Access to all articles, new health classes, discounts in our store, and more!
Nitrates: Possible Poison Grown Into Foods
Published in Let’s Live, July 1961.
* * *
The conversion of organic nitrogen of the soil into the highly oxidized form of nitrate (saltpeter) is a natural microbial activity in any fertile soil. This soluble kind of nitrogen is taken readily from the soil for normal plant nourishment. But under disturbed conditions for the plant and for the soil microbes, like drought and its disruptions, these may cause the nitrates to accumulate within the plant rather than to function normally for their synthesis into plant proteins.
Excessive nitrates in forages and foods have brought disturbing publicity in the last few years. That was a sequel to the shortages of soil moisture in the mid-continent for the highly-fertilized corn crop in particular.1 The damaging effects of the high temperature on the plant’s physiology have not been discussed. Emphasis has gone to the dangers from nitrogen applied as a fertilizer. There have been animal deaths on some soils not given fertilizers and such even before the wider use of them, as soil treatments.
Proof is Difficult
It is extremely difficult to prove any substance poisonous in feed or food, especially when it comes from a fertilizer–by way of the soil and the crop plants–to animals and man. For such proof in court, it seems almost necessary to exhibit the corpse, or some morbidly clinical case. A sub-clinical one is not sufficient. Animals killed by nitrate poisoning have been periodic exhibitions. They represented terrific losses to individual, but scattered, farmers over many years. They have been reported in the publications of the Experiment Stations in certain climatic sections of the United States and with careful diagnoses as far back as 1895.2
Potassium nitrate (saltpeter) was found crystallized out on drought-stricken corn stalks which had poisoned cattle eating it with the fodder. The burning of the dried vegetable matter gave a sparkling effect of miniature explosions of gun-powder. That phenomenon was considered a simple test for nitrates as poisonous feed.
Precautions Taken
The pioneers, who used nitrates in the salt mixture for tanning pelts and hides, made certain they were kept away from livestock, which were inclined to lick or chew the salty skins with lethal results. In the first decade of this century studies were under way at the University of Illinois which used humans in so-called “poison squads” to test the effects of saltpeter “cure” of pork on men consuming such in contrast to those taking similar meat cured without nitrates.
Nitrates in different vegetables have also been under study. They, like the forages for animals, proved that the soil, modified by the variation in the weather during the growing season or by certain soil treatments, could be a disturbing factor responsible for wide variation of the concentration of nitrate nitrogen in the vegetative mass. Naturally, the concentrations were low in the seed parts of the plants.
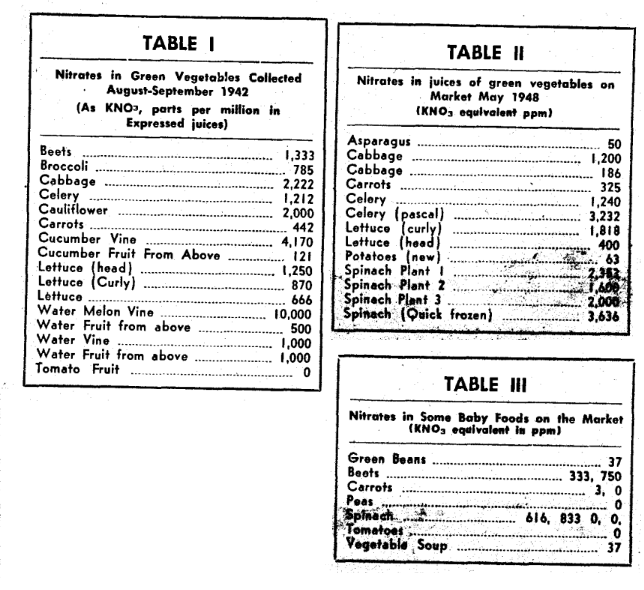
Professor J. K. Wilson, a distinguished (deceased) microbiologist of the Cornell Experiment Station, who was interested in the biochemical transformations of soil organic matter, reported many determinations of nitrates in plant juices, extracts and dry matter as early as 1942.3 His observations deserve attention anew, buried as they are now in unopened volumes.
Revelations Made
Wilson’s report revived interest in an earlier publication of the Kansas Station. In that,2 N. S. May reported on cattle killed by eating drought-stricken corn fodder in 1895. That grain crop was then just going west, in its move from the humid to the semi-humid area. C. S. Gilbert, et al., of Wyoming, reported nitrate poisoning for cattle by weeds under drought effects in 1946.4 two years later, the Easter Sunday Edition of the Kansas City Star had the headline, “Win in Dash for Life”, on March 28, 1949, reporting the recovery of a baby from nitrite poisoning via shallow well water used in its formula, after a dash to a Kansas City hospital.
In the Kansas situation, a very dry autumn and early winter were followed by an accumulation of later winter snows. Those were melted by the early spring rains to move the soil nitrates into the wells of mid-Kansa, where only the third one of the reported babies so poisoned was saved from death. It was those buried ideas in the older reports that prompted Professor Wilson’s extensive testing of leafy vegetables, frozen foods, baby foods, and other possible nitrate carriers among foods to learn whether they may contribute to hemoglobinemia, or other toxic, if not lethal, conditions for adults. Some of his data for vegetables are given in the accompanying Tables I, II, and III.
Chemical Reactions
Those earlier reports tell us that, “When the nitrate is ingested, it passes into an environment where oxygen is in demand; the nitrate is reduced to nitrite; and the latter is toxic. This nitrite, once in the blood stream, combines with the blood. It produces a methemoglobin to prevent the blood from supplying oxygen to the tissues, and thus the body suffocates….The disease was reported similar to poisoning by hydrocyanic acid.”
Wilson cited the report that 1.5% of potassium nitrate in the dry matter of feeds was known to be lethal to livestock, or to cause abortion. Missouri more recently cites that amount as deadly; reproduction difficulties at 1.0-1.5%; symptoms suggesting vitamin A deficiency at 0.6-1.0%; and no trouble with less nitrates if a normal ration is fed. For the concentration of nitrate nitrogen in water for babies, J. G. Heart suggested the maximum safety limit of 10 ppm.5
We are a bit slow to learn that the soil is a biochemical matter about which we should be concerned, lest we upset nature as we try to manage her efforts more for our economic advantages in labor and monetary outlay. Hidden ideas in the past can help us see more of the natural in the garden and the field, if we will but open to some of these basic facts in the recorded experiences escaping us because the books remain unopened.
References Cited:
- George B. Garner, “Learn to Live with Nitrates,” Agr. Expt. Sta. Bul., 708, June 1958.
- Mayo, N. S., “Cattle Poisoning by Nitrate of Potash,” Kansas Agr. Sta. Bul., 49, 1895.
- Wilson, J. K., “Nitrate in Plants: Its relation to fertilizer injury, changes during silage-making and indirect toxicity to animals,” Amer. Soc. Agr., 35:379-290, 1943.
- Gilbert, C. S. et al., “Nitrate Accumulations in Cultivated Plants and Weeds,” Agr. Exp. Sta. Bul., 277:1-37, 1946.
- G. Heart, “New ‘Blue Baby’ Diseases,” Science News Letter, 53: No. 18, 275-276, 1948.