Access to all articles, new health classes, discounts in our store, and more!
Nutritional Role of Calcium in Plants: Prominent in the Non Legume Crops Sugar Beets
Published in Plant and Soil, Vol. 33, No. 2, October 1970
* * *
Summary
Studies, using the sugar beet as a non-leguminous crop in the Imperial and San Joaquin Valleys of California, have shown by soil tests, measured crop yields, and the storage of the disaccharide sugar in the root, that calcium is a plant nutrient deficient in these desert soils.
It was demonstrated by trials over three years that the drilling in seed contact of a natural gypsiferous mineral, known in commerce as “mineralag”, gave decided improvement in the crops as increasing stands and root yields on the neutral and alkaline soils under cooperative supervision of the Holly Sugar Corporation of tests by Geyser Minerals Corp., Denver, Colorado.
It was established that these desert soils with organic matter contents of barely one per cent, or less, a pH of 8.0 and higher, and of excesses of other cations above standard percentages saturation of exchange capacities, the slight variations in applications of nitrogenous, phosphatic and mixed fertilizers in conjunction with calcium, can represent imbalanced plant nutrition as shown by reduced concentrations of the disaccharide sugar in the beet root. The absence, in near total, of both the soil organic matter and the hydrogen cation, or acidity, show clearly how badly unbuffered these soils are to be ‘physiologically shocked’ by more ‘salt’ applications in fertilizers.
Accordingly, when so poorly buffered, the excesses of the other cations, namely, magnesium (possibly the most disturbing), potassium and sodium beyond adsorption potentials to be active as salts, are serious disturbers to any attempt to balance plant nutrition for higher, or controlled sugar concentrations, along with higher beet yields.
These studies recognize the need to consider the balance of the cationic fertility as a factor pointing to the concentration of the disaccharide sugar as well as to the tonnage yields of roots. These studies point to the need to consider the quality, first, along with the quantity of the crop as guides for wisest soil management.
Introduction
Liming the soil for improved agricultural production of crops, especially the legumes, was a practice, even with the Romans, before the Christian Era. The belief, that liming was beneficial for legumes because it counteracted an acid condition of the soil, was introduced as late as the first or second decade of the twentieth century.12 That became a more firmly established idea when the chemists told us about concentrations of the hydrogen ions, measured more accurately by the delicate dye indicators and, soon thereafter, by the hydrogen (glass) electrode, and specially designed machines with dial indicators reporting hydrogen ion concentration (activities) on a logarithmic scale.
But with more knowledge through research about plant nutrition, it soon seemed more logical to consider that in case of improved legume crop growth from liming the soil, it is the nutrient calcium, not the alkalinity,5 that is operating as the cause. Such a view emphasizes the properly balanced nutrition of the plant, especially as concerns the four essential cationic nutrients, calcium, magnesium, potassium and sodium, as soil-borne factors in healthy plant growth and survival, more than the reduction, of the ionic, acidic hydrogen of the soil as the environment of the plant roots, later discovered as a beneficial one.6
One would be expected to envision nutrition certainly more essential, and acidity not so damaging when the plant’s uptake of nutrient cations occurs because its root environment, or rhizosphere, is made acid by its own respired waste, namely, carbon dioxide, which, in water, makes the acid encircling the root with ionic hydrogen actively exchanging itself to the surrounding colloidal clay-humus, which offers adsorbed or available cations like calcium, magnesium, potassium, sodium, and others, to be exchanged for it (another cation) to the root as nutrition and growth.6 When acidity of the soil has been resulting in plant nourishment during all the past ages of the plant’s evolution, one can scarcely indict soil acidity as damaging, except as it represents nutrient exhaustion, and thereby plant starvation–an inimical soil environment, a priori, for healthy plant growth.
This belief in supposed damage to legume crops by soil acidity was emphasized in connection with their production of nitrogen-fixing root nodules in which the Rhizobium microbes enabled those plants, on soils of higher saturation by exchangeable calcium, to elaborate atmospheric nitrogen into the plant proteins required for good healthy growth. The accumulated studies concerning calcium required as such nutrition of legume plants for their symbiotic soil microbes,9 have now come to raise the question whether the soil-borne, nutrient cation, calcium, in balance with other cations does not play a similar and highly beneficial, physiological role in non-legumes. Some studies during the three years, 1965-1967 inclusive, with sugar beets in the desert soils of the Imperial and San Joaquin Valleys of California have suggested the validity of such a theoretical consideration.
Historical and Previous Research
Research studies about legume inoculation to determine how early in the life of the soybean it becomes nodulated under the influence of various cationic nutrients in quartz sand, demonstrated calcium much more stimulative than either magnesium or potassium in early germination and better growth of the plants.4
In another study, soybean seeds were started in two sets of watered quartz sand as growth medium to one of which pulverized, calcareous limestone had been added, and to the other only water. Then ten days after planting, the two sets of plants were separately transplanted into the same kind of soil, with low saturation by calcium of its exchange capacity and allowed to grow there for sixty days. The plants given the calcium through early root contact with carbonate of calcium; (a) had carried along 30.14 mg of calcium per hundred seedlings, the smaller plants given no calcium at the planting carried but 17.07 mg; (b) had nodule numbers ranging from 36.6 to 38.9 while those without initially applied calcium had 3.4 to 15.0; (c) had heights of plants of 12 cm in contrast to the other set but 7.0 cm. in height; all as facts testifying to the effectiveness of early uptake of calcium in establishing the young plants for better growth and survival.1
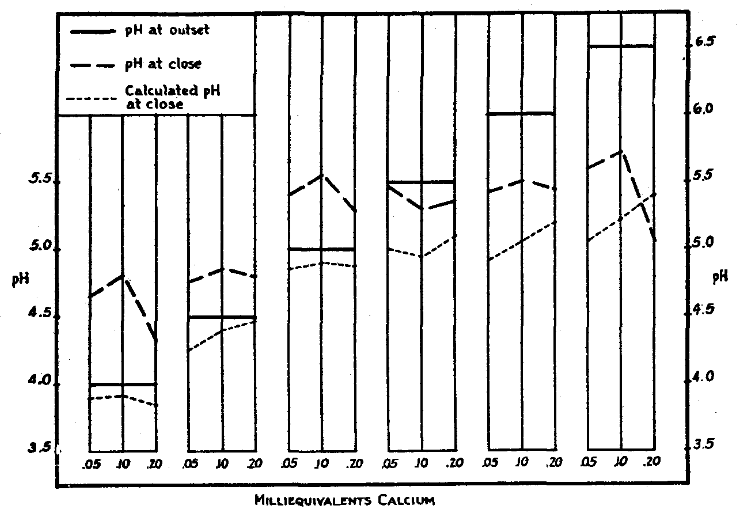
The importance of the higher degree of saturation of the soil’s capacity for colloidal adsorption of the essential cations and their later entry into the plant root as balanced plant nutrition, emphasized itself in a study of the inoculation of soybeans as seemingly related to the degree of soil acidity.2 Germinated seeds were placed in quartz sand as skeleton medium carrying electrodialyzed acid, colloidal clay with an initial pH of 3.35 and titrated with calcium hydroxide to provide calcium-hydrogen clays of the chosen degrees of acidity; and reciprocally degrees of saturation by calcium; namely, a pH (water) series of 4.0, 4.5, 5.0, 5.5, 6.0, and 6.5. These clays were then taken in such amounts as needed to supply a specified, constant amount of calcium per plant in each of the above six clays of degrees of acidity, or controlled pH. There were three duplicate pH, or varied acidity, series of them. One of the three series had 0.05 me of total exchangeable calcium per plant; another series had 0.10 me; and the third one had 0.20 me of calcium per plant. In the three series of the increasing pH values, each of the six represented increasing degrees of saturation of the clay’s exchange capacity, but each series had a constant total quantity of exchangeable calcium offered each plant. These three represented roughly ‘low’, ‘medium’, and ‘high’ levels of total calcium through a series of calcium saturations of the soil’s adsorption capacity of 17, 31, 45, 59, 73, and 87 per cent, accompanied by the reciprocal percentage (on basis of 100) concentration by the hydrogen cation.
Nodule production, as index of healthy plant nutrition, was limited to the two highest pH values and also to the higher values of saturation by calcium in the ‘low’ total calcium; and to the three highest pH values as the two higher total calcium values offered to the roots. Each pH series, from the most to the least acid soil, was an exhibition of increased or improved crop growth as pH values (increase percentages saturation) went higher, or degrees of acidity became less. (See Table A).
Table A: Nodulation and growth of soybeans (first crop) as influenced by the calcium and by the pH of calcium-clay soils. From Ref.2
| Plant characters | Calcium per plant me/plant | pH at outset (first crop) | |||||
| 4.0 | 5.5 | 5.0 | 5.5 | 6.0 | 6.5 | ||
| Nodules 50 plants | 0.05
0.10 0.20 |
0
0 0 |
0
0 0 |
0
0 0 |
0
8 60 |
7
28 69 |
14
40 127 |
| Height, cm | 0.05
0.10 0.20 |
11
9.5 8 |
26
27 25 |
28
34 40 |
31
42 45 |
36
44 48 |
36
45 52 |
| Weight of 50 plants in grams | Tops
0.05 0.10 0.20
Roots 0.05 0.10 0.20 |
4.8 4.2 4.6
1.5 1.7 1.0 |
6.3 6.3 6.0
2.5 2.2 1.7 |
6.8 7.3 8.7
2.0 2.1 2.5 |
7.0
8.9 9.2
2.0 4.3 |
7.9
9.5 9.4
4.0 4.3 |
7.6
8.7 9.9
3.6 4.2 |
But, more significant was the fact that of the lower pH values the plant roots still managed to take some of the calcium from the clay which should have been made more acidic thereby. But yet, contrary to these expectations, the clays had been raised in their pH values, indicating cations moving from the growing plants to the soil, or a case of the acid-clay colloid robbing the plants of cations during their growth period. At the higher pH values, or higher degrees of calcium saturation of the clay, larger amounts of calcium were taken off the clay by the plants to lower its pH and make the clay more acid. (See Fig. 1)
Fig. 1. Changes in pH of calcium clay soils in consequence of soybean growth (first series). From Ref.2
Chemical analyses of the crops and tests of those clays for nitrogen, phosphorus and potassium after crop growth, showed that all three of those elements (two anions and one cation) moved to the clay from the plants to give crops (tops and roots combined) with less of each of those three elements than were initially in the planted seed.
We had grown plants which were holding on to their calcium, but were losing other cationic nutrients back to the soil. The startling facts were (a) the suggestion that the calcium, in particular, was not readily taken away from the plant by the clay of such high hydrogen saturation and, reciprocally, of such low calcium saturation; and (b) that nodulation did not occur at all the higher chosen percentages of calcium saturation. The losses from the plant back to the soil occurred in seventeen of the eighteen pans in the three series. It required a percentage saturation of the highest listed above to bring about what would seem like legume plants able to move nutrients in normally expected amounts into themselves from the supplies adsorbed on the clay.
That the lowest percentage of saturation, by calcium, of the soil’s exchange capacity for legume growth and nitrogen fixation (in soils of low organic matter) lies near but above sixty, was duplicated in another study.3 (In the preparation of the colloidal clay from the Putnam Silt Loam8 as a plant growth medium on which the concept of plant nutrition as a balanced cationic ration is based, its final chemical analysis showed that it carried but little organic matter. But even after treating it with 30% hydrogen peroxide at 100°C for 24 h it still contained 1.50% carbon and 0.15% nitrogen. Hence, the technique of using colloidal clay must be considered using a soil highly devoid of humus, save as that might be a part of the clay11 molecule.)
Other studies and experiences from soil tests have suggested similar standard values for other nutrient cations at the following ranges of percentages saturation as balanced nutrition from the soil; magnesium, 7-15% (possibly 10-20); potassium, 2-5%; and sodium 0.5-3%. With calcium at a figure so much higher among the four cations, it is evident that small excesses in the saturations of any of the latter three would quickly represent imbalances in the plant diet from the soil by them in relation to the calcium, when its high percentage saturation required would so often be too low.
In these citations of past experiments and experience, the calcium uptake in the very early plant life of seed germination and sprouting of the rootlet with such lasting effects on plant growth, suggests that, in practice, the calcium should be applied in contact with the seed at planting. The lasting effects suggest that the root growth carries calcium, and the favorable effects of that, along with the root’s extension of itself, when nodulation results in the deep, or distant and even inimical, untreated soil horizons.10 Dare we envision the root hair as a calcium-membrane of which the seemingly ‘selective absorptions and exclusions’ of nutrient elements manifested only when cell-wall’s normal calcium saturations, activities and effects are exhibited only according as the surrounding clay, too, has a relatively high calcium saturation and the exchange services of which to the root-hair are not offset by competition of its excessive saturations by magnesium, potassium and sodium?
Membrane studies under observations by means of the electron microscope by Preston13 exhibit cell-wall structures made up of parallel fibrils of closely-laid cellulose, bound by cross-fibrils of calcium at regular spacings with their filtering functions, seemingly, dependent on this calcium arrangement. Dare we not believe this calcium arrangement and its functions in the root-hair membrane are modified according to the varied degrees of calcium saturation of the soil’s colloidal clay-humus enshrouding the roots and thereby also to that clay’s degrees of saturations by magnesium, potassium and sodium as disturbers? Can these latter high saturations not be particularly significant in desert soils where cations, other than calcium, are so recognized, and more so than we imagine by our agricultural experience and studies on only acid, humid soils?
Plan of Testing the Theories
The sugar beets seemed to offer themselves as a unique, nonlegume crop for testing the validity of the theoretical considerations prompting this study, since their seeds are borne as clusters in a glomerule, or brood-seed. That simple, natural fact would permit measuring the increased field stands of beets from planting the clusters treated with a natural gypsiferous mineral, in contrast to similarly treated and measured stands and yields of beets from planting the single seeds from clusters broken and the seeds covered as preparation for precision-spaced plantings. Such procedures would allow the use of sugar beets from seeds, treated and untreated by calcium, as a physiological test by stand counts and crop yields, of the soil’s offerings of, and needs for, this nutrient element in the early life of these non-legume seedlings.
Also the yields of the sugar beet on field dimensions, harvested carefully and measured accurately as tonnages per acre when the freshly harvested crop goes directly from the field to the sugar mill to make the data collection an integral part of gathering the crops commercially for simultaneously measured quantities and qualities resulting from calcium treatments of the soil growing the sugar beets; all seemed to offer an inviting experimental test of the theoretical considerations on such an extensive field scale.
The soil treatments and crop tests were made in the desert area of the Imperial and San Joaquin Valleys of California, by applications of the partially granular7 commercial product known by the trade name of ‘Mineralag.’ (Mineralag is a natural resource, a ground rock fertilizer and soil additive of volcanic, hydro-thermal alteration and mineral deposition [Colorado, e.g. Sec. 9-10; T47N; R11E] of mainly calcium sulfate consisting of a collective balance of primary, secondary and micronutrient (trace) elements for plants, with other unique physical, chemical and biological assets.)
The field trials extended over the years 1965-1967, inclusive, in both arid valleys in close proximity of the mills of the Holly Sugar Corporation by whose cooperation through their agronomist and other interested scientists, the tests were carried out.
Results
Stand counts. Imperial valley. Planting 1965. Harvest 1966.
In making the stand counts, usually some area of the adjoining two field plots under test was selected, say, one hundred paces, 275 feet, from one or the other ends of the field, to eliminate irregularities occasioned by flooding, back-up, or washout by irrigation or any other accidental disturbance. At this point two strings, 25 feet apart, were stretched across the plot and all the young beet plants counted per row between them. These row counts of 25 feet each were repeated in other areas of the plots to arrive at an average number of plants, or mean, for 20 to 48 such row lengths.
In connection with the 1965 planting, for the 1966 harvest, only four of the total of seven growers’ tests were subjected to stand counts, of the treated and the untreated plants. In the Table No. 1 giving the data of stand counts, there are given additional data, namely; (a) acres of test plot area; (b) rates mineralag applications, pounds per acre, (c) placement of it; (d) total row-length counted; (e) average numbers of plants per 25 feet row length treated and (f) untreated; and (g) increase as percent of plants per row due to the single mineralag treatment at planting.
Table 1–Stand counts, Imperial Valley, planting 1966, harvest 1966
| Grower’s contract number | Acres in test plots | Mineralag application lbs/A** | Placement | Total length counted (feet) | No. of Plants per 25 Ft. Section | Increase by mineralag (per cent) | |
| Treated | Untreated | ||||||
| 112 | 11.0302*
5.5151 |
475 | On planted row | 500 | 94.50 | 87.00 | + 8.62 |
| E. 109 | 6.1746
6.1746 |
414 | On preshaped bed | 600 | 72.00 | 64.75 | + 11.19 |
| 252 | 5.454
5.454 |
1012 | Before listing | ||||
| W. 109 | 6.106
6.106 |
419 | On preshaped bed | 600 | 95.25 | 80.37 | + 18.51 |
| 35 | 2.651
2:651 |
346 | On listed bed | ||||
| 539 | 7.552
7.552 |
180 | On listed preshaped bed | 1200 | 82.95 | 76.25 | + 8.78 |
| 30 | 112.1952
112.1952 |
209 | On planted row | ||||
*The upper figures represents plot treated mineralag, the lower figures, the untreated ones.
** In addition to mineralag, the plots were given also nitrogen at 200-250 lbs per acre of ammonia, or the equivalent, and also 200-250 lbs per acre of ammonium phosphate, 11-48-0.
It is significant to note from the data that the single seeds of sugar beets, planted in 1965 by the various placements (a) on listed and/or pre-shaped bed; and (b) on top of planted seed row, showed the minimum increase in stand of 8.62 per cent, and the maximum of 18.51 per cent. Those occurred when the associated additional soil treatments were nearly a constant, to include nitrogen (most commonly anhydrous ammonia) and ammonium phosphate at closely constant rates, but none of which carried applications of calcium.
Such data giving those increases in the numbers of plants from single seeds planted, leave no doubt but that these non-legume crops of sugar beets needed calcium sadly on those heavy, desert soils, if the potential crop stand was to be obtained from the seeds planted and normal to beets on fertility-balanced soils.
They also point to the small application of the calcium carrier needed, even when not in direct contact with the planted seed, to increase the germination of the seed numbers, planted at the customary rate of six pounds per acre.
Those data were the early suggestions that the excessive saturations of the soil’s exchange capacities by the cations of magnesium, potassium and sodium in contrast to the deficient saturations by calcium and no saturation by hydrogen, or acidity–shown by the soil test–made the chances for the calcium cation as competitor for entrance into the plant root-hairs all too low for them to manage the nutrient intake as we believe is common when ample calcium is taken by the crop during the early growth of the seedlings.
The high stand counts of 95 plants per 25 feet of row suggested an excessive rate of planting to limit each beet to less than a 4 inch diameter as row space, to say nothing of soil volume per beet too severely limited for ample calcium nutrition in a soil so hostile and not what would suggest a balanced one, especially with respect to sufficient calcium.
Harvests of sugar beets. Imperial Valley. 1966.
There were seven test areas of sugar beets, including untreated soil and such treated with mineralag (a gypsiferous material), planted in the fall of the preceding year with stand counts taken and harvested in 1966 in the Imperial Valley. The resulting harvest data are given in Table No. 2 as tonnage increases per acre due to treatment over the yields per acre for the companion area given no mineralag. Also, the increases of yields are given as percentages increase and monetary earnings per acre over those from no such treatment.
Along with those data, there are also given such of several soil fertility factors revealed by soil tests, (a) the soil’s total exchange capacity; (b) the calcium deficiency as percentage relative to the desired degree of calcium saturation; (c) the excesses of magnesium; (d) of potassium; (e) of sodium; and the imbalance of cations (exclusive of sodium) against calcium, calculated as the addition of percentage figure for calcium as deficiency, and for magnesium and potassium as their excess, or a total quantitative evaluation for these three prominent nutrient cations, commonly held on the exchange capacity of the soil’s colloid.
Against these hindrances of the desert soil’s imbalanced cationic array, it is quite surprising to find the application of calcium in mineralag increasing not only the stand counts cited in the first of the preceding two tables, but it is of more significant note that the early entrance of calcium into the seedlings and its nutritional effects carry through the growing season to give increases as tonnage of the harvested beets in six of the seven trials. With only one of the tests giving a decrease in crop harvest, the increases of it as percentage over no treatment in the six, in the order listed, ranged from 1.89 to 10.69 with a mean of 5.03 per cent.
Table 2–Increases in yields of sugar beets 1966 and related soil test data, Imperial Valley 1966
| Grower’s contract number | Tons | Increase per acre | Exchange capacity me/
100 g*** |
Calcium deficiency
% |
Soil test data | ||||
| Per cent | Earnings | Magnesium excess
% |
Potassium excess
% |
Sodium excess
% |
Imbalance (sodium excluded)
% |
||||
| 112 | -0.649 2
26.3821* |
– 2.46 | – 2.19 | 44.00 | 18.0 | 71 | 122 | 166 | 211 |
| E. 109 | + 0.3530
18.6360 |
1.89 | 16.22 | 45.50 | 18.0 | 74 | 102 | 164 | 194 |
| 252 | + 0.7526
16.2767 |
4.62 | – 5.11** | 37.00 | 6.9 | 42 | 79 | 122 | 128 |
| W. 109 | + 0.7616
17.3771 |
4.38 | 7.04 | 49.00 | 19.0 | 75 | 44 | 172 | 136 |
| 35 | 0.7767
16.4328 |
4.72 | 4.43 | 51.00 | 25.0 | 74 | 60 | 274 | 159 |
| 539 | + 0.9871
25.4973 |
3.87 | 31.79 | 37.50 | 11.0 | 44 | 60 | 235 | 115 |
| 30 | + 2.1714
20.3325 |
10.67 | 27.77 | 47.50 | 23.0 | 70 | 103 | 240 | 196 |
* The lower figure represents the yield of the untreated plot (given no mineralag) while the upper figure is the increase in tonnage per acre by that soil treatment.
** The reduction in percentage of sugar in the beets in connection with heavy application of nitrogen and also of mineralag (discussed in the text) points to cation need for ‘balanced’ plant nutrition, especially in case of soils low in organic matter.
*** These high exchange capacities, and other test data, point out (a) these very ‘heavy’ clay soils; their alkalinity, or high pH, with all, save one, at 8.0 or above and (c) their low organic matter content, all below 100% save one, contract number 112.
That calcium as an alkaline earth, moved into the plants early for such lasting seasonal effects, represents significance when that was competition against the combined excess of potassium, an alkali, and of magnesium, another alkaline earth, all of them combined to be more than 100 per cent as excesses. Then, when the pH figure of all these soils were 7.9 and above, it is all the more expectable that not very much calcium would be ‘available’ from such soils to the roots of the sugar beet, at least not for long after the calcium was applied and the soil’s equilibrium state had become established. Then, too, with the organic matter contents of these soils scarcely up to 1.0 per cent, there would be little microbial dynamics as seasonal decay making the nutrient cations active.
Certainly, when their test data are viewed, these soils represent themselves deficient in mobility of calcium into any plant roots. Accordingly, calcium emphasizes itself prominent as a needed nutritional element, not only for legumes but here for a non-legume, when for sugar beets its fresh application in less than 500 pounds per acre with the seeding does so much in both increasing the stand count and in carrying forward its growth-stimulating effects through the growing season to give increases as much as a mean of five per cent in the harvest of this biennial root crop.
There is suggested, in these data, one item of caution against excessive application of the calcium carrier on the sugar beet, illustrated by the grower contract No. 252 when the fertilizations associated with mineralag applied 238 pounds of the element nitrogen and simultaneously the mineralag application was 1012 pounds per acre equivalent of 185.8 pounds calcium in this 1966 harvest.
While there was a yield increase of .7526 tons per acre, or 4.62 per cent over the check plot, yet there was a decrease of $5.11, or loss, per acre between the two plots because of the extra calcium or excessive nitrogen.
Since calcium is physiologically effective in the plant’s synthesis of proteins in opposition, or antagonism, to potassium’s activities in sugar, or carbohydrate, synthesis, there is the suggestion that the high rate of calcium application was an excess to disturb sugar production and to reduce the financial returns even when the yield as tonnage was favorable by nearly five per cent.
Stand counts. Imperial Valley. 1966.
The resulting data from the stand counts and yields from the initial phase of this study 1965-1966 gave the suggestion that the application of the calcium carrier for the sugar beets might be more effective if placed in closer contact–and possibly at higher application rates–with the planted seeds. Accordingly, such was the theoretical consideration tested in all the plantings of the plots in 1966 for the stand counts in the Imperial Valley and the measured yields as increased by the single soil treatment of mineralag for the harvests in both the Imperial and the San Joaquin Valleys in 1967. No stand counts were taken in the San Joaquin Valley due to the single seed, precision planting used there in 1966.
The same procedures for the stand counts in the Imperial Valley, 1966 were followed as were used there the preceding year. The data for the latter are assembled in Table No. 3 in arrangement duplicating that of the first Table No. 1.
In the 1966 plantings for the 1967 harvest in the Imperial Valley, the applications of the mineralag were made directly on the seed while planting, except in the case of the Contract Number 312, where it was a sidedressing. The rates of application ranged from a low of 54 to a high of 210 pounds per acre. The additional fertilizations carried no significant amount of calcium, when 200 to 300 pounds per acre of 11-48-0 were applied in most all cases, and additional nitrogen, from 75 pounds to heavy applications in the several forms of this element commercially available and encouraged for direct application in connections with crop plantings.
Table 3–Stand counts, Imperial Valley, planting 1966, harvest 1967
| Grower’s contract number | Acreage in test plots | Mineralag application lbs/acre *** | Placement | Total length counted | No. of Plants per 25 ft row | Increase by mineralag (per cent) | |
| Treated | Untreated | ||||||
| 371
312 (Burson) 716 ‘Y’ 307 ‘X’ 307 Jordan 530 Jordan 531 373
|
5.52*
0.992 5.30 3.716 3.224 3.224 5.89 11.78 2.88 |
54
160 105 172 200 124 210 125 107 |
On seeding
side-dress On seeding On seeding On seeding On seeding On seeding On seeded row On seeding |
700
600 600 600 400 400 700 700 600 |
62.3
148.4 148.2 74.6 68.9 62.4 84.7 100.8 100.3 |
61.6
124.7 118.2 69.0 62.7 57.8 53.3 94.2 90.3 |
1.0
18.0 25.3 8.1 9.9 7.9 59.1*** 6.9 11.0 |
* Treated and Untreated plots by mineralag were of same dimensions, hence only one acreage figure is recorded.
** Additional soil treatments, much similar to those reported in Table 1 were again used including generous applications of nitrogen and up to 400 lbs/a of 11-48-0. The plant spacings in the row varied from ¾“ to 3“.
*** Mineralag served as an anticrusting agent in connection with heavy rains just after planting. Nematode infestations were also present.
In all nine cases of stand counts in 1966 for the 1967 harvest, there were increased stands ranging from 1-59 per cent, due to the applied calcium carrier of two hundred pounds and less per acre directly on the seed while planting. Since for both the 1966 and 1967 harvests in the Imperial Valley, the single seeds were planted, so prepared by breaking the seed clusters, and in some cases coating the seeds for more precision in spacing the plants in the row, the effect of the treatment on improved germination spells out an improved physiological response shown as increased plant stand, and not one hidden in the planting of seed clusters. This fact suggests that the beets’ roots must retain this extra calcium well into the growing season to improve also the root’s uptake (or exclusion) by the calcium-laden root hair of other nutrient elements, possibly out of balance in the soil as normal plant nutrition.
A startling observation, made soon after seeding the 1966-67 beet crop, deserves mention in connection with the heavy rains on the freshly planted seeds on September 15, 16, 18, and October 3 and 4. Such rains usually give crust formation of those soils, prohibiting seedling emergence and crop survival. Mineralag treatments gave a stand increase of 59.1 per cent over no treatment by the stand counts. Instead of crustation, there was excellent granulation of the surface soil layer containing the shallow-planting of the beet seeds. Some questions arise here as to the efficiency of applied (distilled) water by the sprinkler system of irrigation in contrast to salt-laden water delivered by furrow irrigation. Some questions arise also as to possibly more effective calcium uptake by the plant in its early life and possibly improved adsorption of this cation on the clay in competition with the excessive magnesium and others in the cationic array there, by such kind of salt-free water as sprinkler treatment.
Harvests of sugar beets, Imperial Valley, 1967
There were nine test areas of sugar beets, managed for the 1967 harvest data, much as was done for the 1966 harvest in the same area. The data for 1967 are given in Table No. 4 in arrangement similar to that for the yields of 1966.
Table 4–Increases in yields of sugar beets and related soil test data. Imperial Valley 1967
| Grower’s contract number | Tons | Earnings
$ |
Exchange**
capacities me/100g |
Calcium***
deficiency % |
Magnesium
excess % |
Potassium
excess % |
Imbalance
(sodium excluded), % |
Sodium
excess % |
| 272
371
312
332
‘Y’ 307
‘X’ 307
716
530
373
|
– 1.0925
21.1170* – 0.8531 29.1250 – 0.2645 21.6586 – 0.1727 21.2839 – 0.0559 23.7484 – 0.1149 24.2965 0.8977 34.5618 1.9663 23.3254 2.0486 29.4739 |
– 18.14
– 12.62
– 4.39
– 2.89
– 0.92
1.91
15.35
32.54
34.03
|
37.0
38.0
40.0
40.0
40.5
36.0
41.0
47.5
25.0
|
16.1
9.6
13.6
11.1
10.9
10.0
9.1
17.4
– 11.1
|
63.6
30.2
54.8
53.7
61.5
51.5
62.4
70.5
16.0
|
13.6
56.4
0
44.6
77.9
33.3
9.0
68.4
– 58.0
|
93.3
96.2
68.4
109.4
150.3
94.8
80.5
156.3
– 52.1
|
270
425
304
200
135
250
141
222
70
|
* The lower figure represents the yield of the untreated plot (given no mineralag) while the upper figure is the increase in tonnage per acre by that treatment.
** Exchange capacities are given as milligram-equivalents per 100 g dry soil. Most of them are high values to indicate ‘heavy’, clay soils, save Reese Ranches sample, approaching a silt loam. These high cationic exchange capacities were not due to soil humus or organic matter ranged by soil tests from 0.1 to 1.3 percent.
*** Of the four cations calcium was deficient in all but one case, viz Contract 373. The other cations, magnesium, potassium and sodium are all excesses except again for contract 373 pointing to the soil as responsible for the order of arrangement of improved yields and earnings.
Again the soil factors, as measured by soil test, are tabulated with the crop yields, and offer their suggestions that calcium should be considered the major deficiency in nutrition of planted seed, especially in soils of such heavy texture, so devoid of organic matter and carrying so much excess of the cationic salts.
Harvest of sugar beets, San Joaquin Valley, 1967
Sugar beets were planted also in San Joaquin Valley for 1967 harvests on six test areas, ranging in test plot sizes from 2.56 to 6.64 acres, duplicated for treated and untreated soil trials, representing a total each of 26.99 acres in this arid valley of California.
Precision plantings of single but coated seeds, in larger spacings, ranging from two to eight inches within the row were used, with the calcium carrier applied in direct seed contact at planting. No stand counts were taken. The yield data related to the soil factors, by soil tests, are assembled much as in preceding style, in Table 5 for the six test areas.
Table 5–Increases in yields of sugar beets, San Joaquin Valley, 1967, and related soil tests
| Grower’s contract number | Increases per acre | Soil test data | ||||||
| Tons | Earnings $ | Exchange capacity me/100 g*** | Calcium deficiency
% |
Magnesium excess
% |
Potassium excess
% |
Imbalance (sodium excluded)
% |
Sodium excess
% |
|
| 254
232
286
254
241
282
|
0.1440
16.4002* 1.0148 10.9954 1.1847 35.2871 1.7344 5.0546 2.6747 31.6001 6.7389** 29.7388 |
1.93
13.60
15.88
23.06
35.84
90.31
|
27.0
38.0
30.0
8.0
40.0
34.5
|
0
10.8
-13.3
-21.6
10.8
0
|
50
60.6
14.0
8.8
9.5
12.1
|
56.9
-6.0
11.0
-68.1
71.4
14.2
|
106
71
38
92
91
26
|
0
217
33
0
505
94
|
The lower figure represents the yield of the untreated plot (given no mineralag) while the upper figure the increase in tonnage per acre by that soil treatment.
* The seed spacings in the row of this sixth grower were 8″; the fifth grower 4″; growers No. 2 and 3 were and for growers No. 1 and 4, 2″.
The yields of sugar beets indicate clearly that these soils did not carry such excessive salts as was true of Imperial Valley soils. The column of ‘imbalance’ indicates similarly by its lower percentage values, which suggest four soil areas of ample or near ample calcium, by soil saturation values as two negative and two of zero deficiencies, or near ample soil calcium supplies. Similarly, two cases show negative excess of potassium, or represent deficiencies rather than excess of this salt in relative degree of soil saturation, hence the ‘imbalance’ values as calculated were of lower percentages than were found for the soils of Imperial Valley. Accordingly this arid soil is less inimical by both soil test data and sugar beet crop-yield test results than the soils of Imperial Valley.
That the calcium deficiency, in its mobilization into the sugar beet, a non-legume crop, is the major problem for growing crops in such soils is verified by the response of the sugar beet by yields to the mineralag application at rates per acre no larger than 93 to 166 pounds. All six tests were positive with yield increases from 0.87 to 34.0 per cent, because of possibly the less inimical soil, but very likely also because of larger plant spacings in row planting to escape excessive crowding of plants and cutting down potential soil fertility supply per plant.
Attention is directed to the two separate 254 tests, namely B-1 and A-2; the former a heavier silt loam of 27.0 me exchange capacity, and the latter a sandy soil that with of but 8.0 me. The beets on the sandy soil were observed to have a ‘heavy’ infestation of nematodes while the silt loam had but a ‘light’ one. One expects lesser infestation in soils of heavier clay, a fact reported in the earliest California studies of this pest on citrus. In these cases it is significant to note that the soil factors showed the sandier soil the less hostile as an alkaline one, but yet the application of the mineralag calcium carrier gave a larger improvement in tonnage yield (over 30 per cent) above the untreated soil with but 5.0546 tons per acre to suggest that given extra calcium, the sugar beet built improved self-defense against this plant pest.
Chemical Analysis of the Sugar Beets Grown on Untreated and Treated Soils
Since the extra calcium offered to the beet plant by the soil treatment gave extra tonnage harvests per acre, some ash analysis were made of the dry matter of beet samples from the Contract Number 307, Lot ‘Y’ in 1967 to get suggestions as to which elements represented increased or decreased effects by the calcium treatments on possibly the plant root hair activity as one of either increased uptake or exclusion as effects modifying the plant’s chemical composition. The beet samples were collected and prepared with meticulous care, washed, sectioned to use one eight of each of twenty beets, quickly frozen, to represent the lot for ash analysis of the dry matter, from both treated and untreated test plots.
The results of the analysis are given in Table No. 6 for six major nutrient elements as percentages; and for six micronutrient elements as parts per million, with indicated increase or decrease as the modification of the sugar beet’s chemical composition by the gypsiferous soil treatments. No determinations were made of the nitrogen, carbon or volatile and organic combinations expected to be modified compositions because of the generous nitrogen fertilizations, and those affected by calcium, which combination is a factor in protein synthesis and, thereby, possible reduction of the beet’s concentrations of stored sugar. The respective sugar percentages of each of the beet harvests are not reported in tabulations, but are indicated in some instances of shifts in the earnings by the soil treatments. Those dollar values are given along with the percentages increase in the yields per acre by the soil treatment.
Table 6–Chemical analysis of the treated and untreated sugar beets
| Elements | Grown on treated soil
(per cent) |
Grown on untreated soil (per cent) | Increase, or decrease by Mineralag soil treatments
(per cent) |
Change
(per cent) |
| Calcium
Magnesium Potassium Sodium Phosphorus Sulfur Iron Manganese Copper Cobalt Molybdenum Zinc |
0.399%
0.351% 1.059% 0.275% 0.086% 0.026% 799.7 ppm 42.8 ppm 11.8 ppm 0.1 ppm 0.5 ppm 21.6 ppm |
0.379%
0.351% 1.395% 0.263% 0.092% 0.051% 465.2 ppm 33.8 ppm 10.4 ppm Nil ppm 0.3 ppm 27.2 ppm |
Increase
None Decrease Increase Decrease Decrease Increase Increase Increase Increase Increase Decrease |
5
0 24 4 6 49 72 26 17 00 60 20 |
As to the shifts in the beet’s chemical composition connected with the dozen elements measured, it is significant to note, first, that the addition of the calcium sulfate to a soil, already high in sulfur, served to reduce the sulfur concentration in the beets by nearly fifty per cent. This suggests a physiological effect by the application of the cation, calcium, rather than of the anion, sulfate. The increase in the amount of calcium within the beet by but 5.2 per cent, according to the table, was effective in bringing about (a) neither reduction nor increase in the seemingly excessive magnesium; (b) a reduction of potassium by 24.0 per cent, and only a very slight increase in sodium, as the changes among the cations in their prominent imbalance. There was a slight reduction in the beet’s concentration of phosphorus, indicated by test so excessively present in the soil; but there was a decided reduction of the sulfur in the beets considering these two major anions.
Among the trace elements, it is significant to note the relative increase of iron concentration in the beets by 70 per cent; (b) an increase in manganese by over 25 per cent; (c) also increases of copper, cobalt and molybdenum; but (d) a decrease in the beet’s concentration of zinc.
These are decided changes in the chemical composition brought about in the beet as the ‘seed’ plant of a biennial plant cycle, when there was but such a small change by the limited soil treatment in the plant’s concentration of calcium. Accordingly, the calcium suggests for itself the physiological roles as tools, or controls, rather than as materials in plant construction. Dare we not envision its role possibly as membranous control within the root hair, or other activities, requiring little change in the supply or concentration of calcium while functioning to control many elements entering the plant from the soil, some as increases and some as decreases in the plant’s uptake?
If the increased growth of a ‘seed’ plant, like the biennial sugar beet, and the associated changes in its chemical compositions in connection with seed treatment by calcium are considered as gains in the struggle of the species to survive in a given environment, then the absence of any shifts in magnesium concentration might be interpreted as if this element, as another alkaline earth chemically so closely similar to calcium in its biodynamics, is unaffected in its movement into the beet by the small alteration of this root’s calcium content occasioned by the small applications of calcium. These two alkaline earths, apparently, cannot be considered ‘antagonistic’. But for potassium, the beet’s better growth registered a reduction of, or a ‘defense’ against, the high potassium saturation of the soil. Such defense against the soil’s excessive sodium was weak, and likewise against the excess of the anion, phosphorus; but against the other anion, sulfur, there was a strong defense shown by a reduction of 50 per cent of it in the beet’s dry matter, even if calcium was applied as the sulfate.
Amongst the trace elements, five of the six increased their concentrations in the beet’s dry matter to suggest these shifts as advantages in survival, or as hindrances in deficiency unless the calcium was added to the soil. That same line of reasoning would apply to the zinc as an excess, or hindrance, in the soil in contrast to deficiencies or low availabilities in the soil of the preceding five in the set tested.
Discussion
The series of soil fertility tests under the sugar beet crop exhibits the problem of balancing most effectively the soil’s nutrient availability with respect to both the crop yield as tonnage and the concentration of sugar in the beet root. The reduction within the latter of the mono- in relation to the di-saccharide is a challenging criterion for more successful soil fertility management. The effects of the higher supplies of available calcium taken into the beet when heavy applications of nitrogen fertilizers were simultaneously made suggest reduction in concentration of the sugar. That was clearly shown by the monetary earnings, sometimes as reductions in spite of increased tonnages yielded. None of those data are recited in detail in these studies, however economically important they become since the tonnage yield, multiplied by the percentage of disaccharide sugar determines the pay check for the crop at the sugar mill.
The excessive degrees of the soil’s saturation of its exchange capacity by magnesium in these desert soils, and the beet root’s manifestation of little, or no, control of magnesium uptake in relation to its increased uptake of calcium–shown by chemical analysis of the root’s dry matter–suggest the magnesium, an alkaline earth, as the one most serious disturber in the plant’s uptake of, and functions by, calcium, another very similar alkaline earth.
The excess of magnesium saturation on the soil colloid, even enough to leave extra salt, bringing plant behavior equivalent to a calcium deficiency, has come into prominence in recent years under wider observation of the nutritional help from drilling gypsiferous, natural minerals for crops, both legumes and non-legumes other than the sugar beet. This has been the case, especially in areas of high concentrations of magnesium in the drainage water of the United State, as has been indicated accordingly by the map of such magnesium (over 20 ppm) drawn by S. B. Detweiler, from the Data of Geochemistry, F. W. Clarke. More critical study should offer more information.
In considering the major plant nutrient cations, viz: hydrogen, calcium, magnesium, potassium, sodium and others, in that order of their decreasing energy of adsorption which holds them exchangeably on the clay-humus colloid in the surface horizon of the soil profile, dare we not envision such a particular energy situation giving also the same order of the cations as decrease in their bio chemo-dynamics in soil development, and in nourishment for evolution of the plant and microbe species on the earth’s surface in what is called “The Living Soil”?
In the humid soils, hydrogen takes on its tremendous biochemical importance at the ionic concentration of carbonic acid by which it weathers rock, leaches out salts, and leaves the clay residue with its unique colloidal capacities holding, against leaching, the array of nutrient cations exchanged therefrom by root-respiration’s carbonic-acid hydrogen emphasizing the clay’s requisite highest saturation by calcium and its connection with protein synthesis and nitrogen fixation. The other cations follow in order of lesser amounts and lesser chemo-dynamics on the clay as more nearly naturally balanced, and available plant nutrition.
The moderate degree of acidity of humid and semihumid soils, near pH 6.0 with their calcium reserves, makes them most productive for plant’s synthesis of protein-rich tissues; their liberal reproduction; and their specific compounds for immunity and self-protection against diseases and pests. Calcium within the root-reach in the profile and its application on the soil surface as liming material of humid soils have lacked our appreciation of its importance as a plant nutrient. The cation, hydrogen, has suffered similarly under its consideration as the arch-enemy in soil acidity. This confusion, covered by the term ‘soil acidity’ so widely, has prevented their recognition of calcium and hydrogen as major cations supporting nutrition of the entire biotic pyramid. The hydrogen, root-made by its respiration, has been the mobilizing agent, via its high adsorption energy, of the other essential cations from the clay-humus colloid into the plant roots by exchange phenomena. Soil acidity has been nutritionally beneficial, in the presence of ample reserves of calcium first, and all other cations following in order to give these two, major cations energywise, their foremost nutritional importance in both legumes and non-legumes.
But in the neutral or alkaline, arid soils, there are no measurable hydrogen ions adsorbed on the clay, and there is too little soil organic matter under microbial decay to maintain active hydrogen equal to the benefits from carbonic acid. Accordingly, dare we not envision calcium, the second prominent cation in the order cited, coming into prominence in evolution and survival of the plant species via early plant functions suggested by help in germination and seemingly early control of nutrient intake as well as exclusions by which the plant’s nutrition may have been naturally managed for survival? The manifestations by calcium’s nutritional roles in the sugar beet, as the equivalent of a ‘seed’ state in its biennial cycle, seem to transcend highly any similar roles we might associate with the cations magnesium, or potassium. Those manifestations suggest themselves as reasons for the higher degrees of saturation by calcium of the soil’s exchange capacity, especially for only connection with processes like protein synthesis and nitrogen fixation, and more so for calcium’s reported requisite role by presence for the amino acid synthesis brought about by the electrical spark’s discharge in the gaseous combination, duplicating the earth’s primitive atmosphere by which such phenomenon probably gave origin to primitive living substances.
Acknowledgements
The author acknowledges the cooperative service by the Holly Sugar Corporation, General Offices, Colorado Springs, Colorado, in this project, by whose help the field scale of measurements, the numerous chemical determinations, weights and other contributions, the project was made possible.
This report was prompted by many requests from The Geyser Minerals Corporation, C. George Swallow, President, Denver, Colorado, for suggested tests of their natural, gypsum-like, fertilizer, named ‘Mineralag.’ The author suggested field-crop tests of their product under seed contact treatments; on soils inventoried by chemical tests of exchangeable and available nutrients; for increased germination of planted seeds, consequently better stands by field counts; as harvested yields from treated and untreated equal rows of, say, sugarbeets; and measured chemical differences in composition of the beets as major and trace elements due to treatment. The study was carried out according to the suggestions and as they are reported in this paper.
The author was given opportunity to observe the field tests under harvests; was supplied with the extensive collection of assembled data; and given cooperation in his voluntary preparation of the manuscript; all of which added up to verification by the sugarbeet crop, a nonlegume, of many facts established initially for the legumes. The opportunities to make the report are hereby acknowledged by the author.
References Cited:
- Albrecht, William A., “Nitrogen fixation as influenced by calcium.” Proc. Sec. Intern. Congr. Soil Sci. Leningrad-Moscow Third Com. 3, 29-39 (1930).
- Albrecht, William A., “Inoculation of legumes as related to soil acidity.” J. Am. Soc. Agron. 25, 512-522 (1933).
- Albrecht, William A., “Some soil factors in nitrogen fixation by legumes.” Trans. Soc. Soil Sci. New Brunswick, N.J. R. 71-84 (1939)
- Albrecht, William A., “Calcium as a factor in seed germination.” J. Am. Soc. Agron. 33, 153-155 (1941).
- Albrecht, William A., “It’s the calcium, not the alkalinity.” Soybean Digest, September, 1941.
- Albrecht, William A., “The fertility problem of Missouri soils.” Proc. Missouri State Hort. Soc. Nov. 30, 1942, pp. 94-100.
- Albrecht, William A., “Relative effectiveness of coarsely ground and finely pulverized limestone.” Soil Sci. 61, 265-261 (1946).
- Albrecht, William A., “Colloidal clay cultures: Properties of the clay and procedures in its use as a plant growth medium.” Soil Sci. 62, 23-21 (1946).
- Albrecht, William A., “Soil fertility in its broader implications.” Missouri Agr. Expt. Sta. Research Bull. 765, 72-88 (1961) (Bibliography).
- Anonymous, “Lime soil to feed crops–not to remove soil acidity.” Missouri Farm News Service May 5, 1954.
- Ferguson, Carl E. (Wm. A. Albrecht). “Nitrogen fixation and soil fertility exhaustion by soybeans under different levels of potassium.” Missouri Agr. Expt. Sta. Research Bull. 330, 9 (1940).
- Harris, J. E., “Soil acidity.” Michigan Agr. Coll. Expt. Sta. Tech. Bull. 19 (1914).
- Preston, R. D., “Structure of plant polysaccharides.” Endeavour 23, 153-160 (1964).