Access to all articles, new health classes, discounts in our store, and more!
Report of Laboratory Investigations on the Physical Properties of Root Filling Materials and the Efficiency of Root Fillings for Blocking Infection From Sterile Tooth Structures
Read before the National Dental Association, Chicago, Illinois, August 1918. Published in The Journal of the National Dental Association, Vol. V, No. 12, December 1918, pp. 1260-1280.
* * *
Root fillings are castings made within the pulp chambers of teeth. They must conform so exactly to the size and shape of that chamber that neither micro-organisms nor fluids, which may be external to the tooth, can enter or find lodgment. The form of this chamber is complex, uncertain and indeterminate and may vary thru a very wide range, which fact makes it necessary that the root filling material, or a sufficient part of it, be inserted in a moldable and plastic state. The physical properties of the materials used for this purpose are such as to make the task extremely difficult, even under ideal conditions of pulp chamber and readiness of access. The degree of exactness required is so great that few plastics can fulfill the requirements since they do not have the same dimensions after taking a condition of set that they have in a moldable state. The problem is rendered still more difficult by the fact that the operator must work in a restricted field, often of very difficult approach and requiring slender, hair-like instruments to be used in curved or tortuous channels, even having islands and bays, and often many estuaries, all of unknown form.
That “a new truth is a new sense” has been abundantly demonstrated in our profession by the changes in methods of practice that have followed the demonstration of the effects of oral focal infections on the general health of individuals. A similar change is in progress regarding methods of root sterilization and an equally marked change of methods must follow the establishment of the truth in the minds of the members of the dental profession, that the physical properties of the root filling materials which are in general use today are such that exceeding great skill and effort and favorable conditions are necessary to make a casting within the pulp chamber which has the fundamental qualities required of a root filling. We must acquire a root filling sense.
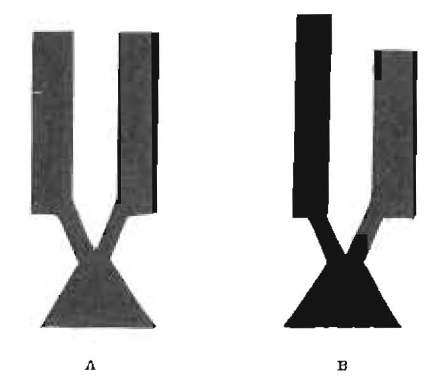
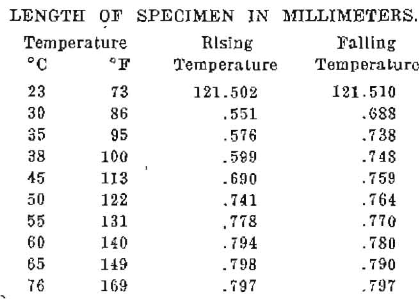
The materials that have been in most general use for root fillings are based upon adaptations of gutta-percha, the variations being chiefly in the solvents used to render it plastic or semi-plastic. We have not found in the literature, dental or otherwise, data on the involved physical properties of gutta-percha and the preparations of it as used in dentistry and it has been necessary for us to work these out in detail. We have had this work in progress for several years and have been fortunate in being able to engage the assistance of Dr. Dayton C. Miller, Professor of Physics, Case School of Applied Science, Cleveland, a physicist of international reputation. Gutta-percha is a gum and behaves similarly to the various waxes and gums. We have demonstrated, in previous reports on the physical properties of waxes,1,2 that the waxes and gums are likely to retain locked elasticity in the form in which we handle them; that, if two pieces of wax cut to the same dimensions have their elasticity released, they may change in relative length eighteen per cent, depending upon the direction in which the wax was in a condition of stress at the time it was chilled. This is illustrated in Figure 1 in which A represents two pieces of inlay wax which are identical in length and cross section. After they are warmed one has elongated and the other shortened, as shown in B. These are photographs from the actual specimens. The details will be found in the article referred to. We suspected that gutta-percha had corresponding physical properties, and one of our first undertakings was to establish the extent to which gutta-percha retains a locked elasticity. One piece of gutta-percha 70 x 70 mm, on being heated slowly, changed its dimension to 55 x 76.5 mm. On being raised to the temperature of boiling water the total area contracted 14.7%. The details will be found later in the more technical part of this report by Professor Miller. There was a progressive change in dimension beginning at about 40°C (104°F) and continuing to a temperature of 76°C (169°F). The length of the sheet decreased twenty-one per cent and the width increased twenty-nine per cent. In the second experiment, with another sample of base plate gutta-percha, the area and volume were determined and it was found that, when starting with a piece 70 x 70 mm, the length decreased to 63 mm and the breadth increased to 72.5 mm; the thickness increased from 0.843 to 0.912; the area decreased from 4900 sq. mm to 4567 sq. mm; the volume increased from 4131 cm to 4165 cm. The intervening dimensions are given later in the report. With this piece the length diminished ten per cent and the breadth increased three per cent. The linear expansion, due to heat, of a piece of heated gutta-percha from which the elasticity had been released was determined and it was found to change from 121.502 mm to 121.797 mm. on being increased in temperature from 23°C (73°F) to 76°C (169°F). But a very important condition is disclosed by comparing the dimension at various temperatures on cooling with the dimension at the same temperature when rising. There is a lag which indicates a different dimension for a given temperature in the cooling process and it probably takes a considerable period of time for this lag to entirely disappear. This corresponds with the hysteresis in a magnetic field. This is graphically shown in the curve in Figure 8 Table 3, of linear expansions which gives the detailed dimensions in the rising and falling temperatures. An additional observation made to determine the change in volume showed that from room temperature to the temperature of boiling water the pink gutta-percha increased in volume 2.4%. A piece of drab gutta-percha treated in the same way increased in volume 2.2%.
Figure 1–Two similar wax bars placed in a warm lavestment and cast. A, before heating. B, after heating.
The above determinations explain why fluids and infection always enter cavities around gutta-percha fillings, for every gutta-percha filling is from one to two per cent smaller after it has taken the body temperature and its final molecular state than at the time it was molded in the cavity. If we will think of a cavity with a dimension of 5 mm and a volume contraction of 1½%, which would occur if the gutta-percha were heated to about 75°C at the time of insertion, the crevice formed by the contraction would be sufficient to permit twenty-five bacteria, each having a diameter of one one-thousandth of a millimeter to enter abreast, and smaller and larger ones in proportion. Linear contraction is one-third volume contraction. For this reason even temporary gutta-percha fillings, if they are to protect dentin from irritation and infection, should always be inserted in cement. We have made determinations of the dimension change when heat is used as a means of making gutta-percha plastic, as follows: Glass cylinders were packed with gutta-percha, made plastic by heat and the gutta-percha submitted to high pressure while cooling. After cooling, ink was applied and it readily entered beside the gutta-percha, which it could not do while it was hot. Figure 2 is a photograph taken thru the glass tube, which has a diameter of about 5 mm. We cannot maintain pressure on the gutta-percha while cooling in a cavity as was done here and, notwithstanding this maintained pressure which kept the gutta-percha flowing as long as its resistance was less than the maintained pressure, the contraction was sufficient to let the ink penetrate freely around the gutta-percha as shown. The pressure used was approximately the limit of the glass tube and equalled several hundred pounds per square inch. These studies have been primarily to determine the actual and relative efficiency of root fillings made of gutta-percha. We have not included the cements in this series but expect to later.
Dr. A. E. Webster, Toronto,3 has studied the efficiency of cements and chloro-percha and gutta-percha and found them all defective. He did a splendid piece of work which has not had proper attention paid to it. The rendering of gutta-percha plastic by means of heat is not at all adequate as a means for molding it to pulp chambers, and the required plasticity is usually secured by means of solvents. For this purpose chloroform and oil of eucalyptol are chiefly used. Also chloroform in connection with rosin. Studies have been made to determine the physical properties of these solvents and of the combinations of gutta-percha with them. Chloro-percha has been used for many years as the plastic form of gutta-percha and the excess of shrinkage has been presumably controlled by the addition of cones of gutta-percha placed in the liquid. Altho exact observations had not, to our knowledge, been made, this material has been considered inefficient by many operators and a solution of gutta-percha in oil of eucalyptol was used by them in its stead, as advocated by Dr. John Buckley. Many dentists have been using a solution of chloroform carrying rosin, to act as a solvent for the gutta-percha points when introduced and as a sealing material for the dental tubuli and foramina, suggested by the late Dr. Callahan, who has contributed so much to the problem of root fillings. It must be remembered that, if the pulp chamber is filled partly by fluid and partly by solid, the change in dimension of both of these must be taken into consideration. Gutta-percha dissolved in chloroform, to a consistency of a thick cream or thick paint, has a volume 300% greater than that of the original material, or, to express it otherwise, if the pulp chamber is filled with gutta-percha having the consistency of a heavy paint, when chloroform has evaporated from it, the chamber will be one-third full. If we put sufficient gutta-percha into this paint or cream to occupy 90% of the total volume, the chamber will still lack 6.6% of being filled after the chloroform has disappeared.
Probably one of the greatest errors we have made, when thinking of gutta-percha in connection with a solvent like chloroform or eucalyptol, is the presumption that the softening of the surface of the gutta-percha cone with chloroform, in order to make it plastic and moldable to the form of the canal, does not materially change its dimension. The dimension is changed greatly with even a small amount of chloroform, as shown by these data. Gutta-percha dissolved in chloroform to form a paint has certain very definite qualities which we should study. It has adhesiveness while it contains an abundance of chloroform but loses this quality almost completely when the chloroform disappears from it. Consequently, root fillings made with this as a solvent tend to leave the walls and shrink toward a center within the mass, leaving the space caused by the shrinkage at the periphery of the mass. It was, doubtless, to overcome this quality that Dr. Callahan sought for and developed the rosin-chloroform preparation, and Dr. Buckley the eucapercha compound. Unfortunately the ultimate contraction is very great and corresponds relatively, whether the solvent used is chloroform, oil of eucalyptol, or chloroform carrying rosin. The gutta-percha in oil of eucalyptol (eucapercha), as supplied in the market, has a volume of 417% greater than the volume of the gutta-percha alone. In other words, a pulp chamber filled with eucapercha and submitted to long and complete drying would ultimately have 24% of the total volume of the pulp chamber filled. It solidifies into a lumpy and curdled mass and does not adhere to smooth surfaces. This extreme drying-out process, however, does not occur for a long period of time. After eight weeks of spontaneous air drying at room temperature, it reduces to 87% of the original. During a similar period gutta-percha and chloroform would have diminished to 76% of its original volume. It is only by the addition of artificial heat, which condition the body would furnish, that the rate is hastened. See tables. Moisture does not hasten it. Rosin and chloroform also undergo a very great reduction in volume when mixed to the consistency of a mobile fluid, approximately corresponding with sewing machine oil, as suggested by Dr. Callahan. It has a volume of 406% of the volume of rosin. In other words, if a pulp chamber were filled with a rosin and chloroform solution of the above consistency, it would ultimately only be 24.6% filled. The addition of gutta-percha cones to occupy say 90% of this total volume would still leave approximately 7.5% of the pulp chamber unfilled, when the complete drying out process has occurred. If a gutta-percha cone is placed in this liquid, it very rapidly swells so that it will occupy more space, soon expanding to ¼ more or to twice its normal volume, because of the softening process. The pulp chamber can only contain a given amount of material, whether fluid, semifluid or solid, and it is a physical impossibility to put in material to make up for the reduction in volume produced by the evaporation of the solvent, while the solvent is present. It is, therefore, a physical impossibility to completely fill a pulp chamber with gutta-percha made plastic by any of the above solvents, except at the time the gutta-percha contains the solvent. The chloroform and rosin compound has very distinct and favorable qualities in that it is very adherent and tenacious in all stages to both smooth and rough surfaces. It is unlike each of the preceding compounds in this respect. The result of this quality is that when a root filling is made of this material and gutta-percha cones, the contraction tends to express itself, in a large part, by a shrinking towards the walls if dry, rather than as a continuous disc at the side of the mass and between it and the wall, and herein lies the great virtue of this material as a means for filling pulp chambers.
Figure 2
It will be a matter of surprise, doubtless, to most of us to find that a skin is formed on the surface of each chloropercha, and rosin and chloroform, particularly the latter, which quite effectually delays the drying-out process. In a case of the chloroform and gutta-percha in a narrow chamber this skin or film separates from the vessel or container, leaving a fault which ultimately makes a series of caverns, each projecting from the last until the entire mass is honeycombed. In large chambers it contracts to a central free mass. Chloroform and rosin in a narrow chamber of the same form retains the continuity of the protecting surface skin by its adherence to the wall and underneath this film the liquid retains nearly its original fluidity, due to the control of the vapor tension by this membrane. Consequently, it is not only a matter of weeks but of many months before our root fillings inserted with any of these three compounds will have attained their maximum contraction. After air drying in an open vessel for two weeks rosin-chloroform had reduced to 65% of its original volume; in five weeks 59%; eight weeks 36.4% and the last 12% of the total shrinkage, requiring either a long period of time or the addition of artificial heat nearly to the boiling point of water for many hours. When we consider the great variety of forms of pulp canals and the variations in size, shape and direction of the many foramina, it is evident that either the root filling material must seal these foramina by a process of retreating while retaining a close adhesion to the surface of the foramen, instead of contracting to a common center of the mass, producing a fault or space. Different types of multiple foramina are shown in Figure 3 and Figure 4. In Figure 3 are seen two lateral canals each containing root filling material. Granting that all the tissue was removed from them, which is doubtful indeed, they would soon after root filling lack considerable of being completely filled, since the solution of gutta-percha which could be forced into them contained so much solvent, which would in time largely or entirely leave the mass. The space the solvent occupied would be open to periapical fluids either from the tissues or periapical space, unless a sealed joint is formed at the external opening of the canal. This sealing appears to form only on dry surfaces and only with the rosin chloroform of the above three methods. Figure 4 is a specimen made by Dr. Callahan and shows a large number of foramina. He found seventy five in one tooth and several in most teeth.
Figure 3
Figure 4
Root Filling Efficiency Tests
During an extended period we have also been making observations and determinations as to the practical results that can be obtained by the various root filling methods for comparison with the above. A large number of extracted teeth that had been root filled months or years previously by splendid operators, were opened after extraction, cultures were taken from the root fillings and dentin, and they were almost universally found to be infected. This, however, did not mean so much, since they were usually extracted because of known peridental involvement. We, accordingly, made a series of determinations of the efficiency of root filling materials when placed in sterile teeth to prevent the entrance into the pulp canal and dentin of infection placed externally. In this work we were assisted by Drs. Milton J. Damlos, on the staff at the Research Institute, and Thomas J. Hill, a practicing dentist, and finally by several dentists who kindly made operations for critical study. The results of these are included in the following tables:
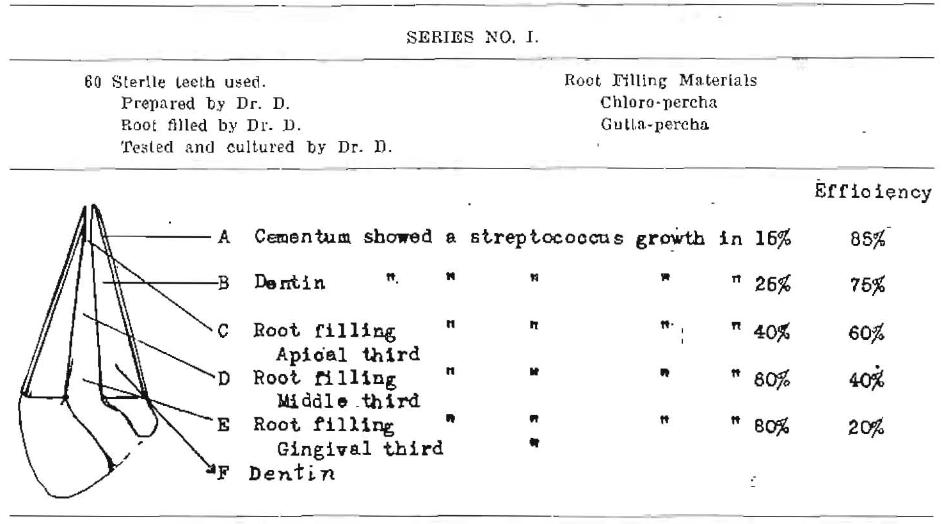
Series No. I
Note–Teeth were boiled two hours. Gutta-percha points were heated to 180°C for one hour. Chloropercha was heated In autoclave for thirty minutes at fifteen pounds. Root fillings were made under glass. Gingival end of canal was filled with gutta-percha. After root filling, teeth were placed in infected mixed cultures, frequently renewed and incubated for fifteen days at 37°C. Teeth were sterilized externally in full strength formalin and alcohol before opening. These results are immediate before contraction has had time to be completed.
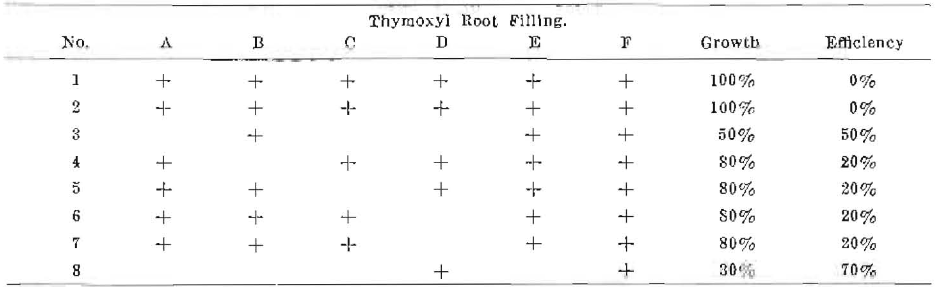
Series No. II
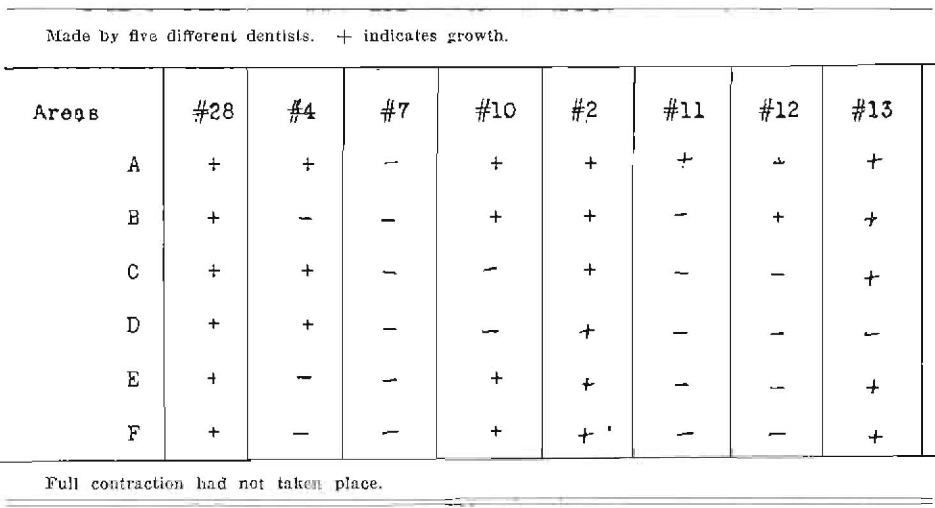
Note–These results are Immediate before complete contraction, due to evaporation of solvent. Efficiency would be less as time is extended. C, D and E refer to root filling infection.
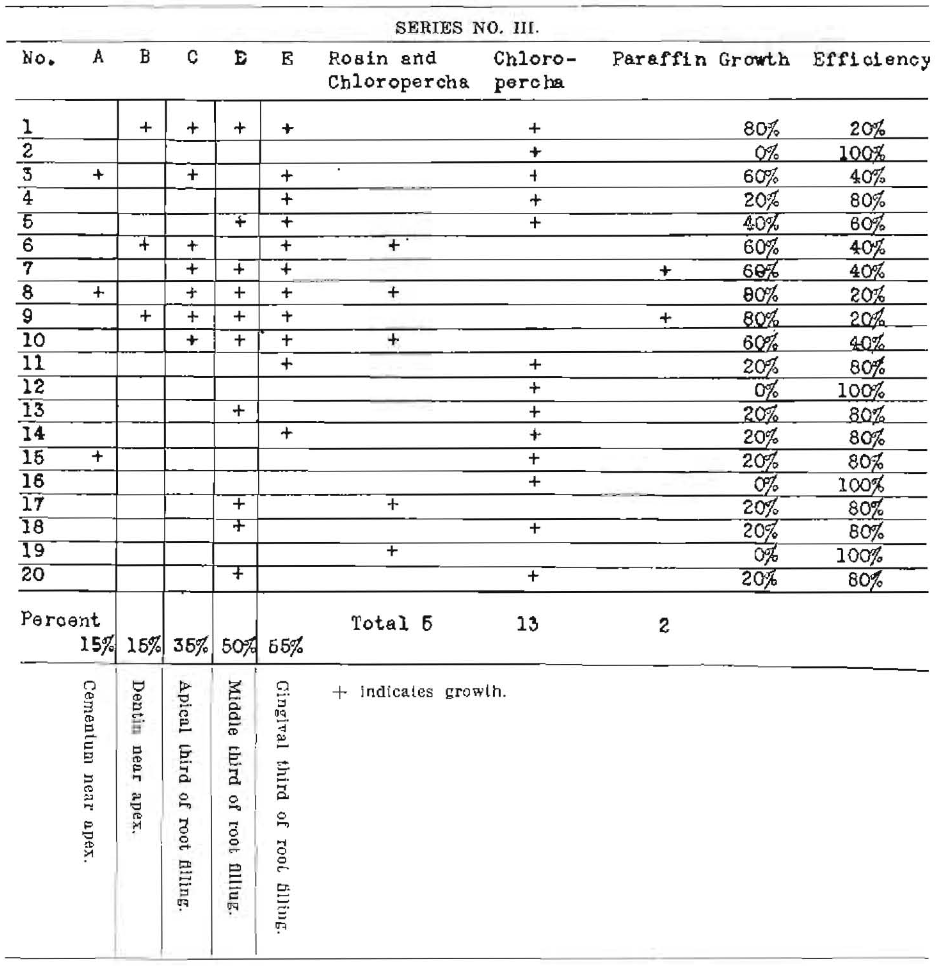
Series No. III
Note–Out of twenty teeth only four equaled 100% efficiency, or a net efficiency of twenty per cent. These results are immediate before full contraction of root fillings.
Series No. IV
Note–No tooth was free from infection. Net efficiency, 0.0%. These results are immediate before full contraction is possible. Crown ends of roots were covered with paraffin after root filling. + indicates growth.
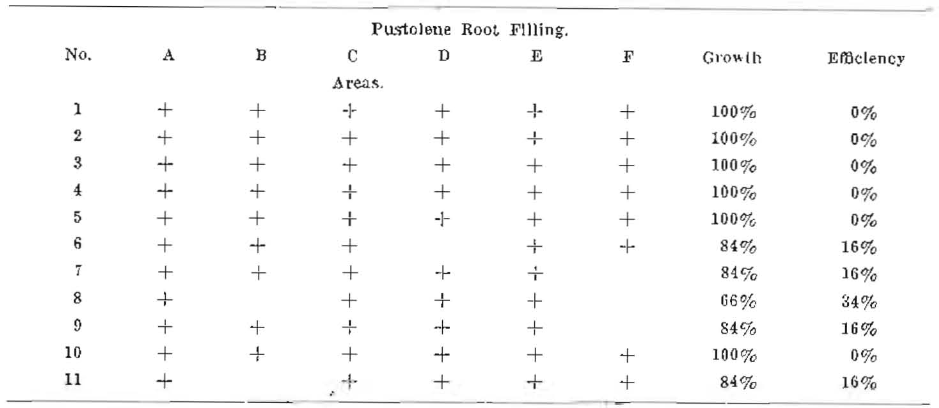
Series No. V
Note–Net efficiency, 0.0%. Full contraction of root fillings had not taken place. Crown ends of roots were covered with paraffin to protect the root fillings.
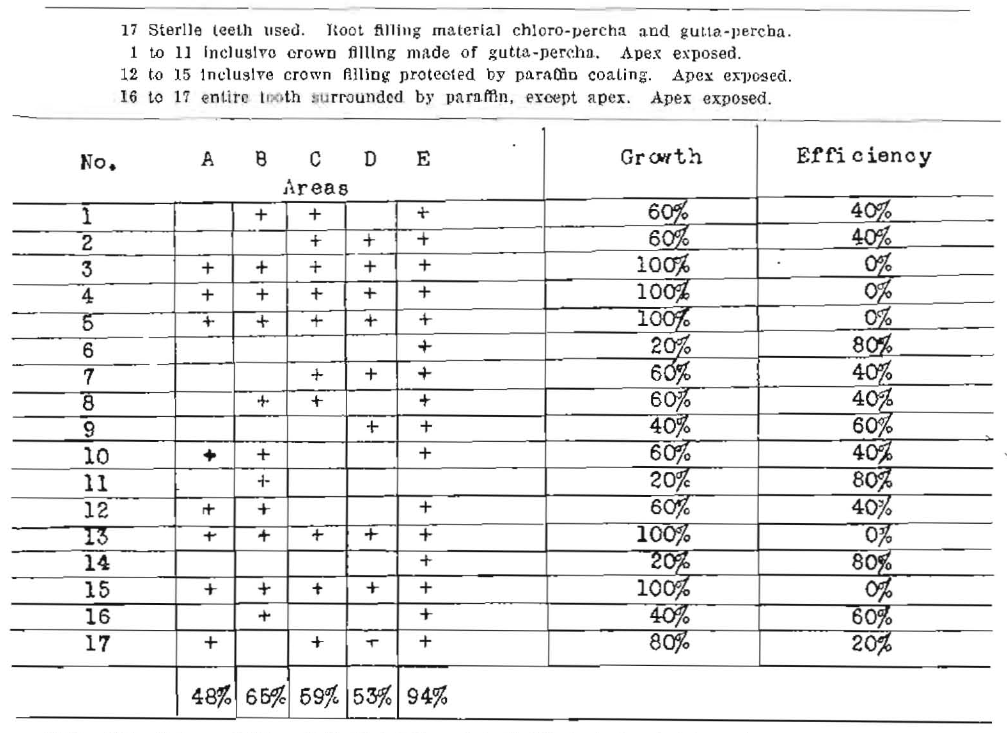
Series No. VI (+ = growth)
Note–Net efficiency 0.0%. Full contraction of root fillings had not taken place.
Series No. VII
In general the results are as follows: When teeth were sterilized by autoclaving and kept moist until root filled by the various methods, in the short space of time of even two weeks, the great majority were found to permit the entrance of organisms from the external surface into each pulp chamber, the root filling material and the dentin, notwithstanding the filling material and solvent used or its method of insertion. These results were obtained by placing the teeth, which had been root filled immediately or soon, into the infected culture medium for from a few days to two weeks and, accordingly, as bad as they are, these results certainly are much better than they would have been after the root fillings had been in place several months, which time is required for the complete contraction to take place. This suggests also that practically every tooth, the root filling of which is not protected from infection from the mouth by a competent barrier, is a direct port for infection to enter the body thru and past the normal defenses. There are several possibilities to be considered. Is it possible for the body always to withstand, without injury, the amount of infection that will enter the root when it is filled as well as possible? Is it possible to develop a technic that will effectually seal the bacteria from a large proportion of root filled teeth? Is there much hope of effectually filling the tortuous multi-canals of multi-rooted teeth in difficult and almost inaccessible parts of the mouth?
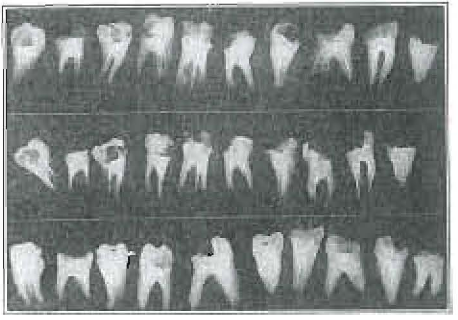
In Figure 5 we show a series of root fillings made by a splendid operator who said he thought he could fill 95% of multi-rooted teeth successfully. These operations, tho better than most operators can make, are practically all failures from the standpoint of mechanically blocking the canals.
Figure 5.
The teeth submitted to members of the profession for root filling were nearly all single rooted straight teeth, as shown in Figure 6, which also shows their root fillings.
Figure 6.
The physical changes which occur in the various uses of gutta-percha are so great and so important that every dental operator should study them carefully, and for this purpose we give in full Professor Miller’s detailed statements.
Department of Physics, Case School of Applied Science, Cleveland, Ohio.
Dayton C. Miller.
Charles D. Hodgman.
Robert E. Snyder.
Report by Dayton C. Miller on the Expansion of Sheet Gutta-percha, submitted by Dr. Weston A. Price.
The gutta-percha material is in the form of sheets about 150 x 75 x 0.85 mm. in size, of two colors, drab and pink. The purpose of the investigation is to determine the coefficient of thermal expansion.
I. Preliminary Volume Expansion.
Experiments were made to determine the volume expansion by placing the material in a pyknometer bottle and filling the bottle with water; the bottle was heated and the relative expansion was observed on a graduated glass tube. By computing the expansion of the glass bottle and of the water, the expansion due to the gutta-percha was determined. The same experiments were made by filling the bottle with mercury instead of water, but there was great difficulty due to the air enclosed in the capillary spaces where the mercury came in contact with the pieces of material. After much trouble these methods were abandoned, since they gave anomalous and irregular results.
II. Area.
Experiments were next made with a sheet of the pink material about 75 mm. square. Lines were marked on the surface forming a square 70 x 70 mm. This sheet was successively heated and cooled, and the dimensions of the square were measured.
The sheet contracted greatly in the direction which appeared to be that in which it was rolled in the process of manufacture, while it expanded laterally. The thickness was not measured in this experiment, but was measured in a repetition described later.
Table I gives the results of the measures:
Table I.
The results show that the material as received for the supply house is under great internal strain, some dimensions being too great, and some less than normal, and that the strains are progressively released by increasing temperature.
The first observation shows that at blood heat, 36°C, the strains are not at all released.
At 45°C. (113°F.) the material is slightly pliable, and the strains just begin to disappear.
At 56°C. (133°F.) the strains were relieved very considerably, so that at first the sheet was badly warped by unequal strain, and it was easily seen to shrink, with the unaided eye. The sheet shrank about 5 mm. in length, being 65 mm. long, and 72.5 mm. wide.
Measurements were made at 64°C. and then at 70°C. (159°F.) At the latter temperature the material was very pliable, but not sticky. It was found to have changed in dimension to 57.0 mm. x 75.5 mm.
At 76°C. (169°F.) the dimensions were 55.0 x 76.5 mm. The material became sticky and so soft as to take the impression of the finger tip. It seemed unsuitable for measures at higher temperatures.
However, it was heated to 78°C., 86°C., 91°C. and 99.5°C. (211°F.) the boiling point. The dimensions did not change, but remained 55.0 x 76.5 mm.
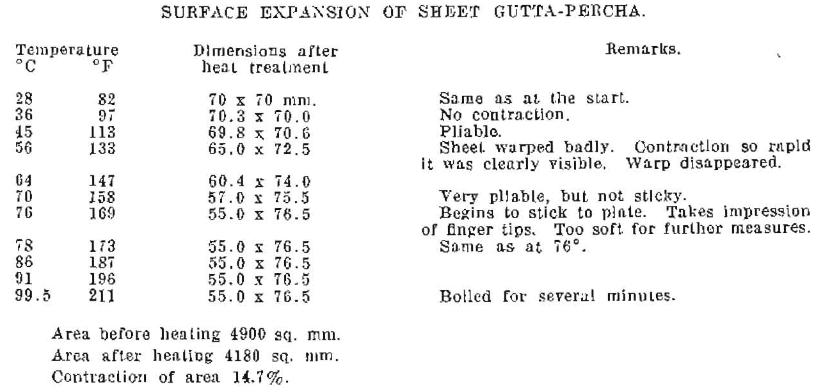
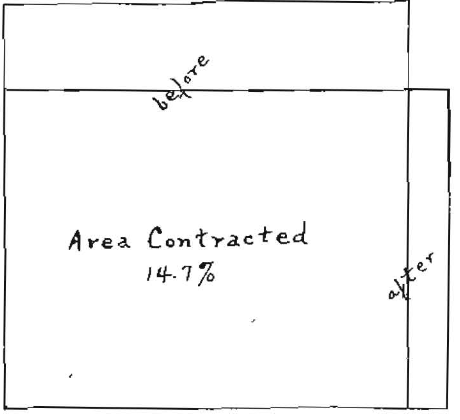
The experiments show a progressive change in dimension beginning at a temperature of about 40°C. (104°F.) and continuing to a temperature of 76°C. (169°F.) In this particular specimen of material, the length of the sheet diminished about 21%, while the width increased about 9%. The area of the sheet diminished 14.7%, due to shrinkage produced by heating.
The actual size of the piece of material at the beginning and at the end of the experiment is shown in Fig. 7.
Fig. 7. Shrinkage in area of a sheet of dental gutta-percha, due to heating to 76°C (169° F).
III. Area and Volume.
A second experiment was made with a sheet of pink gutta-percha, as before, using a marked square 70 x 70 mm. The thickness of the sheet was carefully measured with calipers at eight places, and the average thickness is used in calculating volume. The measurements are shown in Table II.
Table II.
These experiments lead to the same general conclusions as before, tho the changes in dimension for this specimen are less than for the first. The length diminished about 10%, while the breadth increased about 3%. The changes had taken place before the temperature of 85°C was reached. Continued boiling in water for fifteen minutes did not produce any further change.
Taken in connection with the measure of thickness, the observations show that tho the area diminished very much, the volume does not vary appreciably. There is an apparent permanent increase in volume of less than 1%, but this may be due to the fact that the heat slightly roughened the surface, making the thickness appear too great.
IV. Linear Expansion.
A piece of the drab colored gutta-percha was subjected to temperature treatment by heating and cooling several times to 55°C. and 75°C. and finally by boiling in water for fifteen minutes, and then it was cooled slowly. Fine marks were ruled near the ends of the sheet, 121.5 mm. apart.
This strip of material was placed on a special warming plate with arrangements for changing and controlling the temperature by circulating hot water.
The apparatus was placed on the Micrometer Microscope Comparator.
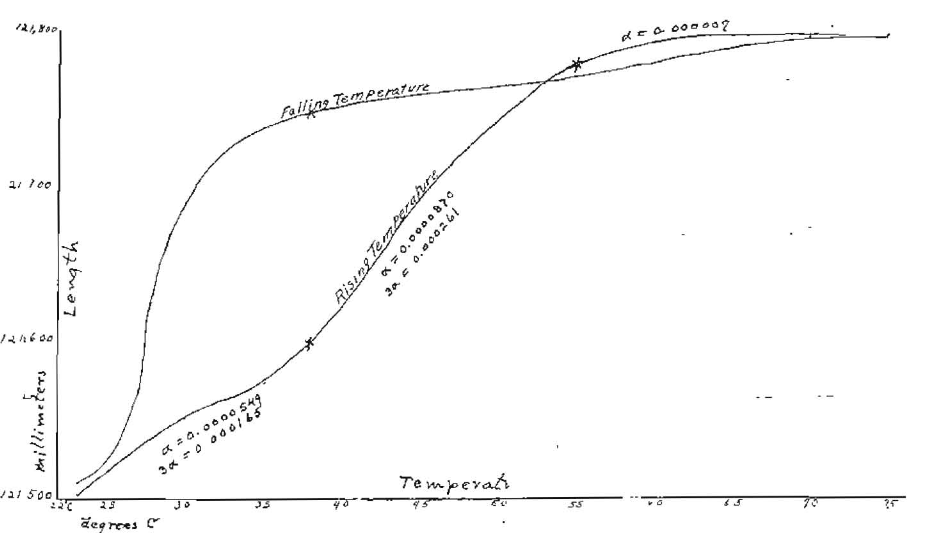
The expansion of the gutta-percha was directly measured with the micrometer microscope, with as great precision as possible. The observations were taken twice around a cycle of rising and falling temperatures. The average results of the measures are shown numerically in Table III and graphically in Fig. 8.
Table III.
Fig. 8
The coefficient of linear expansion and of volume expansion (equal to three times the linear expansion), computed in accordance with the data plotted in Fig. 8, are given in Table IV.
Table IV.
The observations show that for rising temperatures the gutta-percha has a coefficient of expansion of 0.0000549 up to about 38° C. (100°F.) and a coefficient of 0.0000807 from 38°C. to about 55°C. (131°F); above 55°C. the expansion is very slow and practically vanishes at 75°C. (167°F). This agrees exactly with the measures made on the area of the sheet material as shown in Table I; the material becomes wholly plastic at this temperature. The curve for falling temperatures shows a very decided lag in the contraction at the beginning. This may be due to very poor heat conductivity, which causes the temperature to lag, and it might disappear if the material remained a longer time at each temperature. Common experience with this material shows that after being softened by heat and then being apparently cooled to room temperature, its normal rigidity returns slowly.
V. Remarks.
Before being used, this material should be “annealed” by being heated to a temperature of 75°C. (167°F.) Otherwise a long slender cone of the material might be placed in a cavity, and there might later be a contraction in length of 15% which would pull the cone out of the cavity.
If the upper end of the cone were softened with a hot tool, this end would probably be safely scaled.
If a cone of this material were “annealed” and then placed in a cavity at a temperature of 55°C. (131°F.) and then cooled to a temperature of 20°C., there would be a lateral contraction of 0.007 mm. (0.0003 inch) in a cone 3 mim. (about 8 inch) in diameter.
The results of the area measurements, Table I, agree remarkably with the linear expansion observations, Table III, and interpreted in Fig. 8. The material acts like a normal solid up to a temperature of 38°C., expanding at a uniform rate and without the peculiar contraction shown at higher temperatures. At 38°C., it may be that some ingredient of the composition begins to melt, causing a release of any existing internal strain and producing a large contraction in the unannealed and stretched material which wholly masks the regular thermal expansion. After annealing, however, the regular expansion occurs in increased amount from 38° to 55°. Above the latter temperature the material is in no sense a solid.
Respectfully submitted,
(Signed) Dayton C. Miller, Professor of Physics. Cleveland, Ohio, July 8, 1916.
VI. Additional Expansion Experiments.
A piece of pink gutta-percha was “annealed” in boiling water for ten minutes with the following results:
The length diminished…….18.0%
The width increased.…….7.7%
The thickness increased. …….15.9%
The area diminished.…….11.7%
The volume increased..…….2.4%
A piece of drab gutta-percha “annealed” in boiling water for fifteen minutes, was changed as follows:
The length diminished…….17.4%
The width increased…….7.6%
The thickness increased…….14.8%
The area diminished…….11.2%
The volume increased…….2.2%
A piece of the latter material was measured as accurately as possible, to determine its volume, which was 4.764 cubic centimeters. The volume was then determined with precision by hydrostatic weighing, giving the results 4.760 cubic centimeters.
This result indicates that the measures so far reported have a precision of about one part in a thousand.
This material was then treated with chloroform, for determining the changes in volume; but the behavior of the solvent was such that it was necessary to proceed by a new method, described in the next section.
VII. Effects of Chloroform on Gutta-Percha.
It was expected that chloroform might be slowly absorbed by the gutta-percha, but the first touch of the liquid produced a true solution, which proceeded very rapidly, the result being a kind of “rubber paint.” Any portion of the original material not yet dissolved retained its original elastic properties.
The material was wholly dissolved, and the solvent was allowed to evaporate slowly until the solution just began to set, and was just firm enough to hold its shape but could be easily indented. The volume of this thick solution was 11.085 cubic centimeters, by hydrostatic weighing.
This amount of material was then allowed to dry thoroughly, the solvent being wholly evaporated. The volume was now 4.267 cubic centimeters.
There is thus an expansion of volume in the gutta-percha from a solid to the condition of a solution which is just firm enough to hold its shape, of about 260%.
After being dissolved in chloroform and then being dried till the solvent has all evaporated, the gutta-percha returns practically to its original elastic condition. In its original condition the gutta-percha is elastic and tenacious; the material dissolved and dried is inelastic and breaks very easily.
Various Coefficients of Linear Expansion.
Department of Physics, Case School of Applied Science. Cleveland, Ohio.
Dayton C. Miller, Paul E. Sabine, Charles D. Hodgman, George B. Obear.
Report by Dayton C. Miller on Some Physical Properties of Dental Root-Filling Materials.
1. General Properties.
Investigations were undertaken to determine the volume, density, contraction, hardness, and general consistency of several materials used by the dental profession for filling root cavities. The three materials tested were submitted by Dr. Weston A. Price and are described as follows:
- Rosin dissolved in chloroform.
- Gutta-percha dissolved in chloroform and eucalyptol oil, a proprietary preparation known as “Eucapercha.”
- Gutta-percha dissolved in chloroform.
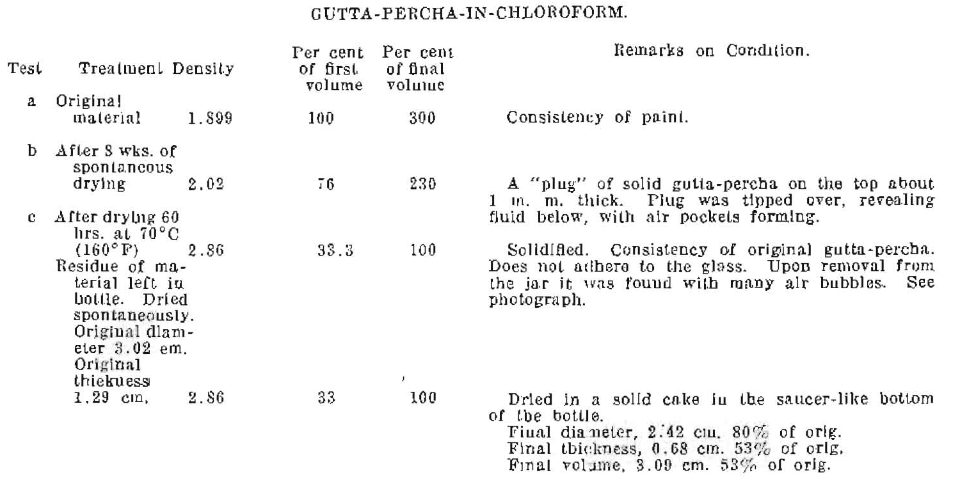
The first efforts were made to determine the rate of contraction, the density, and the firmness of the material, from its fluid condition as put into a tooth cavity, to the condition of being solid. To secure accuracy, and at the same time to approximate the form of a root filling, the materials were placed in cylindrical glass graduates each of a normal capacity of 10 cubic centimeters, but actually holding about 13 cubic centimeters. Each jar was provided with a glass cover having a ground joint, and was accurately calibrated. Fig. 9, shows the three jars, containing the materials, after some evaporation had taken place as described later.
Fig. 9. Jars Containing Root-Filling Materials.
After careful weighing, the covers from the glasses were removed and the solvents were allowed to evaporate at room temperature, standing in the sunlight when practicable (in May and June). It was thought that the materials would shrink steadily in volume, and become solid in a few days’ time; and by making weighings, volume tests and hardness tests at successive stages, the information sought could be secured. This expectation was not realized, for the evaporation proceeded very slowly, and for the gutta-percha materials in a very non-uniform manner.
After eight weeks of spontaneous evaporation in the air at room temperature, the jars remaining uncovered during the whole time, the conditions were as shown in Fig. 9.
The rosin-in-chloroform had steadily contracted in volume, but very slowly in the last three weeks. It was reduced to about one-third of the original volume, the surface being at the level indicated by the arrow at the side of the jar, the measured volume being 36% of the original. The inner surface of the jar above the mass of rosin was covered with a firm, adherent coating of rosin, which shows in the photograph as a discoloration of the jar above the arrow. The surface of the rosin was hard, but upon examination with a hard instrument, it was shown to be only a thin shell of hard rosin, not more than a half millimeter thick, while below this the material was still in a very fluid condition. The consistency of this fluid was that of a thin syrup, such as common maple syrup.
The Eucapercha had contracted much less, to about 86% of its original volume, indicated by the arrow. For about 12 millimeters near the top, the substance had assumed the consistency of dry chalk, a fine granular condition, and it had dried with the formation of cracks, like clay. Below this chalky deposit the material was in its original fluid condition with no evident thickening.
The gutta-percha-in-chloroform had contracted to about 75% of its original volume. The upper surface was hard gutta-percha. Upon touching this surface, it tipped over revealing the formation of a “plug” about 2 millimeters thick, with large irregular air spaces beneath. The material was, however, very fluid in the interior of the portions which appeared to be solid, and in the bottom of the jar it was as fluid as at first.
These results indicated that the necessity of forced evaporation, and the jars were placed in a water bath and subjected for various times to temperature of about 70°C. (169°F.) and 96°C (205°F.)
The conditions of these tests are best described for each material separately.
2. Rosin-In-Chloroform.
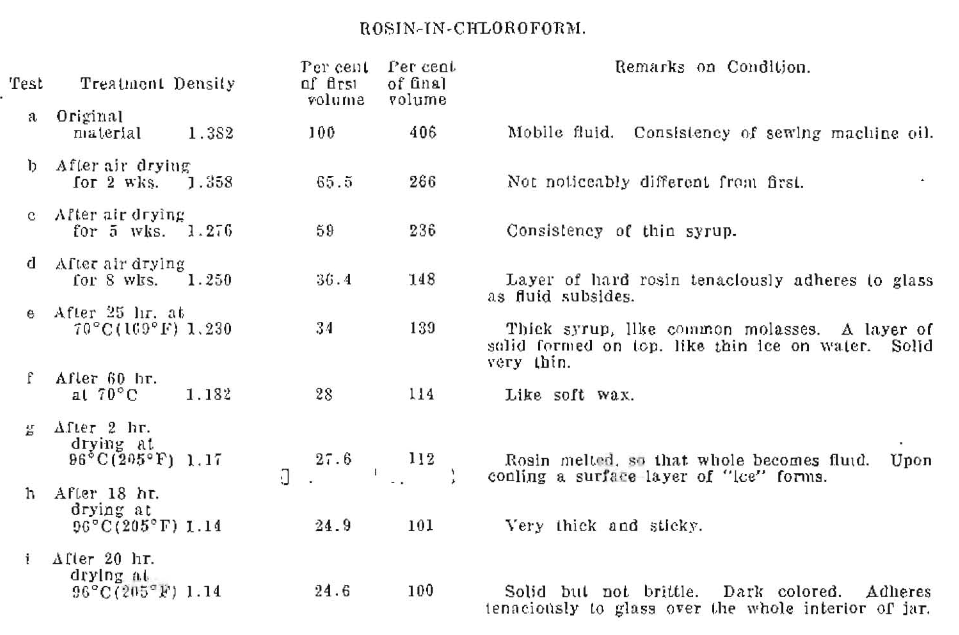
The rosin-in-chloroform at the beginning of the experiments was a mobile fluid of the consistency of sewing machine oil, having a density of 1.382. After two weeks of spontaneous drying its volume was reduced by about one-third, and the density was 1.358. The conditions of the material at the various stages of the experiments are shown in tabular form, Table V.
Table V. Rosin-in-Chloroform.
As the evaporation proceeded, the material left a tenaciously adhering coating on the walls of the jar, which shows in the photographs, Figs. 9 and 10, as a cloudy appearance of the glass jar above the line of the solid portion. It forms a coat of rosin-paint on the inside of the cavity which seems to be very permanent and which may be of great importance in dental uses. The evaporation to the solid state required long heating at temperatures much above body-temperatures as shown by the Table. The final volume was 24.6% of the initial, as shown by the arrow in Fig. 10, and the final density is 1.14. The density of purified “violin bow rosin” was determined experimentally to be 1.07. This indicated the presence of some of the solvent even at the end of the experiment; or perhaps, the continued heat treatment drove off some of the natural, lighter, components of the rosin.
Fig. 10. Rosin-in-Chloroform. Evaporated in jar.
Experiments have been made to determine whether the rosin will harden more rapidly in water, on the possible supposition that water dissolved solvent.
The results show that the water retards hardening, by preventing the evaporation of the solvent, and that its absorbing effect, if any, is imperceptible.
3. Eucapercha.
The eucapercha material used was in the original package, which had not been opened. The glass jar was filled with the material, there being 12.75 cubic centimeters at the beginning, the density being 1.432. The results of the experiments at the four principal stages are shown in Table VI. The appearance after 8 weeks of spontaneous drying is shown by the middle jar in Fig. 9, and the material fully dried is shown in Fig. 11.
Table VI. Eucapercha.
Fig. 11. Eucapercha. Evaporated.
The peculiarities of this material showed in a tendency to curdle, or become granular (soft granules), and in drying on the surface to separate, forming drying cracks. The solvent seems to have destroyed to some extent the rubbery texture of the gutta-percha. This remains in the material when fully dried; it is much less tenacious than the gutta-percha treated with chloroform.
The material, in drying, contracts in about the same proportion as does the rosin, but it adheres very lightly, or not at all, to the walls of the glass jar. When completely dried it is easily removed from the glass, the latter being left clean. This is in marked contrast to the rosin.
4. Gutta-Percha-in-Chloroform.
The gutta-percha was dissolved in chloroform, producing a white paint, similar in appearance to the Eucapercha. The conditions of the material at the various stages are given in Table VII, and the final appearance is shown in Fig. 12.
Table VII. Gutta-Percha-in-Chloroform.
Fig. 12. Gutta-Percha-in-Chloroform. Evaporated.
This material dried by forming a hard surface layer, like ice, which even when thin, would almost wholly prevent further evaporation of the solvent. This action would be complete except for the contraction of the partly solidified material, which pulls away from the glass, and allows further evaporation thru the opening. This further evaporation takes place in pockets, producing a porous or cellular solid. This is clearly shown in the picture of the final condition, Fig. 12.
When the material dries in the bottle, it shrinks away from the edges and rounded corners, and does not form the inner pockets.
The peculiarities of this material are that it remains tenacious and rubber-like even tho it becomes filled with air bubbles; it does not become granular; and it does not adhere to the glass jar.
5. General Remarks.
The first noticeable fact in the experiments was the extreme slowness with which evaporation proceeded under spontaneous air drying. It would have required a year, or more, for the materials in the jar to have solidified. Even when heat of 70°C. (169°F.) was applied, the process was not much accelerated. A temperature of boiling water was required, and this was sufficient to melt the rosin and to soften the solid gutta-percha. It is not improbable that the rate of drying in a tooth-cavity, the walls of which are more or less porous, would be different from that in a glass jar.
It is possible that the body-fluids may affect the rate; but experiments show that the placing of the material in water does not assist hardening, but rather prevents it.
A conspicuous physical property of the materials is the great contraction in volume, the final volume in one instance being less than a fourth of the original volume.
Both of the gutta-percha materials not only contract in volume, but in doing so they fail to adhere to the surface of the jar, leaving it clear of protecting covering, and the materials become porous or granular in structure. It would seem that these properties would render the materials useless for the filling of root cavities. As an illustration it may be assumed that root canals have diameters varying from 0.001 inch to 0.015 inch that is from 0.025 millimeters to 0.375 millimeters, and that bacteria may have a size varying from 0.0005 mm. to 0.005 mm. The possible contraction of fluid gutta-percha root-filling material in the smallest root canal may be 0.005 mm. while for a large canal it may be over 0.075 mm.; these contractions are much larger than the sizes of bacteria. While these statements of the contractions are not exact because of the uncertainties in the manner of drying, yet it is certain that there will be little protection against the entrance of bacteria.
The rosin solution when drying differs from the other in that a solid coating remains tenaciously adherent to the whole inner surface from which the fluid has receded, and the solid does not at any time part from the walls. Thus the whole inner surface of the cavity remains protected, and no cracks are left where bacteria may enter thru the small canal. There is an inner hollow space, but it is wholly surrounded by the painted walls, and closed at the bottom by the solid.
These conclusions, however, are not final in regard to the filling of teeth, for the conditions in a living tooth are certainly different from those in the glass jar. But the facts of physical contractions, etc., are exactly the same in the two cases.
Note:
Density of violin-bow-rosin…..1.07
Density of Chloroform…..1.48
Density of Ether…..0.72
Density of Gutta-percha…..2.90
Respectfully submitted,
(Signed.) Dayton C. Miller, Professor of Physics. Cleveland, Ohio, August 3, 1918.
It will be seen from the above detailed physical report by Dr. Miller, that we are using materials which do not have the properties that our needs require. While contraction begins at once, it will continue slowly for months and possibly for years. This probably explains why so many root filled teeth which at first seem normal fail months or years after root filling. Exhaustive researches should be made to develop adequate root filling materials.
The human body is an hermetically sealed container, the alimentary tract being an infolded tube and continuous with the exterior. Pulpless teeth are openings thru nature’s protective armor and as such will be the port of entrance of infection into the body, unless they are hermetically sealed. These studies show that it is extremely difficult and in many teeth impossible to accomplish this with the methods and materials in vogue. Until we can do very much better than this report shows, no dentist is justified in devitalizing a healthy pulp except in case of most extreme necessity, and seldom, if ever, for ordinary bridge work. Every non-vital tooth, whether root filled or not, is, by the above, to be considered under suspicion and must be kept under observation. An apparently tolerant nature has been entitled to much of the credit for the seeming success of dental root fillings. We have still to learn when nature is only apparently tolerant.
References Cited:
- Price, “The Laws Determining the Behavior of Gold in Fusing and Casting.” Dental Cosmos, March, 1911.
- Price, “Special Researches in Physics.” Journal of the National Dental Association, October, 1914, page 110 (Vol 1. No. 4).
- Webster, “Temporary and Permanent Fillings as Barriers to Bacteria.” Dental Review, April, 1903.