Access to all articles, new health classes, discounts in our store, and more!
Magnesium in the Soils of the United States
Published in Let’s Live, August 1966.
* * *
In the series of discussions of the importance of magnesium in plant nutrition, in these columns, from December 1964 to September 1965 inclusive, no attention was given to the broader pattern of magnesium of the soils of the United States as a whole. Yet for its companion cationic element, calcium, our country divides itself, soilwise, naturally into an East and a West. The 98th meridian of longitude marks the division closely. The division by soils runs roughly south from the northwestern edge of Minnesota to the southern tip of Texas. Those to the west are called “pedocals”–“pedo”, for soils and “cals” for calcium, with free carbonate of it in horizons or concretions in the soil profile. Those soils of the East are called “pedalfers”–again soils with aluminum “al” and iron “fe” prominent in the profile under conditions implying the shortage of any calcium as active there in its carbonate.
In the development of a soil from various rock minerals, the movement of magnesium down through the profile has not been given as much prominence as calcium. Yet in the drainage waters, as an average, from the country in its entirety, the mineral residues on evaporation of those waters–more often appreciated as the so-called “lime” clogging the coils in the water-heater–contain one-fourth as much magnesium as calcium, by weight. Calcium carbonate makes up nearly half of that residue from the waters, to make “lime in the tea-kettle,” a common observation in Western United States.
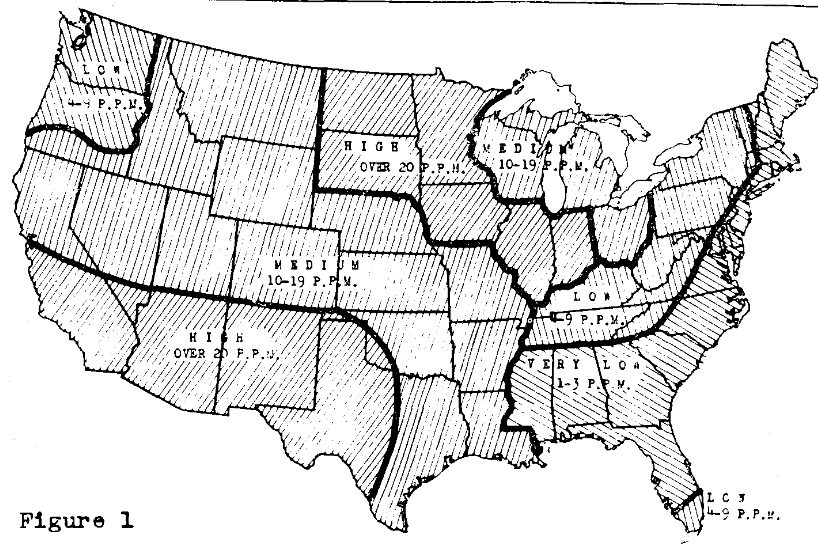
The United States Geological Survey has done extensive chemical analyses of drainage waters. In its Professional Paper No. 135, 1924, Dr. F. W. Clarke gave an extensive collection of data. From those Mr. S. B. Detweiler, now retired from federal services, gave us the generalized magnesium map for the country, shown here as Figure I.
These data suggest their relation to the varied degrees of soil development under the climatic forces, especially the rainfall as it is in inches per year in excess of the evaporation from a free water surface, also in inches per year. The former may also be given as percentage of the latter. Consequently, we would expect low magnesium in our soil waters in the Southeastern states. Accordingly, in the states northeast and east of Ohio and east and south of the Mississippi and Ohio Rivers, the “very low” and the “low” magnesium, namely 1-3 ppm and 4-9 ppm, respectively, are expected as the map reports. Then, in the high rainfall of Washington and Oregon, where the extremes of rainfall occur, magnesium is also “low,” or 4-9 ppm. In the rest of the area and a much larger one, magnesium is reported as “medium” and “high,” at 10-19 ppm and over 20 ppm. The Dakotas, Minnesota, Iowa, Illinois and Indiana with increasing annual rainfalls in that order from less than 20 to 40 inches per annum all show high magnesium. That fact serves to point out that the geological factor and not only the rainfall must come in for consideration. We, therefore, speak of the geo-climatic setting as determining the development of the soil.
The “low” supply of magnesium in the soil as a deficiency in crop production came into prominence some thirty years ago1 in the citrus groves of Florida, when attention first turned to the so-called “trace” elements. Emphasis had gone to the Marsh seedless grapefruit, which was giving larger yields as boxes of fruit than were the “seedy” ones. But when fertility elements, not common in commercial fertilizers, were applied separately on highly calcareous, sandy soils, it was the magnesium that increased the yields of the “seedy” fruits far above those of the seedless. But also, the flavor and taste were so much improved that there was not much premium on the seedless for the customer looking for quality along with quantity.
Magnesium demonstrated its biochemistry connected with the better survival of the species through healthy reproduction. But with that there came also the other products giving the quality as taste, possibly also assets in the reproduction of the species in passing over dormancy from the season of seed production to the planting of that, as it occurs in Nature. With our concern for the production of big bulk per acre, we are apt to forget that quality as fruit may be in the many compounds unknown, but coming along as aids in species survival. Magnesium is associated with many enzymes, and that element, like the enzymes, is a case where so little can do so much, without being necessarily spent in the service.
While magnesium, as mapped “very low” and “low” in Figure I may be a deficiency in highly developed soils, the converse, or excess of magnesium, may be the case in the less-developed soils in areas of lower annual rainfalls and mapped as “high” in magnesium. Early botanical literature says much about the required calcium-magnesium ratio. Irrigators suggest caution of magnesium-potassium ratios and calcium in relation thereto in the applied waters.2 More recently with our appreciation of the adsorption-exchange activities by calcium, magnesium, potassium and sodium on the soils, colloidal complex and their suggestions as to what ratios of these cations on it are a balanced diet for legumes and other more nutritious plants, there comes the ecological suggestion that areas “high” in magnesium on the map may be disturbing to fuller plant nutrition and crop growths. Excess magnesium may be equivalent to calcium deficiency, if not also of potassium.
Calcium plays a role as a fertilizer defined as an element entering into the plant’s growth of its tissues for larger yields. But it also serves as a “soil amendment,” whereby the root hairs are maintained as highly calcareous, so called “semi-permeable” membranes. In that state, they are prohibiting the outgo of nutrients from plants back to the soil. Simultaneously they are fostering the ingo of nutrients–including nitrogen, phosphorus and potassium from the soil to the plant. If magnesium in excess on the clay will saturate the root membrane by replacing the calcium there, should we not interpret that as a need to saturate the immediate seedling zone with calcium? Such is the suggestion coming from many farmers in the areas on the map where magnesium is labeled as “high” in the drainage water.
Agricultural crops as well as citrus are telling us that magnesium is coming into more concern with our soil exploitation of both the organic and inorganic aspects of fertility. The larger look at this element includes the soils of the country as a whole, and the testing of soil for magnesium makes this the fourth, even for labeling of commercial fertilizer bags, though commonly connected with calcium under the term “lime.”
References Cited:
- R. R. Fudge, “Relation of Magnesium deficiency in grapefruit leaves to yield and chemical composition of fruit.” Florida Agr. Expt. Sta. Bul. 331. January, 1939.
- Frank M. Eaton. “Formulas for estimating leaching and gypsum requirements for irrigation waters.” Texas Agri. Expt. Sta. Misc. Pub. 1954.