Access to all articles, new health classes, discounts in our store, and more!
Imbalance of Vitamin B Factors Pyridoxine Deficiency Caused By Additions of Aneurin and Chalk
Published in British Medical Journal, March 31, 1945.
* * *
Pyridoxine Deficiency Caused by Additions of Aneurin and Chalk
In the clinical literature during recent years there have been numerous indications of a certain interrelationship or antagonism between various factors of the vitamin B complex. Pellagrins, after treatment with nicotinic acid, often show signs of beriberi or of ariboflavinosis (Spies, Vilter, and Ashe, 1939; Sebrell and Butler, 1938, 1939; Sydenstricker et al., 1940). There are recorded instances also of converse antagonisms. Thus Bichel and Meulengracht (1941) report a case of pellagra arising after treatment of the Plummer-Vinson syndrome with riboflavin (i.e., vitamin B2); and Salvesen (1940), Brændstrup (1940), and Lehmann and Nielsen (1939) all found signs of pellagra in patients who had been given large injections of vitamin B1. There is little doubt that in many of these cases the deficiencies in the diet were multiple, and it is assumed that successful treatment of the predominant deficiency with a single factor of the B complex showed up other deficiencies which had previously been masked. At the same time there is the possibility, in some cases at least, that secondary deficiencies were induced by the excessive dosing with one particular factor of the B complex, and that the unbalanced proportions of the different factors gave rise to a deficiency which was not present under the original conditions. Spies, Vilter, and Ashe (1939) made the significant remark, without apparently realizing its possible implications, that in pellagrins treated with nicotinic acid and continuing on their usual diets, the associated deficiencies (i.e., of B1 and riboflavin) often became worse.
One or two attempts have been made, but without much success, to induce symptoms of deficiency of one B factor by excessive dosing with another. Klopp, Abels, and Rhoads (1943) found a transitory increase in urinary excretion of riboflavin in some patients after administration of thiamine (i.e., B1), but could not induce either clinical or chemical evidence of riboflavin deficiency in these individuals even when large amounts of thiamine were given daily for 73 days. Unna and Clark (1942) could find no evidence of adverse effects on the growth of rats following administration of excess of individual vitamins in the presence of deficiencies of other vitamins of the B complex.
Effect of Overloading with B1
Recent experiments on rats in this Institute, however, have produced clear-cut evidence of the adverse effects that may be caused by a disturbance of the balance of the vitamin B factors in the diet, and have shown that overloading with one component, B1, can produce a definite deficiency of another component, B6. The experiments were designed to follow up a series of which the results were reported a year ago. In studies of the influence of the dietary factor on reproduction and lactation the results showed the beneficial effects on the breeding performance of rats of additions of chalk, dried yeast, and milk to a poor human diet (Richards, 1943). On the other hand, addition of pure vitamin B1 as aneurin, seemed to have a definitely harmful effect, as the weaning weights of the young rats were low and the mothers were in very poor condition after rearing their litters. It was therefore suggested that caution must be exercised in the addition of vitamin B1 to a poor diet, and the present experiments were planned to investigate this point more closely. Using a synthetic diet* as basal ration in place of the human dietary of the earlier experiments, vitamin B1 (as aneurin) was given at 3 levels–low, medium, and high–and these 3 levels were repeated in 3 additional groups which received also a supplement of chalk.
In the growth tests, which lasted from weaning till the rats were mated at about 115 days of age, the chalk group at each level of B1 intake showed a higher weight increase than the corresponding group without chalk, but these differences were not great. The groups receiving high B1 both with and without added chalk, were somewhat lower in weight than the corresponding groups on medium B1, but there was no noticeable difference between the animals in their general condition. Thus the growth tests, like those of Unna and Clark (1942), gave no very marked evidence of untoward effects arising from the variations in the diet. It is generally recognized, however, that a diet which may be reasonably adequate for growth, and even for reproduction, may not be adequate for successful lactation, and the lactation test in these experiments, even when the diet was improved by milk supplements, revealed differences between the groups which had been quite unsuspected from the growth test and the reproduction records. In certain cases the litters failed completely to survive to weaning; in others a few members of the litter survived, but were much below normal weight and in very poor condition; while in some instances 5 litters which were nearly normal in weight and apparently quite healthy suddenly showed the convulsive fits which enabled their condition to be diagnosed as pyridoxine (i.e., B6) deficiency. Chick, El Sadr, and Worden (1940) reported the occurrence of fits of an epileptiform nature in rats maintained for long periods on a diet deficient in vitamin B6. The seizures were characterized by hyperexcitability and circular running, tonic-clonic convulsions, and a comatose recovery period. Still more apposite, from our point of view, were the findings of Daniel, Kline, and Tolle (1942), who reported similar seizures in young rats while being nursed by mothers on pyridoxine-deficient diets, the symptoms appearing suddenly towards the end of the lactation period. The syndrome was “characterized by frantic running about the cage, accompanied by loud cries and convulsive seizures,” and the symptoms could be prevented or cured by pyridoxine supplements. Patton, Karn, and Longenecker (1944), who studied the incidence of sound-induced seizures, also recently reported the occurrence of spontaneous convulsions in young rats suckled by mothers maintained from parturition on pyridoxine-free diets.
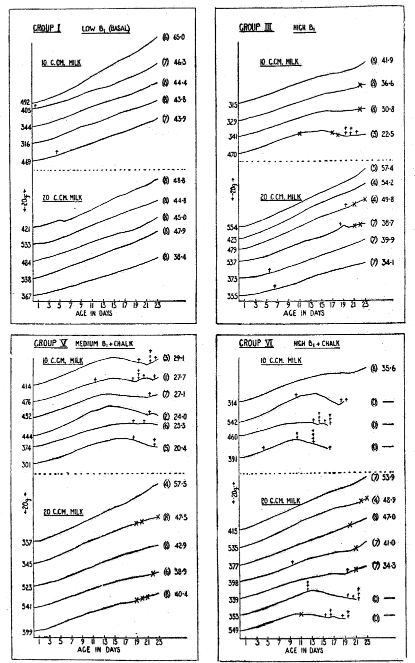
These descriptions fitted so exactly the symptoms observed in our young rats that there seemed to be little doubt that we were dealing with a conditioned pyridoxine deficiency, since the only variables in the diet were the vitamin B1 and chalk. Comparison of the groups showed that the lactation performance deteriorated as the level of B1 increased, and that conditions were made worse by the addition of chalk. At each level of B1 the group receiving chalk was worse than the corresponding group without chalk. Thus the best group was Group I–the basal group with low B, and no added chalk. The litters were in their approximately normal weight curves, whether the milk supplement was 10 or 20 c.cm., and convulsions were not observed in any of the litters. The worst group was Group VI, with extra chalk and high B1. In this group 10 c.cm. milk was quite inadequate to ensure successful lactation, only one litter surviving to weaning stage. In the section of this group receiving 20 c.cm. milk, 5 litters reached weaning stage, with approximately average weaning weight, but 6 of the 7 litters in the group showed the typical convulsions. In Group V, which had extra chalk with medium B1, 10. c.cm of milk again proved insufficient, although the performance was better than in the high B1+chalk group with 10 c.cm. milk. Representatives of 6 litters reached weaning stage, but there were many deaths during lactation, and the few survivors were low in weight and in poor condition. Convulsions were not observed in rats which were very puny and sickly. With 20 c.cm. of milk the lactation performance was again much improved. Deaths during lactation were reduced to zero, and weaning weights were approximately normal, but the characteristic fits were observed in 3 litters. Group III, with high B1 and no added chalk, was about equal in performance to Group V, with medium B1+chalk, and much better than the group with high B1+chalk. One instance of convulsions was observed in Group II, with medium B1 and no chalk, and two instances in Group IV, with low B1+chalk. The lactation performances of 4 of the groups are summarized in Chart 1, which shows the average weight curves of the individual litters from birth, and indicates the occurrence of convulsions. Groups II and IV are not included, as the weight curves showed little departure from normal.
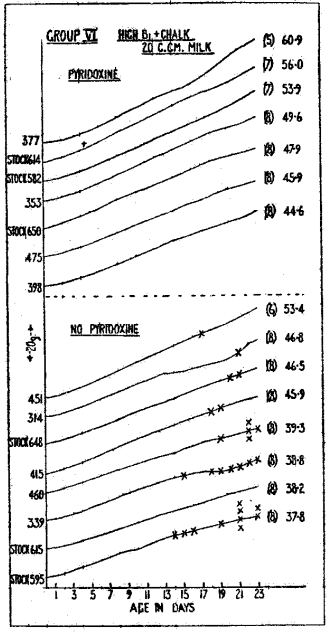
The breeding test was repeated, to obtain confirmation of these results and to test whether the onset of convulsions in the worst groups could be prevented by giving the does a supplement of pyridoxine from parturition. This supplement was given to half the does in Groups III, V, and VI. All rats in this test received a supplement of 20 c.cm. milk, since the 10-c.cm. supplement had proved insufficient in certain groups to bring the young to weaning and convulsions seemed to occur more readily when the young rats were fairly normal in weight. Chart II gives the results for Group VI, receiving high B1+chalk, from which it will be seen that the previous findings are amply confirmed. Seven out of eight litters showed the characteristic convulsive fits, several of them having repeated attacks. Although fits were not actually observed in the eighth litter, it seemed probable that they may have occurred (possibly overnight) when no observer was present, for it was noted that the general condition of the young in this litter at weaning was very similar to that of another litter in the group in which fits had been observed day after day. Moreover, the weaning weights of the young in these two litters were practically identical–38.3 and 37.8 g.
Chart I–Showing average weight in grammes per rat from birth to weaning for individual litters. At the left of each graph is the number of the doe. At the right of each graph in brackets, is the number of young reared. At the right of each graph, unbracketed, is the average weaning weight of the litter. ✝ indicates the death of a rat. x indicates the occurrence of fits.
Chart II. The weight graphs in the upper half represent litters from does which received a daily supplement of 40-y pyridoxine from parturition. The graphs in the lower half represent litters from does which received no pyridoxine. “Stock” indicates that the doe was a stock rat placed on the experimental diet two weeks before mating.
Pyridoxine Supplement.—Seven does in this group were given a pyridoxine supplement of 40 microgrammes daily from parturition, and none of their litters showed convulsive fits. The young progressed quite normally, and the average weaning weight was 49.7 g., as against 41.9 g. in the section of the group which received no pyridoxine supplement. In this experiment also the group with medium B1+chalk was approximately equal to that with high B1 and no chalk. In each of these groups 2 litters showed fits, and again not a single fit was observed in litters the mothers of which received pyridoxine supplementation.
It may be noted that a slight difference is observable through all the groups in this test when compared with the corresponding groups in the previous breeding test. Fresh supplies of flour, casein, and dried yeast had to be used in the repeat experiment, and it is unlikely that the new supplies would agree exactly with the old in their contents of all the B factors. A decrease in the B1 content of the basal ration, or an increase in the B6 content, or both, would tend to minimize the lack of balance of the B constituents, and the findings of the second test are consistent with such a difference in the basal ration. The lack of balance is somewhat less than in the first test, so that the occurrence of fits in any one group is less pronounced than in the same group of the previous experiment. Thus in Group III (high B1 with no chalk) only 2 litters showed fits, although 5 litters were affected in the previous test, and no instance of fits was noted in the second test in either Group II or Group IV. But the worst groups come in the same order as before, and the fits were entirely prevented in all groups when pyridoxine was given.
Comment
It seems abundantly clear, therefore, that pyridoxine deficiency was the cause of the symptoms observed, that this deficiency was induced by excess of vitamin B1 and by excess of chalk, and that the effect was enhanced when both vitamin B1 and chalk were present in excess. Patton and colleagues (1944), who used two pyridoxine-deficient diets in their experiments, one containing much higher amounts of thiamine, riboflavin, and pantothenic acid than the other, found fewer spontaneous seizures in litters from mothers receiving the smaller amounts of the B vitamins. This is in accordance with our findings in regard to B1.
While it is a deficiency of B1 that has become primarily obvious in our experiments, it seems likely that deficiencies of other B factors may also have been induced by the excess of B1. It happens that pyridoxine deficiency produces this spectacular effect in the young rats, and thus can be very readily recognized when other deficiencies may be overlooked. As a matter of fact, skin lesions, which developed during the mating and lactation periods in practically all the females of Groups V and VI, indicated the presence of some deficiency other than that of pyridoxine. The lesions of the extremities which are said to be characteristic of pyridoxine deficiency did not appear, but there was loss of hair on parts of the body, which might possibly be indicative of a B1 deficiency, and numerous body sores. The bald patches occurred mainly on the forehead and round the ears, and in some cases the underside of the body was practically denuded of hair. The body sores took the form of isolated septic spots, with loss of surrounding hair. György (1934) and others (e.g., Chick et al., 1935) have shown that if the diet of rats is deficient in both B2 and pyridoxine the florid dermatitis specific for B6 deficiency does not become evident until B2 is supplied. The amount of dried yeast in the diet was intentionally kept at a somewhat low level, to avoid masking any adverse effects of the additions of vitamin B1. It is thus possible that the supply of B2 I was suboptimal, and liable to be converted to a definite deficiency by excess of B1, just as happened with pyridoxine.
The high dose of vitamin B1 given in our experiments is, of course, far in excess of the amount likely to be found in any ordinary human diet, and the amount of pyridoxine in an ordinary mixed diet will probably be such that there is little risk of a deficiency being induced by the amount of B1 present. Danger may lie, however, in the present-day tendency to prescribe vitamin B1 somewhat indiscriminately, as a dietary adjunct and to give large doses of B1 orally or by injection in the treatment of various diseases. It is precisely in such cases, in which the patient is probably on invalid diet that is liable to be unbalanced, that a large excess of B1 may entail unexpected and dangerous results. In the case described by Brændstrup, for example, the patient, a chronic dyspeptic treated with ulcer diet, was given large injections of vitamin B1, totalling 220,000 i.u. in 3 weeks. When signs of pellagra developed, treatment with a preparation containing the entire B complex permitted recovery. Apart from the development of pellagra, untoward symptoms of various kinds have been recorded after dosing with vitamin B1. Steinberg (1938), who treated cases of chronic arthritis with large doses of vitamin B1, records that in a few patients vitamin B1 therapy caused “typical lesions of herpes zoster,” irritation of the peripheral nerve plates, and spasm of smooth muscle. The pain and irritation ceased when B1 therapy was withdrawn. Leitner (1943) also describes two cases in which injection of pure vitamin B1 over long periods produced unfavourable effects. In one case the symptoms resembled those of thyroid over-dosage, and included insomnia, headache, giddiness, and palpitation. It may well be, in view of our results, that such untoward effects are evidence of some vitamin deficiency induced by the large excess of the single vitamin.
Little is known as yet regarding human requirements of pyridoxine, but some results recorded by Spies, Bean, and Ashe (1939) are of interest. They found that 4 patients who had been treated successfully for pellagra and beriberi, but who remained on their deficient diets, complained of such symptoms as extreme nervousness, insomnia, irritability, abdominal pain, weakness, and difficulty in walking. All the symptoms disappeared within 24 hours of an injection of 50 mg. of pyridoxine. Later, Spies, Ladisch, and Bean (1940), studying the urinary excretion of B6 in human subjects, found indications of B6 deficiency in patients suffering from other clinical deficiency diseases. It is thus evident that pyridoxine deficiency can arise in man under certain circumstances.
It is becoming increasingly recognized that in the treatment of pellagrins with nicotinic acid it is essential to provide other members of the B complex and to prescribe a liberal and well-balanced diet. Our experiments would suggest the necessity for adopting a similar procedure for other B factors, and in particular, when B1 therapy is indicated, for supplying the whole B complex instead of the single vitamin. It was suggested in the previous paper (Richards, 1943) that the improvements effected in a poor human diet by means of such simple supplements as inorganic calcium, milk, and dried yeast provided a useful pointer for the post-war feeding of the starved populations in Europe. The present results emphasize the need for caution in any attempt to improve the diet of these populations by indiscriminate addition of large supplements of single synthetic B vitamins.
Notes:
* The synthetic diet was planned to be approximately equal in caloric value and Ca content to the original poor human diet, which contained a large proportion of white bread. It consisted of: white flour (untreated with chalk or aneurin) 1,020 g., commercial casein 400 g., dried brewers’ yeast 32 g., salt mixture (McCollum 185) 33.3 g., margarine 180 g., radiostoleum (containing 1 g. a-tocopherol acetate in 50 c.cm.) 2 c.cm., KI 0.00616 g., and MnSO4H2O 0.0616 g. In the groups which received extra calcium, chalk was added in the proportion officially recommended in making “fortified” white bread. A small amount of aneurin was added to the basal diet to make the B1 content equal to that of the original basal group in the human dietary experiments. In the groups with “medium” aneurin, the amount added was equivalent to the difference in B1 content between white and national wheatmeal flour, and to the “high”-aneurin groups 10 times this amount of B1 was given.
There were thus 6 groups in the experiment:
I. Basal + low B1
II. Basal + medium B1
IlI. Basal + high B1
IV. As I + chalk
V. As II + chalk
VI. As Ill + chalk
The Ca intake for Groups I, II, and III worked out at approximately 0.33 g per 1,000 Cal.. and for Groups IV, V, and VI at 0.81 g. per 1,000 Cal. The B1 intake for Groups I and IV was approximately 198 i.u., for Groups II and V 554 r.u., and for Groups III and VI 3756 i.u per 1,000 Cal. The B1, KI, and Mn supplements were added as dilute solutions daily to the liquid used in making up the ration, which was fed as a stiff paste. In the breeding tests, milk supplements were added to the diet–10 and 20 c.cm. per head respectively to half of each group in the first test, and 20 c.cm. per head in the second test.
References:
- Bichel, J., and Meulengracht, E. (1941). Wschr., 20, 913.
- Brændstrup, P. (1940). Læg., 102, 95.
- Chick, H., Copping, A. M., and Edgar, C. E. (1935). J., 29, 722.
- —-El Sadr, M. M., and Worden, A. N. (1940). Ibid., 34, 595.
- Daniel, E. P., Kline, O. L., and Tolle, C. D. (1942). Nutrit., 23, 205.
- György, P. (1934). Nature, 133, 498.
- Klopp, C. T., Abels, J. C., and Rhoads, C. P. (1943). J. Med. Sci., 205, 852.
- Lehmann, J., and Nielsen, H. E. (1939). Med., 1, 289.
- Leitner, Z. A. (1943). Lancet, 2, 474.
- Patton, R. A., Karn, H. W., and Longenecker, H. E. (1944). Biol. Chem., 152, 181.
- Richards, M. B. (1943). British Medical Journal, 2, 418.
- Salvesen, O. (1940). Med., 5, 279.
- Sebrell, W. H., and Butler, R. E. (1938). Hlth. Rep. Wash., 53, 2282.
- —-(1939). Ibid., 54, 2121.
- , T. D., Bean, W. B., and Ashe, W. F. (1939). J. Amer. Med. Ass., 112, 2414.
- —-Ladisch, R. K., and Bean, W. B. (1940). Ibid., 115, 839.
- —-Vilter, R. W., and Ashe, W. F. (1939). Ibid., 113, 931.
- Steinberg, C. L. (1938). J. Digest. Dis., 5, 680.
- Sydenstricker, V. P., Sebrell, W. H., Cleckley, H. M., and Kruse, H. D. (1940). Amer. Med. Ass., 114, 2437.
- Unna, K., and Clark, J. D. (1942). J. Med. Sci., 204, 364.