Access to all articles, new health classes, discounts in our store, and more!
Symposium on Chemical Carcinogenesis: Part II. Carcinogenesis Associated With Foods Food Additives Food Degradation Products and Related Dietary Factors
Published in Clinical Pharmacology and Therapeutics, Vol. 4, No. 1, January-February 1963.
* * *
Since the genesis of tumors may be influenced to a lesser or greater degree by the nutrient environment furnished the host, frequent reference is made to the role of nutrition and dietary factors in carcinogenesis. A current and comprehensive review of nutrition and cancer has been presented by Tannenbaum.67 While the obvious association of cancer and nutrition was recognized earlier, a comprehensive assessment of the role of food and diet in the induction of neoplasms must more accurately be viewed in relation to the ingestion of food additives, food contaminants, processing degradation products, and other dietary components.8,74 Rapid advances in modern food technology have focused attention on dietary components other than essential nutrients such as food additives and processing degradation products. The utilization of agricultural chemicals as soil fumigants, plant growth regulators, and pesticides which may remain on fruits and vegetables if not adequately removed also contributes food contaminants to the diet of man. More recently, the nuclear detonations in weapons-testing programs as well as the expanded industrial and medical applications of radioisotopes have introduced radionuclides into the diet as food contaminants. While many of these materials and identified chemical compounds appearing either as additives, contaminants, or degradation products may not be labeled as poisons toxicologically, their latent effect must be assessed in terms of their potential contribution to induction of cancer.
The continuous introduction of synthetic materials as coating materials for films, packages, and containers for foods presents certain problems in terms of migration of these substances from the wrapper or container into the raw or processed food. Whereas some compounds are essential in processing, and for improvement of flavor and texture, other organic compounds are added strictly for coloring purposes and enhancement of acceptability. These food colors, coating materials, and a wide spectrum of other materials must be tested thoroughly for their toxic or carcinogenic properties prior to general use. Perhaps worth considering is the fact that condiments, flavorings, or seasoning agents have had unchallenged acceptance in home and commercial food processing operations for many years. An awareness of the potential toxic and carcinogenic properties of food additives culminated in the adoption of Federal legislation on Sept. 6, 1958, as an amendment to the Federal Food, Drug and Cosmetics Act (Public Law 85-929) stipulating that all food additives must be tested for possible toxicity. In addition, certain requirements relative to the carcinogenic potential of food additives and degradation products from processing methods, among which is the newer concept of preservation by means of ionizing radiation, are specified.9 In 1954, the Miller Pesticide Amendment of this Act also established a procedure for setting safe amounts for residues of pesticides on fruits and vegetables. In 1960, the enactment of the color additives amendment of this original legislation duly recognized the potential carcinogenicity of coal tar colors and prevented establishment of their harmlessness by the Food and.Drug Administration.
The rapid advances in our knowledge of biochemistry, pharmacology, and toxicology have resulted in certain refinements and improvement in methodology for subliminal pharmacology and bioassay for carcinogens. Perhaps a stronger motivating force for refinement in the methodologic approaches to assess long-term subtle toxic and latent carcinogenic effects has been the specific Federal legislation referred to above. Conventional and classic methods involving long-term chronic animal feeding trials have been used for appraisal of a toxic or carcinogenic agent, yielding thereto certain gross observations, important toxicity data, and tumor incidence from ingested food materials. These procedures do not adequately meet current requirements. Accordingly, comprehensive studies dealing with route and rate of absorption, levels of storage in the tissues, and ultimate metabolic fate are now requisite in order to elucidate the mechanism of biologic action of the dietary component under investigation. A comprehensive treatment of this phase of the problem is reviewed in other reports. A general discussion of principles is appropriate, however, before description of the series of observations reported on the genesis of neoplastic disease in animals and man mediated through the multiple factors associated with ingested materials.
General principles and considerations
In the ultimate evaluation of experimental findings, these are conditioned on multiple factors such as (1) satisfactory design of experiment, (2) identity and purity of material under test, (3) selection of animals and number of animals under investigation, (4) selection of range of doses, (5) route of administration–for food additives or contaminants, the oral route is the principal method, with subcutaneous or intraperitoneal injection or skin painting resorted to in confirmation of negative findings in feeding trials, (6) adequate postmortem and histopathologic examinations, and (7) nutritional and dietary variants. A detailed treatment of these factors is presented in a joint FAO/WHO Expert Committee Report on Food Additives and Publication 749 of the Food Protection Committee of the Food and Nutrition Board of the National Research Council.48
Role of nutrition and dietary variants.
There is considerable information on the effect of the macronutrients and micronutrients and enzymes on the appearance, period of latency, and duration of cancer. The general effect of diet may be considered in terms of the origin and the growth of tumors.
Caloric intake. A definitive statement on the influence of ad libitum feeding and caloric restriction on tumor induction is conditioned by age, strain, type of ration, method of caging, and voluntary exercise of experimental animal. In general, chronic caloric restriction inhibits the formation and growth of a tumor once it is formed. Boutwell,7 Iluseby,28 and Rusch,57 to name but a few, have reported on the formation and appearance of tumors induced by caloric restriction. The data in Table I reported by Tannenbaum68 are illustrative of the effect of caloric restriction during stages of carcinogenesis in mice to which benzpyrene was applied and in which skin tumors were induced.
Table I. Effects of caloric restrictions during the two stages of carcinogenesis.
Inhibition of growth of tumors has been shown by Bischoff and Long5 to be effected by underfeeding of starch or fat alone. Growth of transplanted tumors has been retarded through caloric restriction, as shown by Rous55 and by Suguira and Benedict.65 In addition, the variation in animals, the specified experimental conditions, the kind of tumor, and the dosage or potency of the carcinogen determine the critical level of calories which determines the development of a neoplasm.
Several concepts have been advanced in explanation of the mechanism of caloric restriction. It is believed by some that the mitotic activity of the tissue is inhibited by caloric restriction, and as part of this picture, carbohydrate or the intermediates of carbohydrate metabolism are a limiting factor. in cell division. Others believe that restriction leads to adrenal hyperfunction and a consequent increase in glyconeogenesis.7 A third hypothesis is that in caloric restriction, the level of estrogen, considered the primary carcinogen, is reduced, and thereby in the case of the mammary gland, this reduces the incidence of cancer.28
Macronutrients.
FAT. In general, fat enrichment of the diet augments the formation of certain tumors; butterfat, for example, increases the incidence of skin tumors produced by tar substances.2,29 In these investigations, ultraviolet light or hydrocarbons as primary carcinogens induced skin tumors at higher rates in mice on a high fat diet than on the control diet. Sarcomas, on the other hand, were not significantly altered by the high fat diet. A different picture is revealed by autoxidized fats as degradation products of foods, which will be described later. With respect to growth of tumors, variation in level of fat was virtually ineffective on the growth rate of various types.
Tannenbaum69,70 has suggested that the fat effect in genesis of tumors may be due to solvent action of fat deposited in tissue which accelerates the rate of transfer or alteration in dose level of carcinogen. Perhaps the fat alone has a direct effect on the developing neoplasm. Isolated fatty acids had effect equal to the natural triglyceride in the enhancement of tumor formation. Cholesterol seems to stimulate tumor formation, whereas phospholipids such as lecithin may retard, as may some other substances derived from the brain tissue if they are administered in large amounts.
PROTEIN. Although protein has a pronounced effect on the development of somatic tissue, the alteration in this macronutrient is not as significant as might be expected in terms of formation or growth of the tumor. Of course, results of feeding experiments may be confounded by effects of altered caloric intake and body weight. In diets fed ad libitum or calorically restricted, Tannenbaum71 and Rusch56 did not note any appreciable effect on sarcoma formation in mice.
The type of protein and composition, especially total amount of sulfur-containing amino acids, may play an important role, as, for example, in hepatoma formation. Sasaki and Yoshida,60 in feeding rats unpolished rice soaked in olive oil solution of o-aminoazotoluene, produced hepatomas in about 24 per cent of the population and had 25 per cent fatalities. American workers were unable to repeat this when the source of protein was wheat. Kinosita,35 feeding rats unpolished rice soaked in solution of p-dimethylaminoazobenzene (butter yellow), produced hepatomas in shorter time than Sasaki and Yoshida and found a higher yield of neoplasms with polished rice. Again, a bread or wheat protein diet reduced the rate of hepatoma formation. Yeast is particularly effective since a level of 15 per cent of it is almost completely protective, and any high quality protein diet and B vitamins, especially B2, are defensive mechanisms in inhibiting formation of such neoplasms.44
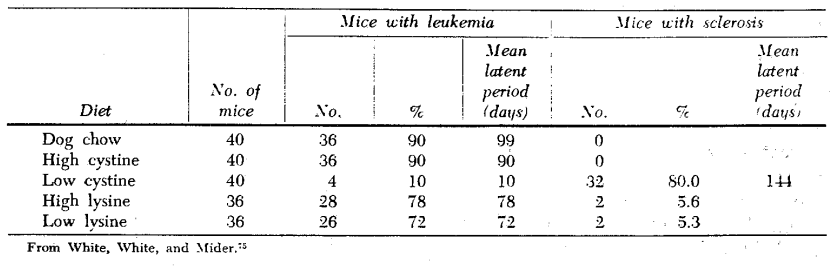
White has shown the influence of a low cystine diet on inhibition of leukemia induced by methylcholanthrene in DBA mice.75 Low levels of cystine and of lysine (the latter not as marked as the former) had a similar effect on the incidence of leukemia and the latency period, perhaps because of effect on a hormonal system (Table II).
Table II. Methylcholanthrene-induced leukemia in DBA mice as influenced by high and low levels of cystine and lysine in the diet.
Micronutrients.
VITAMINS AND MINERALS. Some vitamins appear to suppress while others stimulate malignant growth. According to Cramer,10 certain precancerous lesions of the gastric mucosa such as gastric polyps, atrophic gastric ulcer, and chronic atrophic gastritis have been attributed to deficiencies in vitamin A, riboflavin, and nicotinic acid. These lesions may advance into neoplasia in the presence of a carcinogen. In vitamin A deficiency, papillomas appear on the mucous membranes.10 Kline and co-workers33 noted possible inhibition of skin tumors arising from a pyridoxine-deficient ration administered to mice.
With respect to mammary carcinoma, Morris16 observed that riboflavin deficiency contributed to lowered incidence of these tumors. Day has reported on the procarcinogenic effect of vitamin B12 with rats fed p-dimethylaminoazobenzene.13 According to Russell,58 by depletion of a diet of thiamine and riboflavin, brain tumors were induced with methylcholanthrene.
Perhaps the most interesting observations are those of Japanese and American investigators31,49 on liver neoplasia in which riboflavin-rich diets inhibited tumor formation. This preponderance of evidence on hepatomas associated with low B vitamin diets can probably account for the high tumor incidence among the Bantus and certain Asiatic groups.22
Hepatic tumors have also been reported to occur in rats when a choline-deficient diet was administered to a strain of rats having a high choline requirement.15 In general, the injury of liver cells resulting from a nutrient deficiency may impair normal growth of cells, and carcinogens then may readily induce hepatomatous nodules.
Some attention has been given to the influence of inorganic compounds or minerals, more particularly in recent years since nutritionists have become interested in mineral requirements, their role in certain enzyme systems, and potential toxicity and carcinogenicity in levels above daily requirements for animals and man. Whereas many trace minerals at low levels are essential in metabolic functions, at higher levels they may be regarded as toxic and are potential promoters in presence of certain carcinogens such as the azo dyes. However, some effect of copper salts on the inhibition of tumor formation in the presence of azo dyes has been reported.32 Many of these trace metals are in foodstuffs such as seafoods. These elements are important biologically as metallo-enzymes or chelated compounds and are currently under study.
Some of the cations directly or indirectly implicated in the genesis of tumors are arsenic, beryllium, chromates, radium, and cobalt. The toxicity of arsenic varies widely with the nature of the compounds, and organic arsenicals, such as arsanilic acid, have been used chemotherapeuticaIly without any observed carcinogenic effect. Cancer of the skin and liver of vineyard workers exposed to arsenical pesticides in Germany has been reported.54 Experimentation thus far has failed to establish tumors caused in animals by arsenic. The validity of certain epidemiologic data on arsenic is equivocal.
In the scope of mineral elements, the presence of radioisotopes such as radium, uranium fallout radionuclides (Sr90; CS137, Ba140, I131, Sr59) K40, and C14 in our food, water supply, and other beverages has been considered as to potentiality for gene modification and increased frequency of neoplasms in succeeding generations. Further reference to these radioactive ions will be discussed under food contaminants.
Influence of additives, contaminants, and degradation products in foods on mutagenicity and carcinogenicity. A consideration of food intake in carcinogenesis is not restricted to nutrition per se but must also include additives, contaminants, or processing degradation products which may play a more important role in tumor induction. Inevitably in the processing of food for preservation and increased palatability, certain products are formed such as the hydroperoxides in autoxidation or heating of fats because of the action of oxygen on unsaturated lipids. These lipid peroxides, or organic peroxides, may act as inhibitors of catalase or peroxidases, and on this mechanism rests a possible explanation of the potential carcinogenic effect. Radiation, of course, produces effects comparable to autoxidation or polymerization through heating, and it is not surprising that radiation preservation produces end products in food comparable to other processing procedures. The hydroperoxides are readily absorbed by the organism and soon after ingestion can be detected in anatomic sites, such as the liver or adipose tissues of the muscles. These organic peroxides may attack vitamins, inhibit certain enzymes, affect the mitochondria, interfere with the function of cortisone, reduce the viscosity of deoxyribonucleic acid, attack hemoglobins, and destroy vital sulfhydryl linkages. At significant levels, they will, as result of vitamin destruction, produce sterility in rats and dogs (vitamin E destruction) and hemorrhagic diathesis in rats (vitamin K destruction).36,37 Organic peroxides may produce cleavage in the nucleic acid chain resulting in alteration in desoxyribonucleic acid through depolymerization, thus accounting for their mutagenic action in Drosophila, Neurospora, and Aspergillus.61 These effects are potentiated in some cases by the addition of certain carbonyls, particularly formaldehyde.
The assessment of carcinogenicity with respect to organic peroxides, epoxides, or other polymeric substances is relevant not only to toxicity but also to their potential mutagenic action as demonstrated in unicellular forms of life and insects. Matsuo43 has not only shown that removal of peroxides from autoxidized products enhanced the growth rate of rats, but with in vitro studies, he demonstrated the complexing of autoxidized ethyl esters of highly unsaturated fatty acids with ovalbumin.
The hydrocarbons as contaminants are ubiquitous in ingested materials, and some are classic carcinogens, such as 3,4-benzopyrene in smoked products, which will be discussed later. Recent interest has been shown in the concentration of polycyclic hydrocarbons in the drinking water, and this aspect has not been overlooked in the incidence of stomach cancer among the Icelanders.14
Irritants such as paprika, mustard, and others may possibly act as cocarcinogens or procarcinogens when carcinogens such as dyes or heated or carbonized foodstuffs are present in food or beverages, according to Steyn.63
It is difficult to establish whether the chemical carcinogen acts directly through metabolites, by hormonal mechanism, or potentiation by a viral agent. In the case of food additives or contaminants, it is believed that some hormonal mechanism is implicated. Hence, such compounds in food could be considered indirect agents or carcinogens. Food additives are now classified as those permitted on the basis of exclusion of a carcinogenic or potential mutagenic or carcinogenic agent. Currently, in a consideration of radioactivity (food radionuclides), maximal permissible concentration (MPC) values are set up as arbitrary guidelines below which level no potential hazard may exist. Interestingly enough, according to Public Law 85-929, no tolerance levels are legally permitted where the substance has been established as carcinogenic. It has been the practice to set tolerance levels for substances as alleged carcinogens which occur in the environment and contaminate food.
With secondary contaminants such as a carcinogen identified and extracted from petroleum waxes, plastics, or processing degradation products, i.e., from smoking or heating, certain requirements are set, and the process is modified or limited to reduce the compound to an extremely low level. Pesticide residues, for example, are reduced to the lowest practical level when proved as carcinogenic, or in many cases, their use on field crops is completely banned. Fallout radionuclides, which have implications as carcinogens, are at levels which in some cases are hard to detect even by sensitive methods. Nevertheless, should the concentration increase to a level at which somatic tissue damage is envisioned, remedial measures would have to be instituted to reduce this contamination to levels below the Federal Radiation Council guidelines.17
Specific references to food additives, food contaminants, and food degradation products in carcinogenesis
Food colors and additives. The basis for acceptance or rejection of these substances applied by the food industry has been largely through extensive animal experimentation. Whereas the critical appraisal is on the basis of oral feeding, and certainly a proved carcinogen by this technique would be rejected by Food and Drug authorities, if tumors are demonstrated by skin painting or injection, this may also be the basis for rejection.
Auramine O and tetramethyldiaminodiphenylcetonimine, are representative of carcinogenic food colors, and the component b-naphthylamine in yellow OB and AB or other impurities in this color may be carcinogenic. In the Union of South Africa, according to Steyn,63 nigrosine and benzopurpurin have been removed from permitted list of dyestuffs on the basis of toxic effects and not at the moment because of tumorigenic incidence.
Since carbon blacks have been shown to contain 3,4-benzpyrene as a contaminant, these materials, along with activated charcoals used as food colors, have been investigated. The petroleum waxes used in food packaging and as food additives (chewing gum) have been and are receiving some investigation, since certain petroleum fractions have been shown to be highly carcinogenic.19 In feeding rats D & C red No. 9 for 2 years, Davis and Fitzhugh11 found no apparent effect on the growth rate, mortality, and occurrence of tumors from this food color.
Dulcin or p-phenetylurea (Valzin or Sucrol) used as sweetening agent has been shown to be toxic and hepatomatous to experimental animals.18 Fitzhugh18 compared the chronic toxicities of P-4000 (2- amino-4-nitrophenylpropyl ether), cyclamate sodium, and saccharin, and on these tests, Dulcin was banned by the U. S. Food and Drug Administration as a food sweetening agent.
Most of the flavoring agents are not carcinogenic. However, recent studies by the U. S. Food and Drug Administration72 have demonstrated safrole (component of oil of sassafras) to be carcinogenic. Safrole occurs as a constituent of essential oils of cinnamon, nutmeg, and mace and has been made synthetically. This material is a flavoring agent in root beer and sarsaparilla. At a level of 5,000 p.p.m. when fed to rats, safrole not only caused liver injury but produced tumors. Survival rate was affected down to levels of 100 p.p.m. Within recent years, safrole has been discontinued as a flavoring component in beverages, and this compound (isosafrole and dihydrosafrole) has been prohibited in food. No action has been taken relative to use of cinnamon, nutmeg, or other natural substances containing low levels of safrole. Dietary factors may enhance or protect against carcinogenic or toxic effects of safrole.26 Differences in the hepatic susceptibility may be strain and species dependent, and sex and age may have some influence. Further studies with dogs are under way to confirm these findings in another test species.
Certain emulsifying agents in foods such as Tween 60 (polyoxyethylene sorbitan monostearate) and in an antibloom agent for chocolate retarded growth in rats and hamsters and produced papillomas and carcinomas on skin of mice and sarcomas when injected. This material is an excellent activator or promoter of tumors when applied to skin of mice previously treated with carcinogens such as the polycyclic hydrocarbons.42 There is no evidence that tumors are produced when this compound is fed.51
Polyoxyethylene monostearate (Myrj 45), proposed as an antistaling agent in bread, fed to rats at a 25 per cent level in the diet produced some bladder cancer. At lower levels, these tumors were not produced. Feeding at such high levels is not realistic and is perhaps undesirable from a toxicologic viewpoint; however, the application of this material in bread manufacture has not been permitted.47
As a stabilizer and emulsifier ice cream manufacture, carboxymethylcellulose has been used. Feeding experiments with rats on this material have been negative, but subcutaneous injection in rats for a 2 year period resulted in sarcomas.42
In beverage manufacture and other uses, tannic acid is an effective clearing agent. Tannic acid is also present in tea and other foods. Hepatomas have been produced in rats when this material was administered by parenteral injection, although not by feeding.34 No restriction against its use has been imposed since the tumors observed have not resulted from feeding or occurred at the site of injection.
Orange TX (FD & C orange No. 2) and 1-benzene azo-2-naphthol (orange E), structurally similar to FD & C red No. 32 and citrus No. 32, have been shown by Bonser, Jull, and Clayson6 to produce mammary and lung tumors in mice. FD & C red No. 32, used for dyeing oranges, has been banned for its high toxicity. A preservative used in cheese, 8-hydroxyquinoline, has been shown to produce cancer of the breast, cervix, and brain in rats.
For fattening of poultry and cattle, diethylstilbestrol has been used as implanted pellets and as an additive to rations and has been shown to have carcinogenic properties.21 The fact that a tissue residue remains in poultry from pellet implantation has resulted in prohibition of its use, but in livestock feed it has not been discontinued in the United States.
Food contaminants. The wide scale use of agricultural chemicals and pesticides has invoked certain requirements as to plant residues, specifically those proved experimentally to be carcinogens.
Aramite, used for control of mites in certain fruit crops, has been applied in this country at level of 1 p.p.m. This chemical (2-[p-tertiary-butylphenoxy] isopropyl-2-chloroethyl sulfide) contains a small amount of 2-(p-tert.-butylphenoxy) isopropyl sulfite. In feeding trials with rats (2 years) and dogs (1 year) and subsequent carcinogenicity studies with several strains of rats and mice and dogs, hepatomas were produced in several species at the level of application of 1 p.p.m., which is used on fruits.50,62 Whereas this acaricide had been in use for 8 years, zero tolerance levels for this compound have been established. It is currently used on nonfood crops.
The widespread use of the fungicides thiourea and thioacetamide for citrus fruits was discontinued when feeding of these compounds produced hepatomas in rats.20 Thiourea, which is known to be thyrotoxic, has also been observed to produce thyroid tumors in rats and mice.53 Perhaps a plant residue of more recent national interest is the effective weed killer aminotriazole (3-amino-IH-1,2,4-triazole), which was used in cranberry bogs. While there seemed to be no hazard if this plant growth regulator was used well in advance of the harvesting of cranberries, concern was shown when the compound was applied prior to harvesting and hence there was the possibility of residue. This compound, when fed to rats at 15 p.p.m., produced thyroid adenomas and some adenocarcinomas. It has been stated that similar goitrogenic substances occur in foods, particularly the Brassica, and propylthiourea is used therapeutically with no evidence of tumors in man. Nevertheless, a zero tolerance has been set for this weed killer in this country.30
Another plant growth regulator, IPC (isopropyl-N-phenylcarbamate), also a weed killer, is used in sprout inhibition for potatoes. Again, this chemical leaves a residue in food. By feeding of this compound alone to mice and rats, no tumors were produced. However, if it was given orally to mice in conjunction with painting the skin with croton oil in olive oil, after 6 months, skin papillomas were produced. The incidence of papillomas was greater for the test group than for the control group (solution of croton oil). Another method for sprout inhibition used currently in Russia and Canada is the treatment of potatoes with a 7,000 rad. dose of 𝛾 radiation. Since radiation processing does not introduce the problem of residue (no induced radioactivity with a Co60 source), this method may warrant serious consideration to obviate the use of chemicals. The Netherlands has established a provisional tolerance for IPC.16
The wide spectrum of pesticides developed and applied in this country represents a potential carcinogenic hazard. Indeed, many experimental insecticides such as 2-acetylaminofluorene never reached the marketable stage because of their major carcinogenic properties. The literature is replete with data on the tumorigenic potential of 2-acetylaminofluorene.4 DDT, widely found as a contaminant (milk, meat, fodder, etc.), is a weak carcinogenic agent, but more extensive studies are required on this pesticide.19 Recent studies by Davis and Fitzhugh12 have shown that aldrin and dieldrin, common pesticides, were not considered carcinogens since tumors produced in mice were morphologically benign.
Certain molds and fungi can also contaminate food supplies. Liver injuries, acute, subacute, and chronic, have been induced in ddD mice and Wistar rats fed rice contaminated with Penicillium islandicum according to Miyake and associates.45 These mice had nodular hyperplasia of the liver and adenomatous nodules. The metabolites produced by moldy rice grains were quite toxic, and the etiologic agents have been identified as a yellow toxic substance, which is luteoskyrin, and a colorless toxic substance, which is a chlorine-containing peptide. A diet with as low as 1 per cent moldy rice was fatal to some mice after 200 days. Primary hepatic carcinoma was found in rats dying at 548 to 553 days on a diet with 5 per cent moldy rice.
A new disease called “turkey X disease” has recently been reported relative to high death rate of turkey poults in England in 1960. Postmortem examination of dead poults from field studies revealed acute hepatic necrosis associated with generalized bile duct proliferation. Outbreaks of this disease followed the use of a ration containing Brazilian peanut meal. While the results with poults pointed to hepatotoxicity, other studies with rats fed 20 per cent of this meal in the diet led to reduced food intake, lessened growth rate, and severe liver lesions. The nodules in the liver of rats fed this peanut meal consisted of liver tumors of three types: solid yellowish lobulated hepatomas and blood-filled or bile-filled cysts. The acute liver damage in turkey poults was not produced in rats, but rather, multiple liver tumors (nine out of eleven), of which some had lung metastases, give evidence to the hepatotoxicity and carcinogenicity of this agent. Toxic peanut meal causes hepatic changes in cattle, pigs, and sheep. A dose of 20 µg of the extracted principle from toxic meal fed orally to 1-day-old ducklings was fatal in 24 hours, but lesser amounts produce the liver lesion.40,59
Further work on the extract from meal (which has been characterized in products from countries other than Brazil) has established that the toxin-producing agent or fungus is Aspergillus flavus Link ex Fries. Apparently, certain strains of Aspergillus will produce this pathologic state and others will not since there is a body of information in this country on the feeding of moldy cereal grains in which such findings have not been reported relative to the hepatotoxic or tumorigenic effects. Richardson52 has reported on the effect of a moldy diet on the growth and mortality of chicks and poults with no tumorigenic findings.
With nuclear detonations and increased use of nuclear energy, the diet is now contaminated with radioactivity. An extensive research effort is now under wav by the U. S. Public Health Service and the Atomic Energy Commission to ascertain the effects of low level radioactivity relative to somatic and genetic hazards and, more specifically, the potential leukemogenic or sarcomagenic properties. While the occurrence of radionuclides in foods, other than fallout radioisotopes, is frequently overlooked, it is well to recognize the level of these in foods, beverages, and potable water supplies. Mayneord stated that the British diet contained amounts of radium and thorium equivalent in effect to some 300 times the present intake of Sr90 from fallout. For example, in 4 ounces of Brazil nuts, there is 3 times the amount of radium and thorium found in all the bone and soft tissues of a normal adult.
Kraybill38,39 has tabulated the relative contribution of radioactivity in foods from various sources, including fallout and the naturally occurring elements K40, Ra226, and C14, etc. These are listed in Table III.
Table III. Natural and fallout radioactivity in foods
The body at equilibrium has a burden of 130,000 µµc of K40. The effect of K40 has never been established, as has neither the influence of low levels of radium, but epidemiologic studies are now under wav to ascertain, if any, the influence of uranium and radium in certain rivers and its potential contribution to cancer incidence or other health hazards (Farmington, New Mexico, Studies). With radiation, there is not the issue as to threshold value below which effects are not harmful but rather of a stopping point where dilution in space is so great that the incidence of tumors attributable to fallout is considered negligible in comparison with end achieved.
Food processing degradation products. Within the last decade, investigators have concentrated on the influence of degradation products of foods on biologic response of animals and man. As early as 1939, Widmark73 concluded that extracts of roasted foods contain one or more carcinogenic or cocarcinogenic agents. In this category, one might consider roasted coffee, autoxidized fats, or browned butter. In his early work, Roffo found tumor growths in the stomach, liver, and cecum of rats fed on bread and milk to which was added animal fat or olive oil that had been heated to 3500 F. for ½ hour. Since that time, many experiments have been completed on heated fats showing their toxic effects, vitamin destructive potential. (vitamins A, E, and K), and cocarcinogenic properties with chemical carcinogens. Beck and Peacock3 found that fats (cottonseed and whale oils) heated to 200 to 2200 C. in an iron saucepan for 30 minutes on 30 consecutive days and fed to rats caused papillomas of the stomach associated with vitamin A deficiency.
More recently, Sugar and Kummerow64 have shown that a nonurea adduct-forming fraction isolated from heated corn oil acted in synergism with 2-acetylaminofluorene and enhanced its carcinogenic activity. In this work, only 0.005 per cent of 2-acetylaminofluorene and 2.5 per cent of the nonurea adduct-forming fraction from heated corn oil induced ear duct and liver tumors. Unpolymerized or fresh fats are not capable of inducing these tumors. The inference from these studies is that overprocessing of foods or food fats is unwarranted.
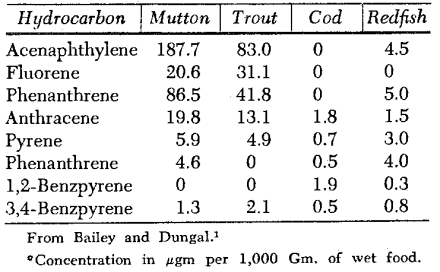
Another processing method or preservation procedure is that of smoking. Smoked food has been shown to contain 3,4-benzpyrene23 (1.9 to 10.5 µg per kilogram for smoked sausage and 1.7 to 7.5 µg per kilogram for smoked fish). The benzpyrene contained in 1 Kg. of smoked mutton corresponds to the quantity contained in the smoke condensate of 250 cigarettes.14 The concentration of this carcinogenic polycyclic hydrocarbon and other hydrocarbons in food is shown in Table IV as presented by Bailey.1
Table IV. Polycyclic aromatic hydrocarbons in smoked food
From epidemiologic studies according to Dungal, the incidence of neoplasms among Baltic or Icelandic fishermen eating these smoked fish is 3 times that for inland populations and specifically for gastrointestinal cancer 4 times that in a population on non-smoked fish diets. The stomach cancer incidence in Iceland represents about 35 to 45 per cent of all malignant tumors.
The use of uncontaminated wood and purified smoke extracts is important, and in many places, liquid smoke treatment is being utilized for preservation and flavor. The channel process for making carbon black, for example, is now used since this material is not contaminated with 3,4-benzpyrene.
Ever since hatchery-reared rainbow trout have been on dry pelleted rations, liver tumors have appeared. The first observations were made on yearling fish, and in some groups, the incidence was as high as 70 to 80 per cent. The lesion is quite specific for one strain of fish, the rainbow trout, and fish maintained on fresh liver, meat scraps, or a purified ration have practically no incidence of hepatomas. Thus far, the causative factor would seem to point to a component of the dry diet. Not all dry commercial rations will produce liver tumors, and only a few “hot diets” will induce a high incidence of tumors. It would appear that extensive processing of the plant and animal protein meals used in the ration may be involved. Recent work by Halver, Johnson, and Ashley24 and by LaRoche and colleagues41 points to a lipid fraction in fish meal or cottonseed meal portion; these materials probably have a high polymerized-oil content which impairs the intermediary metabolic pathways of this macronutrient.
Extensive research is now under way to isolate and identify the etiologic agent and to determine what practical measures can be taken to eliminate the processing degradation products responsible for induction of hepatoma. The disease appears in both the nodular and massive forms, and metastases have been observed in hemapoietic tissue of the kidney and spleen. These studies are important in extending knowledge on the mechanism of dietary carcinogens.27
Relative to hydroperoxides and polymerized lipid products, it is interesting that Harman25 has obtained inhibition of spontaneous cancer by antioxidants as well as prolongation of life span in C3H and AKR mice. At the 1 per cent level in a pelleted diet, 2-mercaptoethylamine prolonged half survival time of C3H female mice from 14.5 to 18.3 months. This antioxidant also produced a marked decrease in the tumor incidence of C3H female mice. Perhaps the antioxidant inhibits the formation of oxidized polymerized material (mitochondrial lipids) and may decrease the probability of free radical attack on cell constituents. As for tumor formation, it is possible that hydroxylamine may depress the respiratory enzyme in tumor cells or may protect some area of the desoxyribonucleic acid involved in initiation of the cancer.
Lymphosarcoma and cancer of the lungs were the frequent malignant states reported in the TM (tropical medicine) strain of mice on the Rockland diet supplemented with raw egg yolk and hard boiled egg white, according to Szepsenwol.66 These results reported by the Puerto Rico investigators have been challenged by others in the United States who have failed to confirm these findings. It is possible that a different strain of mice, presence of a viral agent in the mouse colony, or pathogens in the product tested may have some bearing on these controversial findings. Until these experiments are confirmed or not validated, the concept of egg protein being a carcinogen is indeed equivocal.
While radiation processing is not at the moment an accepted commercial procedure, it is important to recognize that only certain radiation sources would be currently approved relative to absence of induced radioactivity. For food processing, low energy sources (Co60 at 1.13 and 1.33 Mev.) at least below the threshold energy level of 12 Mev. would be recommended. Measurable induced radioactivity would be possible above this energy level. Linear accelerators with beam energies up to 24 Mev. will give rise to induced radioactivity. The radioisotopes produced are Na22, P32, P33, S35, Mn54, Zns65, Rb84, and I126. These radionuclides decay in a short time on storage of food, and the levels are below the maximal permissible concentration values for water as reported in National Bureau of Standards Handbook 69. Despite this, should such a process be used utilizing high energy radiation sources, the non carcinogenic properties of this low level radioactivity in food would have to be established in accordance with the Delaney Amendment (Public Law 85-929).
Concluding Comments
In a critical assessment of the factors involved in the genesis of neoplastic disease, the role of ingested materials, food, drugs, and water cannot be overlooked as a major environmental effect. Earlier studies with experimental animals and more indirect clinical observations with man directed attention to the importance of caloric intake and the effect of protein fat and vitamins on the initiation and growth of tumors. Dietary intake or nutrition per se must in general be viewed as a modifier in development of a neoplasm rather than the causative agent. A low level of nutrition, minimal calories, and submarginal deficiencies of vitamins tend to inhibit and retard the growth of the tumor. However, these conditions may also have adverse physiologic effects.
More recently, attention has been focused not on the role of nutrition but rather on the specific and additive influences of perhaps more important contributors, such as the materials that are added to food (in processing), contaminants such as spray residues, and finally, through processing, canning, or heating, the degradation products that are ever present. It is not surprising with the present state of the art and advances in food research and technology that over 800 chemical substances find their way into foodstuffs. Many of the chemical additives such as emulsifiers and stabilizers for aqueous and fat phases are designed to facilitate processing of many food products. Others enhance color, improve palatability, extend shelf life, and modify texture. To prevent insect damage to fruits and vegetables and to control size of plants, pesticides and plant growth regulators have become an integral part of agricultural practice. Spray or plant residues of these chemicals may not be completely removed in some cases prior to consumption and hence contribute to food contamination.
Recent legislation has required that toxicity tolerances be established for many substances, and if it is proved carcinogenic, such a chemical or additive may be completely restricted. Food and drug regulatory groups have accordingly evaluated many materials for their potential toxicologic or carcinogenic properties; some have been cleared and are on approved lists, while others have been rejected.
Current research has established the fact that some chemicals or food degradation products such as the hydroperoxides may, in the presence of a chemical carcinogen or a latent viral agent, serve as promoters in the induction of certain tumors. Such examples as moldy peanut meal and dry pelleted trout ration with certain degradative products may be illustrative of the effect of these etiologic agents in the formation of liver tumors in rats and rainbow trout. The effect of agricultural chemicals or insecticides which may inadvertently enter food must be removed to obviate the possibility of initiating and accelerating tumor growth. The effect of low level radioactivity, specifically fallout radionuclides, on carcinogenic processes has not been assessed as critical on the basis of current experience or experiments. However, the long-term or latent effect of these radioactive elements does remain in doubt.
More extensive testing and research are needed to achieve a better understanding of the mechanism of action of food additives, contaminants, and especially the lesser-known processing degradation products. With respect to processing and preservation of foods, freezing, freeze drying, cold storage, or refrigeration of foods would appear to present the least deteriorative changes. Since current investigations have directed attention to the effect of processing on nutrient value and potential toxicity, the synergistic action of degradative end products must also be appraised in the presence of a chemical carcinogen and the subsequent induction of a neoplasm.
The effect of heated fats on induction of certain tumors, specifically hepatomas, is illustrative of the property of these breakdown products to promote tumors in rats in the presence of classic carcinogens such as 2-acetylaminofluorene.
The probable influence of multiple factors and the diverse characteristics of different neoplasms contribute much to the complexity of the problem and preclude the establishment of a single chemical entity as a food additive, contaminant, or breakdown product as solely responsible for induction and growth of a tumor. With more extensive research, however, it is hoped that a clearer understanding of the mechanism will provide some insight into the specific role, if any, of all these associated factors and nutrient levels on the fundamental development, growth, or regression of malignant cells.
Summary
Extensive animal investigations and clinical evidence in man demonstrate that cancer is a disease which is enhanced through an optimal nutritional status of the host. Caloric restriction and marginal vitamin deficiencies serve to inhibit tumor formation. There are some notable exceptions, however, where dietary deficiencies potentiate formation of tumors. Current attention through research and testing is directed to the role of food additives, pesticides as contaminants, and heat-processing degradation products (organic peroxides, etc.), in their synergistic action or as promoters in tandem with primary chemical carcinogens in the genesis of neoplasms. Some food colors previously approved and utilized in food industry have now been rejected on basis of cancer-producing properties in animals. Similarly, certain pesticides have been discontinued on the same basis.
The association of processing degradation products arising, for example, from heating and smoking with tumor induction has directed attention to these products as potential etiologic agents. Gastrointestinal or stomach cancer from continuous ingestion of smoked meats containing 3,4-benzpyrene as observed in Icelanders is illustrative. Treatment with azo dyes of rats which were maintained on diets containing heated fats induced hepatomas, showing the effect of autoxidized lipids as promoters. Liver tumors have also been observed in rats consuming certain moldy peanut meals and in trout fed dry pelleted rations containing allegedly polymerized lipids, providing additional evidence of the effect of degradative products and contaminants in tumor production.
The sequence of events leading to the establishment of Federal legislation requiring evaluation of a wide spectrum of food materials in terms of potential toxic or carcinogenic properties is discussed. Examples of food additives, food contaminants, and food degradation products implicated in various types of tumor induction are cited. Exploitation of the.leads from this area of research is considered important in providing a better understanding of the mechanism of carcinogenesis.
References Cited:
- Bailey, E. J., and Dungal, N.: “Polycyclic hydrocarbons in Icelandic smoked food,” Brit. J. Cancer, 12:348-350, 1958.
- Baumann, C. A., and Rusch, H. P.: “Effect of diet on tumors induced by ultraviolet light,” Am. J. Cancer, 35:213-221, 1939.
- Beck, S., and Peacock, P. R.: “Gastro-papillomatosis due to vitamin A deficiency induced by heated fats,” Brit. M. J. 2:81-83, 1941.
- Bielschowsky, F.: “Distant tumors produced by 2-amino and 2-acetylaminofluorene,” Brit. J. Exper. Path. 25:1-4, 1944.
- Bischoff, F., and Long, M. L.: “The influence of calories per se upon the growth of sarcoma 180,” Am. J. Cancer, 32:418-421, 1938.
- Bonser, G. M., Jull, J. W., and Clayson, D. B.: “Induction of tumors with 1-(2-tolylazo)-2-naphthol (oil orange TX),” Nature, London 174:879-880, 1954.
- Boutwell, R. K., Brush, M. K., and Busch, H. P.: “Some physiological effects associated with chronic caloric restriction,” Am. J. Physiol. 154: 517-524, 1948.
- Caspari, W.: Nutrition and cancer, Paris, 1938, J. Hermann.
- U. S. Congress, 85th Session: Public Law 85- 929. Food Additives Amendment to Food, Drug and Cosmetic Act of 1938, Sept. 6, 1958.
- Cramer, W.: “The origin of cancer in man,” J.A.M.A. 119:309-316, 1942.
- Davis, K. J., and Fitzhugh, O. G.: “Pathologic changes noted in rats fed D and C red No. 9 for two years,” Toxicol. & Appl. Pharmacol. 4: 200-205, 1962.
- Davis, K. J., and Fitzhugh, O. G.: “Tumorigenic potential of aldrin and dieldrin for mice,” Toxicol. & Appl. Pharmacol. 4:187-189, 1962.
- Day, P. L., Payne, L. D:, and Dinning, J. S.: “Procarcinogenic effect of vitamin B12 on p-dimethylaminoazobenzene fed rats,” Proc. Soc. Exper. Biol. & Med. 74:854-855, 1950.
- Dungal, N.: “The special problem of stomach cancer in Iceland,” J.A.M.A. 176:789-798, 1961.
- Engel, R. W., Copeland, D. H., and Salmon, W. D.: “Carcinogenic effects associated with diets deficient in choline and related nutrients,” Ann. New York Acad. Sc. 49:49-67, 1947.
- Esch, G. J. van, Genderen, H. Van, and Vink, H. H.: “The production of skin tumors in mice by oral treatment with urethane isopropyl-N Phenyl carbamate or isopropyl-N-chlorophenyl carbamate in combination with skin painting with croton oil and Tween 60,” Brit. J. Cancer, 12:355-362, 1958.
- Federal Radiation Council: Background material for the development of radiation protection standards, Report No. 2, September, 1961.
- Fitzhugh, O. G.: “A comparison of the chronic toxicities of synthetic sweetening agents,” J. Am. Pharm. A. 11:583-586, 1951.
- Fitzhugh, O. G., and Nelson, A. A.: “The chronic oral toxicity of DDT (2,2-bis(p-chlorophenyl-1,1,1-trichloroethane),” J. Pharmacol. & Exper. Therap. 89:18-29, 1947.
- Fitzhugh, O. G., and Nelson, A. A.: “Liver tumors in rats fed thiourea of thioacetamide,” Science, 108:626-628, 1948.
- Food and Agriculture Organization of the United Nations: Evaluation of the carcinogenic hazards of food additives, Fifth Report, No. 29, UN-FAO, Rome, 1961.
- Gilbert, C., and Gillman, J.: “Diet and disease in the Bantu,” Science, 99:398-399, 1944.
- Gorelova, N. D., and Dikun, P. P.: “Problems of oncology,” Vopr. onkol. 4:405, 1958.
- Halver, J. E., Johnson, C. L., and Ashley, L. M.: “Dietary carcinogens induce fish hepatoma,” Fed. Proc. 214390, 1962.
- Harinan, D.: “Prolongation of the normal life- ‘span and inhibition of spontaneous cancer by antioxidants,” J. Gerontol. 16:247-254, 1961.
- Homburger, F., Kelley, T., Friedler, G., and Russfield, A. B.: “Toxic and possible carcinogenic effects of 4-allyl-1,2-methylene-dioxybenzene (safrole) in rats on deficient diets,” Med. Exper. 4:1-1I, 1961.
- Hueper, W. C., and Payne, W. W.: “Observations on the occurrence of hepatomas in rainbow trout,” J. Nat. Cancer Inst. 27:1123-1135, 1961.
- Huseby, R. A., Ball, Z. B., and Visscher, M. B.: “Further observations on the influence of simple caloric restriction on mammary cancer incidence and related phenomena in C3H mice,” Cancer Res. 5:40-46, 1945.
- Jacobi, H. P., and Baumann, C. A.: “The effect of fat on tumor formation,” Am. J. Cancer, 39: 338-342, 1940.
- Jukes, T. H., and Shaffer, C. B.: “Antithyroid effects of aminotriazole,” Science, 132:296-297, 1960.
- Kensler, C. J., Sugiura, K., Young, N. F., Halter, C. R., and Rhoads, C. P.: “Partial protection of rats by riboflavin with casein against liver cancer caused by dimethylaminoazobenzene,” Science, 93:308-310, 1941.
- King, H. J., Spain, J. D., and Clayton, C. C.: “Dietary copper salts and azo dye carcinogenesis,” J. Nutrition, 63:301-309, 1957.
- Kline, B. E., Busch, H. P., Baumann, C. A., and Lavik, P. S.: “The effect of pyridoxine on tumor growth,” Cancer Res. 3:825-829, 1943.
- Korpassy, B.: “The hepatic carcinogenicity of tannic acid,” Cancer Res. 19:501-504, 1959.
- Kinosita, R.: “Special report. Studies on the carcinogenic chemical,” Tr. Soc. Path. Jap. 27: 665-725, 1937.
- Kraybill, H. F.: “The effect of ionizing radiation on the vitamins and other physiologically active compounds.” UN-FAO symposium on wholesomeness evaluation of irradiated foods, Brussels, Oct. 23 to 30, 1961. In press.
- Kraybill, H. F.: “General methods and problems in assessment of potential toxicity and carcinogenicity in radiation and thermal processed foods and food additives.” UN-FAO symposium on wholesomeness evaluation of irradiated foods, Brussels, Oct. 23 to 30, 1961. In press.
- Kraybill, H. F.: “Radiological hazards in processed foods resulting from nuclear warfare,” Food Technol. 16:3-16, 1961.
- Kraybill, H. F., Calvert, C., Decker, W. M., and Terrill, J. G.: “Radiological surveillance of total diets in selected locations in the United States,” Health Physics, 8:27-33, 1962.
- Lancaster, M. C., Jenkins, F. P., and Philip, J. M.: “Toxicity associated with certain samples of groundnuts,” Nature; London 192:1095- 1096, 1961.
- LaRoche, G., Halver, S. E., Johnson, C. L., and Ashley, L. M.: “Hepatoma inducing agents in trout diet,” Fed. Proc. 21:300, 1962.
- Lusky, L. M., and Nelson, A. A.: “Fibrosarcomas induced by multiple subcutaneous injections of carboxy-methylcellulose (C N1C), polyvinylpyrrolidone (PVR), and polyoxyethylene sorbitan monostearate (Tween 60),” Fed. Proc. 16:318, 1957.
- Matsuo, N. J.: “Nutritional effects of oxidized and thermally polymerized fish oils,” J. Jap. Biochem. Soc. 29:773-777, 1958.
- Miller, J. A., Miner, D. L., Rusch, H. P., and Baumann, C. A.: “Diet and hepatic tumor formation,” Cancer Res. 1:699-708, 1941.
- Miyake, M., Saito, M., Enomoto, M., Skikata, T., and Ishiko, T.: “Toxic liver injuries and liver cirrhosis induced in mice and rats through long term feeding with Penicillium islandicum Sopp growing rice,” Acta Path. Jap. 10:75-123, 1960.
- Morris, H. P.: “Effects on the genesis and growth of tumors associated with vitamin intake,” Ann. New York Acad. Sc. 49:119-140, 1947.
- National Research Council, Food Protection Committee: “Approximate total quantities of various substances necessary to produce tumors in animals,” Report 749; Washington, D. C., 1959, National Academy of Sciences-National Research Council, app. A, p. 37.
- National Research Council, Food Protection Committee, Food and Nutrition Board: “Problems in the evaluation of carcinogenic hazard from use of food additives,” Report 749, Washington, D. C., 195 9, National Academy of Sciences-National Research Council.
- Opie, E. L.: “The influence of diet on the production of hepatic tumors induced by p-dimethylaminoazobenzene,” in American Association for the Advancement of Science: Approaches to tumor chemotherapy, Lancaster, Pa., 1947, Science Press Printing Company, pp. 128-138.
- Oser, B. L., and Oser, M.: “2-(p-tert.-Butylphenoxy) isopropyl 2-chloroethyl sulfite (aramite),” Toxicol. & Appl. Pharmacol. 2:441-457, 1960.
- Oser, B. L., and Oser, M.: “Nutritional studies on rats on diets containing high levels of partial ester emulsifiers,” J. Nutrition, 61:235-252, 1957.
- Richardson, L. R., and Webb, B. D.: “Effect of a moldy diet on the growth and mortality of chicks and poults,” Texas Feed Service Report 23, pp. 3-4, March, 1962.
- Rosin, A., and Rachmilewitz, M.: “Development of malignant tumors of the face in rats after prolonged treatment with thiourea,” Cancer Res. 14:494-496, 1954.
- Roth, F.: “Arsen-Leber-Tumoren (Hämangioendotheliom),” Ztschr. Krebsforsch. 61:468- 503, 1957.
- Rous, P.: “The influence of diet on transplanted and spontaneous mouse tumors,” J. Exper. Med. 20:433-451, 1914.
- Rusch, H. P., Johnson, R. O., and Kline, B. E.: “The relationship of caloric intake and of blood sugar to sarcogenesis in mice,” Cancer Res. 5:705-712, 1945.
- Rusch, H. P., Kline, B. E., and Baumann, C. A.: “The influence of caloric restriction and of dietary fat on tumor formation with ultraviolet radiation,” Cancer Res. 5:431-435, 1945.
- Russell, W. O.: “The response of the central nervous system of the rat to methylcholanthrene. II. The effect of a diet deficient in thiamine and riboflavin on the induction of tumors derived from nervous tissue,” Cancer Res. 5:152-156, 1945.
- Sargeant, K., Sheridan, A., and O’Kelly, J.: “Toxicity associated with certain samples of groundnuts,” Nature, London 192:1096-1097, 1961.
- Sasaki, T., and Yoshida, T.: “Experimentalle Erzeugung des Leberkarzinoms durch Futterring mit O-Aminoazotoluol,” Virchows Arch. path. Anat. 295:175-200, 1935.
- Sobels, F. H.: “Peroxides and mutations. I. Implications of organic peroxides in radiobiology,” Argonne National Laboratory Symposium Abstract, May 7 to 9, 1962, pp’. 1.5-19.
- Sternberg, S. S., Popper, H., Oser, B. L., and Oser, M.: “Gallbladder and bile duct adenocarcinomas in dogs after long term feeding of aramite,” Cancer, 13:780-789, 1960.
- Steven, D. G.: “Food and beverages in relation to cancer,” in Symposium on potential cancer hazards from chemical additives to foodstuffs, International Union Against Cancer. Rome, Aug. 19 to 21, 1956, pp. 341356.
- Sugai, M., Witting, L. A., Tsuchivama, H., and Kummerow, F.: “The effect of heated fat on the carcinogenic activity of 2-acetylaminofluorene.” Cancer Res. 22:510-519, 1962.
- Suguira, K., and Benedict, S. R.: “The influence of insufficient diets upon tumor recurrence and growth in rats and mice,” J. Cancer Res. 10: 309-318, 1926.
- Szepsenwol, J.: “The carcinogenic effect of egg yolk and egg white in mice (abst.),” Fed. Proc. 21:161, 1962.
- Tannenbaum, A.: “Nutrition and cancer,” in Homburger, F., editor: The physiopathology of cancer, New York-, 1959, Paul B. Hoeber. Inc., pp. 517-562.
- Tannenbaum, A.: “The dependence of the genesis of induced skin tumors on the caloric intake during different stages of carcinogenesis,” Cancer Res. 4:673-677, 1944.
- Tannenbaum, A.: “The genesis and growth of tumors. III. Effects of a high fat diet,” Cancer Res. 2:468473, 1942.
- Tannenbaum, A.: “The role of nutrition in the origin and growth of tumors.” Approaches to tumor chemotherapy, Lancaster, Pa., 1947, Science Press Printing Company, pp. 96-127.
- Tannenbaum, A., and Silvertone, H.: “The genesis and growth of tumors. IV. Effects of varying the proportion of protein (casein) in the diet.” Cancer Res. 9:162-173, 1949.
- U. S. Food and Drug Administration: Refusal to extend effective date of statute for certain specified food additives (John L. Harvey). Fed. Reg. 25:12412, 1960.
- Widmark, E. M. P.: “Presence of cancer-producing substances in roasted foods,” Nature. London 143:984. 1939.
- Waterman, N.: Diet and cancer: An experimental study. Amsterdam. 1938. D. B. Centen.
- White, J., White, F. R., and Mider, G. B.: “Effects of diets deficient in certain amino acids on the induction of leukemia in DBA mice,” J. Nat. Cancer Inst. 7:199-202, 1947.