter, in any severe infection. It is likely, though the problem has not been seriously studied, that the toxins of the streptococcus and other microbes in periodontal and periapical infection may, in a similar fashion, weaken the surrounding endothelial tissue. Precisely how important this problem is, has yet to be tested under carefully controlled conditions.
Nutritional factors most assuredly play a vital role in the maintenance of endothelial integrity. For example, ascorbic acid is easily linked with the deposition and the maintenance of intercellular cement substance. There are numerous accounts in the literature17 to show the role of ascorbic acid deficiency states in oral bleeding.
There is the chance of a hypersensitivity reaction.18 The syndrome may take the form of an allergic response to a food, drug, or possibly even to an invading microorganism. In other words, the altered vascular permeability and fragility may signify an allergic response to microorganisms in the periodontal structures. The effect of allergy on hemorrhage is clearly shown in so-called anaphylactoid or allergic purpura. The stomatologic aspects of this problem have never been investigated under rigidly controlled experimental conditions.
Finally, evidence exists that hormones, notably those of the hypophysis (ACTH), adrenal cortex and the gonads (particularly the estrogens) play a role in vascular integrity. This is shown, for example, by the observation that a significant number of women bruise easily just prior to menses, that is, at the time when the estrogen levels are lowest.
Intravascular Factors: Hemorrhage may be due to a deficiency in the quantity or quality of: (1) platelets, (2) prothrombin, (3) fibrinogen, or (4) any one or combination of a number of other plasma fractions. Thus, in the final analysis, bleeding due to intravascular defects manifests a disturbance in one of three mechanisms which are termed: (1) thromboplastin deficiency states or hypothromboplastinemias, (2) prothrombin deficiency states or hypothrombinemias, and (3) fibrinogen deficiency disorders or fibrinogenopenias.
Hypothromboplastinemia: The role of thromboplastin in the coagulative mechanism has already been established. Its formation is impeded by a deficiency of: (1) platelets, (2) thromboplastinogenase, (3) antihemophilic globulin, (4) plasma thromboplastin component, and (5) other less well-defined plasma constituents such as plasma thromboplastin antecedent. In addition, a thromboplastin shortage may be the result of circulating anticoagulants which suppress the platelet and plasma elements so vital to the production of thromboplastin.
Hypothrombinemia: Strictly speaking, all coagulation defects, even the hypothromboplastinemias, with the possible exception of the fibrinogen-fibrin link, are the result of a defective production of thrombin. But, in its more limited connotation, hypothrombinemia is the result of a deficiency of: (1) stable factor, (2) labile factor, (3) calcium, or (4) prothrombin. Calcium plays only a minor role since the coagulative mechanism is not disturbed until the calcium level reaches a 50 per cent point–that is, a level below that for tetany.19 The general outline is pictorially represented (Fig. 1).
Fibrinogenopenia: An absence or reduction of fibrinogen may be congenital or acquired. The congenital types are exceedingly rare. The acquired forms are associated with severe primary or secondary liver disease and are of considerable clinical importance.
Incidence of the Hemorrhagic Diathesis
Generally speaking, the plaguing problem in the dental office is the person who reports that he has a “bleeding” tendency or the patient who is recognized as a so-called “bleeder” during or after a dental operation. More puzzling and embarrassing is the situation when the laboratory reports a normal bleeding and clotting time. The problem is then one of establishing the common cause or causes of oral bleeding.
In terms of frequency of occurrence, bleeding is most commonly the result of a defect in the vessel wall. It is therefore quite understandable why most bleeders show normal bleeding and clotting times. This was shown in a research project at the Cleveland Clinic where a group of 527 patients with a chief complaint of hemorrhage was examined extensively from a hematologic standpoint.20 This study pointed up the fact that less than one in every four individuals showed any significant intravascular defect. Such studies, limited as they are, all suggest that bleeding is generally due to increased capillary permeability and fragility. Next in order of frequency are the extravascular problems. Last, and therefore least common, are the hemorrhagic disorders due to a disturbance in the coagulative mechanism.
These epidemiologic observations are of cardinal interest. The literature suggests that coagulation defects such as hemophilia are rare. Yet, the bulk of writing concerns itself with these disorders. Conversely, although vascular pathosis is common, little is written about it. This paradoxical situation stems from the fact that more investigative work has been done and more is known about the intravascular mechanism as contrasted to vascular and extravascular processes. This, in turn, arises from the fact that more reliable tests are available for the study of the coagulative mechanism than for determining the status of the vessel wall.
Control of Hemorrhage
It should be abundantly clear that the mechanism of pathologic bleeding is complex and the result of one factor, or, usually, a combination of factors. Obviously then it is difficult to imagine that there can be one or a small group of therapeutic measures capable of controlling all the hemorrhagic problems encountered in the average dental office.
Broadly speaking, the hemostatic armamentarium may be divided into two groups: (1) local, and (2) systemic measures.
Local Measures: The local measures include those agents and devices applied directly to the bleeding part to reduce or eliminate hemorrhage. These include: (1) local pressure, (2) rest, (3) coagulation-promoting agents, (4) thermocautery, (5) application of ice, (6) local vasoconstrictors, (7) chemical escharotic agents, (8) antimicrobial agents, and (9) sutures.
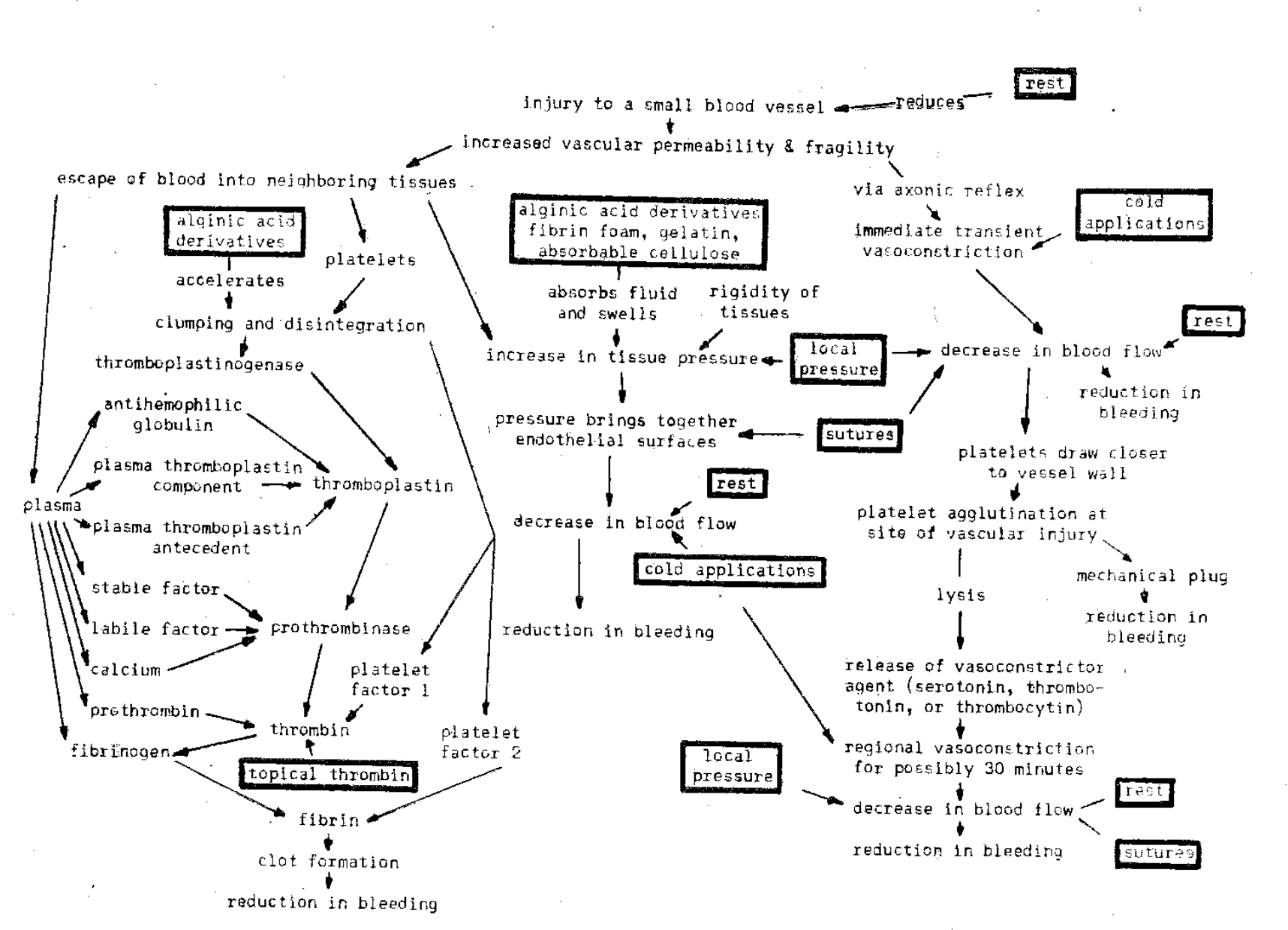
Local Pressure: Direct pressure to a bleeding wound is a simple and highly effective adjunct in the control of hemorrhage (Fig. 2). This is the procedure which is utilized after the extraction of a tooth when the patient is instructed to bite down on a sponge; in part here is the rationale for the use of periodontal packs. It should be emphasized, however, that direct pressure is most beneficial in those conditions in which vascular and extravascular factors play the cardinal causative role in bleeding. Thus, the local application of force is of benefit, for example, in hereditary telangiectasis, in the aged and debilitated patient, and in bleeding associated with nutritional, hormonal and allergic disorders. There are, in addition, agents available which are capable of absorbing many times their weight in blood with resultant expansion. This causes the material to swell, exert pressure and thus act as a mechanical barrier to the flow of blood. The physiologic basis, for example, of starch sponge21 and alginic acid derivatives22 appears to have merit in this regard but requires further evaluation.
Fig. 2–Action of local measures in the control of bleeding. Most of the measures are effective in the control of bleeding due to vascular and extravascular factors.
Rest: The oral cavity is unique in that it is an area which is being subjected to constant microtraumas. Thus, under normal conditions, capillaries are constantly being damaged and repaired. Obviously, the reduction or elimination of such microtrauma is highly desirable when pathologic conditions are present, since capillary permeability and fragility would be reduced (Fig. 2). This fact is clinically recognized as evidenced by instructions to the patient to imbibe liquids, avoid rough foods and reduce speech.
Coagulation-promoting Agents: There is no question but that agents such as fibrin foam,23 gelatin sponge,24 absorbable alkaline cellulose,25 topical thrombin,26 and alginates22 are helpful in the control of bleeding. These materials, combined with local pressure and rest, are highly desirable when the pathosis is of the vascular or extravascular type.
Thermocautery: The control of bleeding by cautery is helpful in those conditions in which the cause of bleeding is either in the vessel wall or extravascular. In other words, thermocautery is not desirable for the control of hemorrhage due to coagulation defects such as hemophilia. Actually, with thermocautery, the tissue is simply destroyed and thus coagulates. The end result is a mechanical plug which, for a time, can assist in the control of bleeding. However, it should be emphasized that other local measures are generally as effective if not more so than cautery.
Application of Ice: There is considerable physiologic evidence that the application of cold produces regional vasoconstriction and therefore a reduction of blood flow (Fig. 2). However, the vascular response is transient. Thus, cold applications can only be regarded as an adjunct in the control of hemorrhage due to vascular and extravascular pathosis. The effect of ice is enhanced when it is used in conjunction with other local and systemic measures for the control of bleeding.
Local Vasoconstrictor Agents: Sympathomimetic drugs are commonly applied locally for the control of bleeding. Most such preparations consist of 4 to 8 per cent racemic epinephrine. These drugs produce prompt but transitory vasoconstriction and thus reduce blood flow briefly. Unless the difficulty is within the vessel wall, its effect is insignificant.
Chemical Escharotic Agents: The use, and particularly the continued application, of escharotic agents such as silver nitrate, phenol, chromic acid and trichloroacetic acid is to be deplored.27 They act through tissue destruction. The hemostatic effect is therefore fleeting and other, more effective measures are available.
Antimicrobial Agents: The evidence, as already pointed out, is clear that infection can produce toxic damage to the vascular endothelium with a resultant increase in permeability and fragility. Thus, it might seem that the local application of chemotherapeutic and antibiotic agents to a bleeding site would be an effective hemostatic adjunct. However, though many such anti-infective preparations are available commercially, no extensive and carefully executed studies are available to support this premise as it pertains to oral bleeding.
Sutures: Suturing may be regarded as a helpful local measure. Here, the intent is to increase the extravascular pressure with resultant compression of the traumatized vessel, and slowing of blood. The use of sutures is helpful only in hemorrhage of vascular and extravascular origin.
Conclusions: It should be clear from the foregoing account that: (1) there are a variety of local hemostatic measures available, (2) they are most efficacious when used in combinations, and (3) they are generally indicated in situations in which the bleeding is largely or exclusively due to vascular and extravascular pathoses. It follows then that these local measures alone do not play a cardinal role in the control of hemorrhage due to coagulation defects.
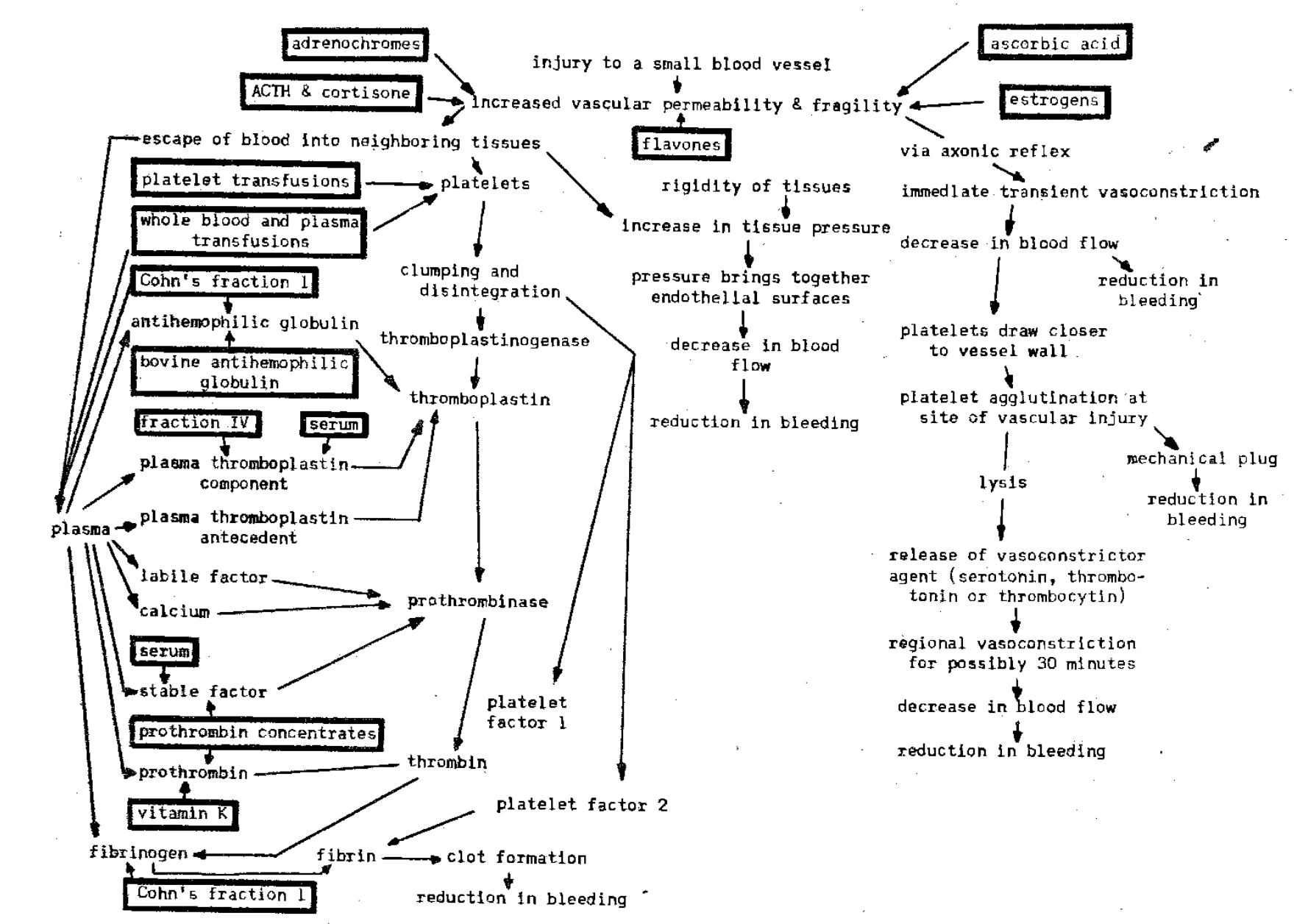
Systemic Measures: The systemic measures include those agents and devices administered by mouth to be swallowed, or injected subcutaneously, intramuscularly or intravenously with the expressed purpose of reducing or eliminating hemorrhage. These include: (1) rest, (2) whole blood, serum and plasma, (3) plasma fractions, (4) platelets, (5) ACTH and cortisone, (6) estrogens, (7) ascorbic acid, (8) flavones, (9) vitamin K, (10) toluidine blue and protamine, (11) the adrenochromes, (12) antimicrobial agents, and (13) splenectomy (Fig. 3).
Fig. 3–Action of systemic measures in the control of bleeding. Most of the agents are effective in the control of bleeding due to intravascular factors.
Rest: Reduced activity, not of the bleeding part, but of the entire patient, is a most important measure, because it invites sleep and because blood pressure and cardiac output are reduced. Thus, in any type of serious bleeding, it is highly desirable to reduce intravascular pressure. This can be furthered by bed rest.
Whole Blood, Serum and Plasma: In only one condition is it clear that whole blood transfusions are necessary–when hemorrhage has been severe enough to cause anemia. In addition, a transfusion of whole blood is helpful in any hemorrhagic state in which there is a depletion of one or more of the coagulation factors. In other words, bleeding due to a coagulation defect can be aided by whole blood transfusions if enough blood can be transfused. However, it should be emphasized that whole blood, serum and plasma may be wasteful and even dangerous. For example, plasma contains about 300 mg. fibrinogen and 7 mg. prothrombin per hundred cubic centimeters.28 Obviously, these amounts are too small to raise significantly the host’s level of fibrinogen and prothrombin in situations in which the coagulation defect is of these types. Thus, it is desirable to give the patient specific, purified fractions directed to replace the deficient fraction.
Plasma Fractions: A number of plasma components are now available and there is every reason to believe that more will become practicable in the very near future. Thus, it is possible to provide the patient deficient in plasma fractions with concentrated amounts of stable factor,29 labile factor,30 prothrombin,31 plasma thromboplastin component,32 antihemophilic globulin,33 and combinations such as Cohn’s fraction I which contains antihemophilic globulin plus fibrinogen.28
Platelet Transfusion: The importance of the platelets in hemostasis cannot be overemphasized. It should be recalled that the thrombocytes participate in almost every phase of the process. Thus, considerable research has yielded a significant advance in the classifications of technics for the collection, preservation and transfusion of platelets.34 The intent, when platelets are transfused, is to deliver not only intact thrombocytes but also their products. The indication for platelets is, primarily, certain types of thrombocytopenia, especially of the idiopathic variety.
ACTH and Cortisone: These hormones have been shown to exert a nonspecific but well-defined effect on the capillary wall.35 Though the mechanism is still open to question, it appears that these agents operate by decreasing capillary permeability and fragility. However, there is also a demonstrable shortening of the bleeding and clotting times, and stimulation of bone marrow activity.
Estrogens: Precisely how the female sex hormones act in the hemostatic process is still unknown. However, it is definite that they play a role. For example, it has been observed that, in women, bleeding seems to be more severe at the end of the secretory phase of the menstrual cycle. This is the period when the estrogen level is lowest and the urinary excretion of gonadotropin greatest. Other presumptions exist that telangiectatic and other types of vascular hemorrhage may be controlled with estrogens.36
Ascorbic Acid: The literature is replete with articles which underscore that ascorbic acid is essential for the synthesis and maintenance of the endothelial cement substance.37 Thus, it follows that an ascorbic acid deficiency leads to increased capillary permeability. This has been confirmed by the observation that the addition of vitamin C to the therapeutic regimen often reduces or eliminates hemorrhage.38 Most interestingly, verification that the effect of vitamin C is on the vessel wall is provided by the fact that ascorbic acid tends to reduce the hemorrhagic effect of bishydroxycoumarin at given prothrombin levels.39
Flavones: Flavones and their derivatives, including flavonones (hesperidin), flavonols (rutin), flavins (epicatechin) and coumarins40 have been shown to decrease capillary permeability. However, their action is slow. Consequently, they can only be used as preventive rather than therapeutic agents.
Vitamin K: This fraction is indispensable for the synthesis of prothrombin41 by the liver. It is often presumed that most hemorrhagic defects are the result of a vitamin K deficiency. At least, this agent is prescribed more often to dental patients than any other hemostatic drug42 despite the fact that it is seldom indicated. Evidence in support of this statement may be found in the Australian study in which only 1 of 232 patients undergoing routine exodontic procedures showed a prothrombin level as low as 75 per cent. Vitamin K exerts its principal effect on the coagulative mechanism and specifically on the production of prothrombin. Since, as has already been shown, intravascular pathosis is not common, it follows that, in most instances, vitamin K serves no purpose in the control of oral bleeding.
Toluidine Blue and Protamine: There are situations in which hemorrhage is due to the presence of circulating heparin-like factors. In such instances, toluidine blue, a complex dye with an affinity for certain circulating anticoagulants, or protamine sulfate may serve as a satisfactory hemostatic agent.43
Adrenochromes: The hemostatic effect of epinephrine has already been mentioned. However, its side effects and transient action preclude its usefulness except in the most minor types of vascular bleeding. The oxidation of epinephrine produces adrenochrome, which has been reported to be an excellent hemostatic drug without the usual sympathomimetic side effects. A product of adrenochrome, monosemicarbazone in combination with sodium salicylate, is presently available for oral and intramuscular administration. It is claimed that its principal effect is decreasing capillary permeability.44 However, its clinical usefulness must await determination by more carefully controlled studies.
Antimicrobial Agents: Mention has already been made of the local application of anti-infective drugs such as the sulfonamides and the antibiotics. Once again it is well to underscore the fact that invading microbes or their toxins or both can damage the vascular wall and so increase permeability and fragility. However, there is still no well-defined experimental study in stomatology to show that bleeding can be controlled with the use of antimicrobial agents.
Splenectomy: The precise role played by the spleen in the hematopoietic mechanism is still unknown. In recent years the term hypersplenism has received renewed attention to suggest that one crr more splenic functions may be overactive. Such overactivity may eventuate in a reduction of erythrocytes, leukocytes or platelets. Of particular importance to this discussion is the possibility of a platelet reduction with its attendant hemorrhagic dangers. Therefore, there are situations, admittedly uncommon, where splenectomy is indicated.
Diagnosis of Bleeding Problems
A diagnosis of any type of disorder, and therefore of a bleeding problem, can be reached only by: (1) the history, (2) the clinical examination, and (3) laboratory investigation.
History and Clinical Examination: It should be quite clear from an examination of the diverse origins of bleeding disorders that symptoms and signs may appear in any of the organs or systems of the body. For example, in the case of chronic, minimal bleeding, anemia is a likely sequela. The result is a reduction in the oxygen-carrying capacity of the blood. Thus, the patient complains of easy fatigability, restlessness, nervousness, dyspnea and general malaise; pallor and other evidences of chronic debilitation can be observed. These symptoms and signs are part and parcel of hundreds of syndromes and therefore are not pathognomonic of any one disorder.
There are some few symptoms and signs, however, which should alert the examiner for a possible hemorrhagic diathesis. These include: (1) ready bleeding after simple cuts and bruises such as during shaving, (2) easy bruisability, (3) a story of a bleeding tendency in other members of the family, (4) oral bleeding with minimal trauma such as with toothbrushing and eating.
Laboratory Examination. The most commonly employed laboratory tests for the detection of a hemorrhagic disease are the bleeding and clotting times. It is not intended in this report to analyze in detail the biochemical basis for these tests. However, the general statement can be made that if either or both of these tests prove positive, there is the strong possibility of a hemorrhagic disorder. On the other hand, if the bleeding or clotting times or both are within normal limits, and this is much more often the case, the possibility of a bleeding tendency cannot be eliminated. Thus, the question which must be answered is what laboratory tests should be performed in order to establish the presence or absence of a bleeding tendency.
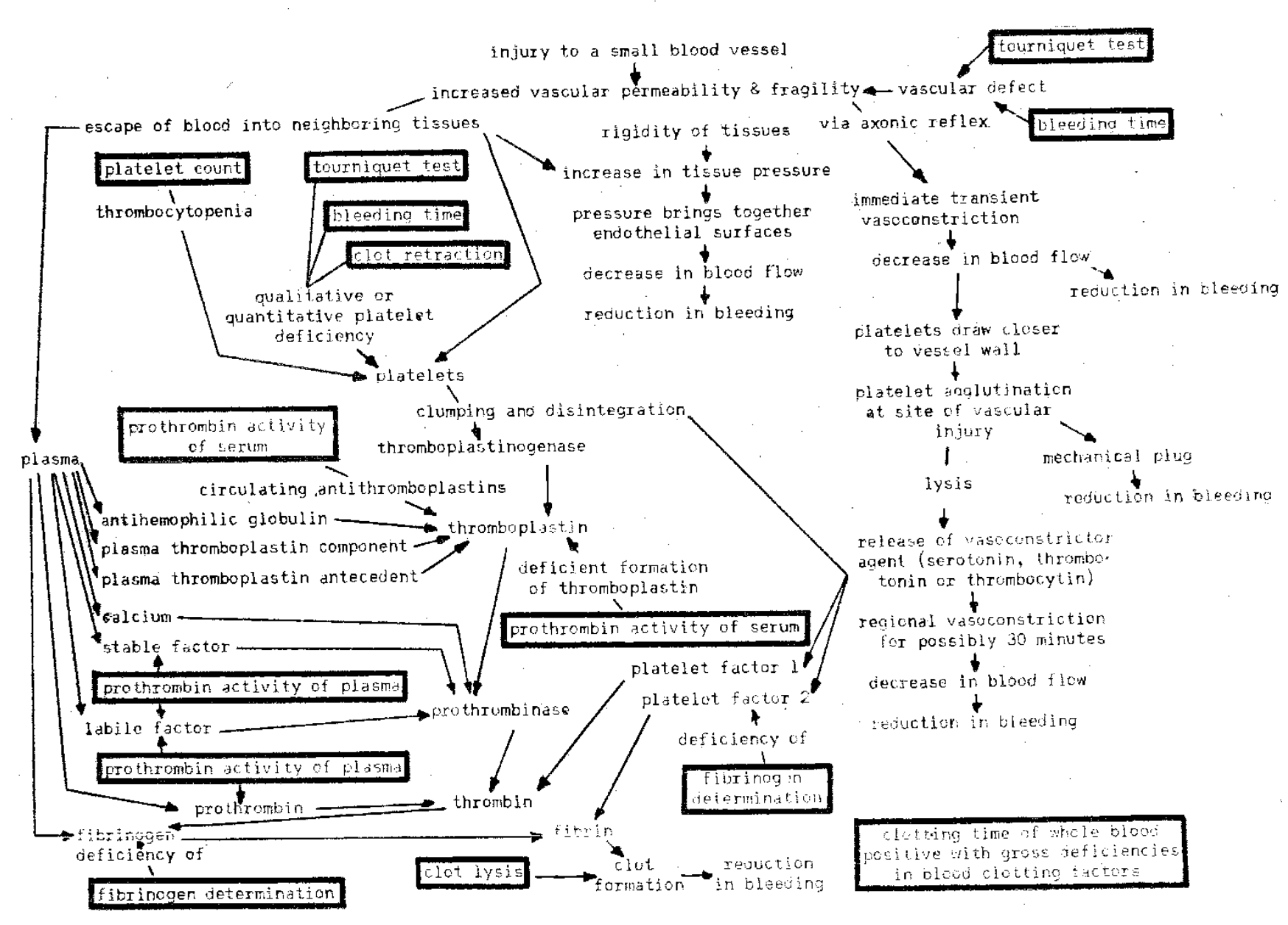
Stefanini and Dameshek27 recommend a battery of screening procedures which they insist should be carried out on every potential “bleeder” (Fig. 4). By means of these nine tests, most patients with a bleeding diathesis can be detected and even grossly classified. All of these procedures, collectively, require about 8 cc. of blood. Most of these tests, with the exception of clot lysis, can be completed within a two hour period.
Fig. 4–Significance of screening tests in detection of the hemorrhagic diathesis.
Summary
- Physiologic hemostasis involves the delicate interplay of reflex muscle contraction, the release of a platelet vasoconstrictor agent, extravascular pressure, and a complex chemical reaction among a number of plasma and platelet fractions.
- For diagnostic purposes, bleeding may be regarded as the result of extravascular, vascular or intravascular defects or all three.
- Studies on the incidence of bleeding suggest that defects in the vessel wall itself probably represent the most common causes of hemorrhage.
- A host of local and systemic measures are available for hemostasis. Generally, a combination of measures produces the most desired result.
- The diagnosis of a bleeding diathesis depends on a careful history, clinical examination and, most important, laboratory studies.
- For screening purposes in any instance of bleeding, it is recommended that the following nine laboratory tests be performed: (1) tourniquet test, (2) bleeding time, (3) platelet count, (4) clot retraction, (5) prothrombin activity of serum, (6) prothrombin activity of plasma, (7) fibrinogen, (8) clot lysis, and (9) clotting time.
References Cited:
- Chen, T. I., and Tsai, C. “Mechanism of haemostasis in peripheral vessels.” Physiol. 107:280 June 25, 1948.
- Stefanini, M. “Basic mechanisms of hemostasis.” New York Acad. Med. 30:239 April 1954.
- Bigelow, F. S. “Serotonin activity in blood.” Lab. & Clin. Med. 43:759 May 1954.
- Zucker, M. B. “Platelet agglutination and vasoconstriction as factors in spontaneous hemostasis in normal, thrombocytopenic, heparinized and hypothrombinemic rats.” J. Physiol. 148:275 Feb. 1947.
- Lewis, J. H., and others. “Relation of certain fractions of plasma globulins to coagulation defect in hemophilia.” Blood 1:166 March 1946.
- Aggeler, P. M., and others. “Plasma thromboplastinin component (PTC) deficiency: new disease resembling hemophilia.” Soc. Exper. Biol. & Med. 79:692 April 1952.
- Rosenthal, R. L.; Dreskin, O. H., and Rosenthal, N. “New hemophilia-like disease caused by deficiency of third plasma thromboplastin factor.” Soc. Exper. Biol. & Med. 82:171 Jan. 1953.
- Brennan, M. J.; Monto, R. W., and Shafer, H. C. “Hemorrhagic diathesis due to Ac-globulin deficiency.” J. Clin. Path. 22:150 Feb. 1952.
- de Nicola, P. “Factor VII (SPCA); its physiological significance.” Blood 8:947 Oct. 1953.
- Fantl, P., and Nance, M. H. “Physiological activation of prothrombin.” J. Austral. 1:128 Jan. 31, 1948.
- Eagle, H. “Studies on blood coagulation; formation of fibrin from thrombin and fibrinogen.” Gen. Physiol. 18:547 March 1935.
- Tocantins, L. M. “Mechanism of hemostasis.” Surg. 125:292 March 1947.
- Fantl, P., and Sawers, R. J. “Hemorrhagic diseases–their detection and management in dental surgery.” J. Den. 59:327 Dec. 1955.
- Spaet, T. H. “Vascular factors in pathogenesis of hemorrhagic syndromes.” Blood 7:641 June 1952.
- Alban, H. “Hereditary hemorrhagic telangiectasia.” Med. 40:86 March 1941.
- Syrop, H. M. “Hereditary hemorrhagic telangiectasia.” Oral Surg., Oral Med. & Oral Path. 10:253 March 1957.
- Schultzer, P. “Studies on capillary resistance; lowered resistance due to vitamin C deficiency and other conditions in hospitalized patients.” Acta Med. Scandinav. 81:112, 1934.
- Ackroyd, J. F. “Allergic purpura, including purpura due to foods, drugs, and infections.” J. Med. 14:605 May 1953.
- Stefanini, M., and Quick, A. J. “Quantitative studies on comparative activity of calcium and chemically treated ions on coagulation of blood.” J. Physiol. 152:389 Feb. 1948.
- Haden, R. L.; Schneider, R. H., and Underwood, L. C. “Abnormal hemorrhage with normal platelet count and normal clotting.” New York Acad. Sc. 49:641 May 11, 1948.
- Diconza, P. J. “Use of Terramycin starch sponge and starch powder hemostatic.” Oral Surg., Oral Med. & Oral Path. 7:1056 Oct. 1954.
- Frantz, V. K. “Experimental studies of alginates as hemostatics.” Surg. 127:1165 June 1948.
- Weiner, L., and Wald, A. H. “Fibrin foam and thrombin as used in surgical removal of large fibromyxoma of mandible.” A.D.A. 33:731 June 1, 1946.
- Light, R. U., and Prentice, H. R. “Gelatin sponge; surgical investigation of new matrix used in conjunction with thrombin in hemostasis.” Surg. 51:69 Sept. 1945.
- Seegers, W. H. and Sharp, E. A. Hemostatic agents. Springfield, Ill., Charles C Thomas, 1948.
- Tidrick, R. T.; Seegers, W. H., and Warner, E. D. “Clinical experience with thrombin as hemostatic agent.” Surgery 14:191 Aug. 1943.
- Stefanini, M., and Dameshek, W. Hemorrhagic disorders. New York, Grune & Stratton, 1955, p. 52.
- Cohn, E. J., and others. “System for separation of components of human blood: quantitative procedures for separation of protein components of human plasma.” Am. Chem. Soc. 72:465 Jan. 1950.
- Alexander, B.; Goldstein, R., and Landwehr, C. “Prothrombin conversion accelerator of serum (SPCA): its partial purification and its properties compared with serum Ac-globulin.” Clin. Investigation 29:881 July 1950.
- Owren, P. A. “Fifth coagulation factor (‘factor V’) preparation and properties.” J. 43:136, 1948.
- Seegers, W.H. Coagulation of the blood. New York, Academic Press, 1 953, p. 180-221.
- Aggeler, P. M.; Spaet, T. H., and Emery, B. E. “Purification of plasma thromboplastin factor B and its identification as a betas globulin.” Science 119:806 June 4, 1954.
- MacFarlane, R. G.; Biggs, R., and Bidwell, E. “Bovine antihaemophilia globulin in treatment of haemophilia.” Lancet 1:1316 June 26, 1954.
- Stefanini, M., and Dameshek, W. “Collection, preservation and transfusion of platelets.” New England J. Med. 248:797 May 7, 1953.
- Kramar; J. “Endocrine regulation of capillary resistance.” Science 119:790 June 4, 1954.
- Whittington, P. B. Jr. “Control of bleeding with intravenous estrogens.” A.D.A. 53:595 Nov. 1956.
- Pijoan, M., and Lozner, E. L. “Medical progress; physiologic significance of vitamin C in man.” New England J. Med. 231:14 July 6, 1944.
- Mead, S. V. “Studies of effect of ingestion of citrus fruit upon gingival hemorrhage.” D. Res. 23:73 Feb. 1944.
- “Blood clotting and allied problems.” Transactions of the First Conference, Flynn, J. E., editor. New York, Josiah Macy, Jr., Foundation, 1948.
- Levitan, B. A. “Biochemistry and clinical application of vitamin P.” New England J. Med. 241:780 Nov. 17, 1949.
- Collentine, G. E., and Quick, A. J. “Interrelationship of vitamin K and dicoumarin.” J. M. Sc. 222:7 July 1951.
- Sutherland, K. J. G. “Scientific evaluation of vitamin K therapy as a routine hemostatic measure.” D. J. 1:30 Feb. 1956.
- Allen, J. G.; Jacobson, L. O., and Grossman, B. J. “Study of the plasma defect in patients whose bleeding is temporarily controlled by protamine sulfate or toluidine blue.” Lab. & Clin. Med. 33:1480 Nov. 1948.
- Perkins, R. E. L. “Clinical investigation of adrenochrome monosemicarbazone sodium salicylate.” Oral Surg., Oral Med. & Oral Path. 10:230 March 1957.