Access to all articles, new health classes, discounts in our store, and more!
Fragmentation and Scarring of the Tarsal and Metatarsal Bones: An Index of Dental Deformity
Read before the Second Annual Seminar for the Study and Practice of Dental Medicine, October 9, 1945. Reprinted from American Journal of Orthodontics and Oral Surgery, Vol. 32, No. 8, pp 486–515, August 1946.
* * *
In my previous paper,a l described our experience with feeding experimental animals diets which produced deficiencies in skeletal and organic development. I demonstrated that these animals returned deficient material to the soil, which in turn produced deficient plants. In these experiments, we were working with only a part of the cycle which governs growth and development.
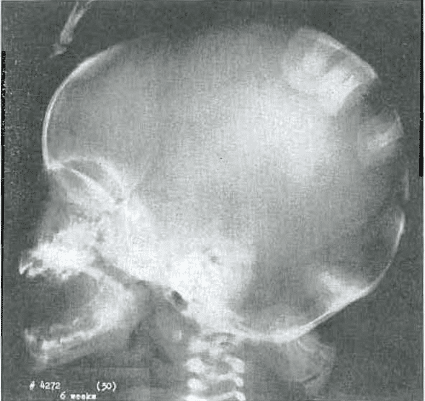
Fig. 1.—No. 4272(30). Skull of female, aged 6 weeks. Note mandibular protrusion and small middle third of face.
The entire cycle consists of soil to plant, to animal, and to man; then back to the soil. Primitive man of optimum development maintained this cycle. Anthropologists have pointed out that modern man has broken the chain, interfering with the regular homogeneous development of his kind. Price1 has made an extensive study of ‘modern primitives’ who have quickly shown degenerative changes on the food of civilized man. Wrench2 has studied the Hunzas with the same general conclusion. McCarriso3 has studied the diets of the men of India by feeding the identical foods to the rat, the pigeon, and the monkey. His results corresponded closely with those which we experienced with our own cats and rats.
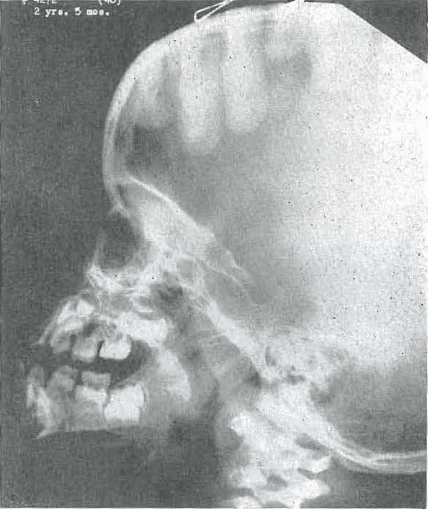
Fig. 2.—No. 4272(46) Skull or female, aged 2 years, 5 months. Note increasing prominence of mandibular protrusion with failure of base of skull and middle third of face to move forward.
We who are practicing medicine and you who are practicing dentistry are confronted daily with the problems brought about by modern methods of agriculture, food processing and preparation, and by the deficient health of parents. If we are to bring intelligent help to our patients, we must employ all of the available information on growth and development that is yielded by a study of the sciences dealing with the health of man.
In this paper, it is my purpose to show you that we have been able to learn a great deal about the effect of malnutrition on the human being from x-ray studies. Just as we found that the shape of the skull of the newborn kitten was an index of its future health, so have we found that the skull of the newborn babe is likewise an index to the future health of the child.
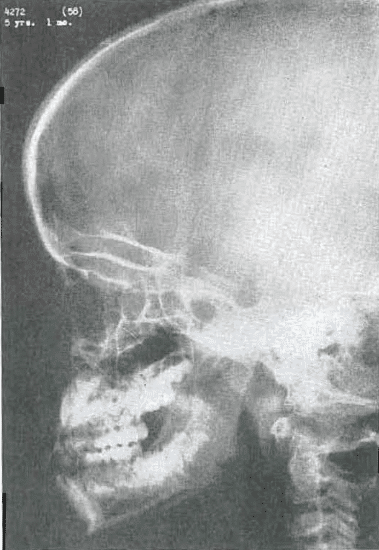
Fig. 3.—No. 4272(58). Skull of female, 5 years, 1 month of age. Mandible still prominent. Sphenoid sinuses beginning to pneumatized. Middle third of face still small. Frontal sinuses not yet developed. Dental alignment fair.
Todd4 called the skeleton the historian of the body. He pointed out that the scoring on the head of the radius and the long bones of the body represented metabolic upsets. In our own patients, we have found that the allergic child shows marked bone damage in the bones under stress at the time they are experiencing their most rapid growth. Fragmentation of the bones of the lower extremities occurs most commonly in the child in the period of life from 3 months up to approximately 6 years of age (Figs. 1 through 9).
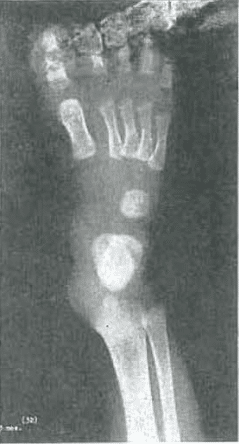
Fig. 4.—No. 4272(32). Foot of female, aged 3 months. Marked angulation and concavity of the distal end of metatarsals 2, 3, 4, and 5. Slight condensation of the internal trabecular structure of the cuboid.
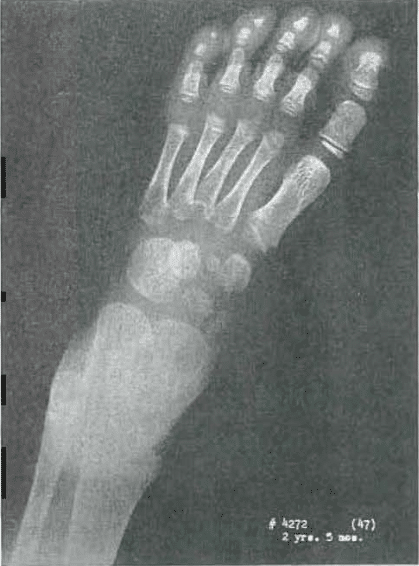
Fig. 5.—No. 4272(47). Foot of female, aged 2 years, 6 months. Slight fragmentation of cuneiform 1 and 3. Mild roughness of navicular, Some roughness and fragmentation of bases of metatarsals of 2, 3, 4, and 6. No epiphyseal head or metatarsal 6. Metatarsal 4 shows epiphyseal heads just appearing. Three is fragmented. Two is poorly developed. Proximal epiphysis of metatarsal 1 is rough.
The past few years, orthodontists have sought to correlate the structures of the mouth with some other structures of the body and to determine skeletal maturation. They have commonly elected to use the hand for this correlation. I propose to show you studies which will demonstrate that an ideal structure for the orthodontist to study in this connection is the foot.
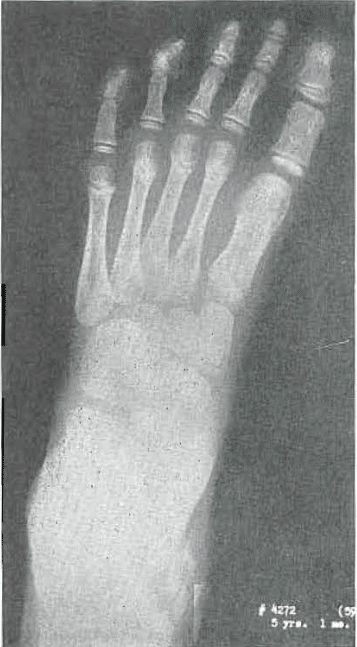
Fig. 6.—No. 4272(69). Foot of female, aged 5 years, 1 month. Lately healed. Slight condensation at base of metatarsals. A little roughness of cuneiform 1.
We routinely take x-rays of the lateral and anteroposterior views of the skull as well as x-rays of the hand and foot of each patient. In the child, we make a particular study of the foot. The bones of the foot in the infant and growing child to 6 years of age are under the stress of beginning to walk (Figs. 10 through 29). This corresponds with the time of the erupting and beginning of shedding of deciduous teeth. At this time the dental arches are expanding to make room for the permanent dentition, while the buds of all the permanent teeth, with the exception of the third molars, are underway. At the same time the heads of the metatarsal bones are developing, epiphyses are taking shape, and the general contours of the cuboid bone are being developed. The navicular bone is forming, and the three cuneiform bones are being laid down. It is also the period in which the bases of the metatarsals are smoothing out and are completing their articulation with the cuboid and cuneiform bones and with each other. They are reaching an oval shape, while the general outline of the tarsal bones is almost completed.
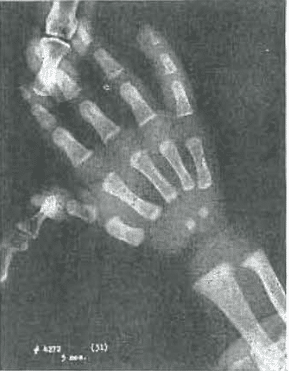
Fig. 7.—No. 4272(31). Hand of female, aged 3 months. Well-developed hand; compares favorably with Todd Standard 1. Slight roughness of capitate.
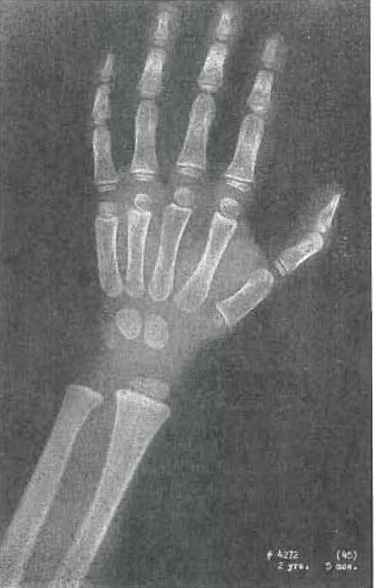
Fig. 8. —No. 4272(45). Hand of female, aged 2 years, 5 months. Slight condensation of trabeculae in capitate and hamate. Near Todd Standard 8, except lunate not yet seen.
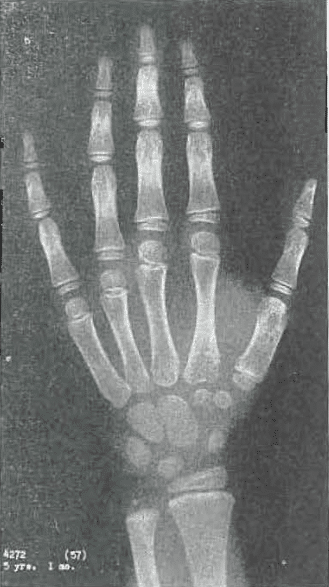
Fig 9.—No. 4272(57) Hand of female, aged 5 years, month. Development slightly ahead of age. Slight internal condensation of capitate and hamate. Compared with Todd Standard 14: 5 years, 9 months.
Though the foot is the easiest of all the structures of the lower extremities to study, the ankle, the knee, and the hip may show disturbances in growth equally well. Although a child may show fragmentation such as is shown in the accompanying illustrations, he may present no external evidence of this problem. However, he may have flat feet or may limp slightly for a time. Evidence of such a period of damage may occur in the ”normally” healthy child when he experiences an intercurrent infection such as measles or any of the usual illnesses of childhood. This child may heal the damage with no one the wiser unless x-ray pictures are made at the time.
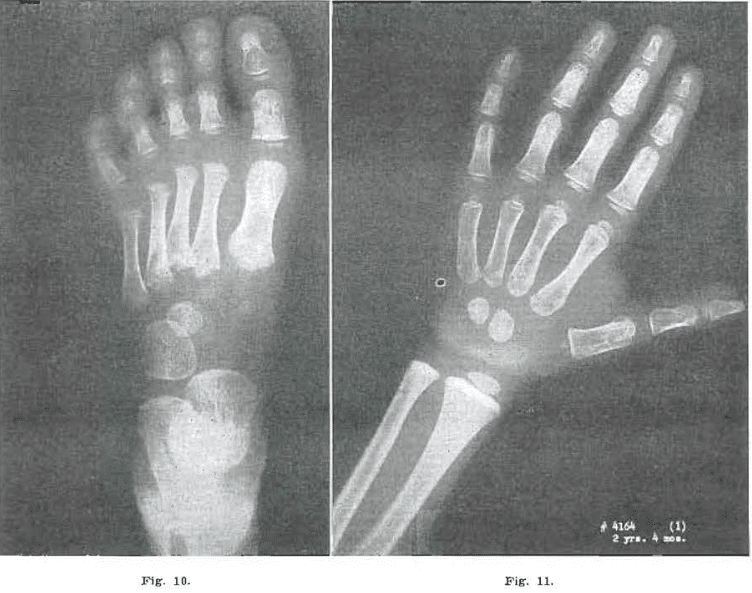
Fig. 10.—No. 4164(2). Foot of male, aged 2 years, 4 months. Marked fragmentation of the bases of the metatarsals of 2, 3, 4, and 6, and proximal epiphysis of metatarsal of phalanx 1. Navicular delayed. Cuneiform 1 slightly fragmented. Flattening and irregularity of the heads, and failure to develop of epiphyses of metatarsals 2, 3, 4, and 5.
Fig. 11.—No. 4164(1). Hand of male, 2 years, 4 months. Coarse trabeculation in all carpal and metacarpal phalangeal bones. Between Todd Standard 7 and Standard 8: 2 years, 3 months, and 2 years, 9 months.
The first evidence of fragmentation of the bones of the foot of the allergic child may be seen as early as the third month. It reaches a maximum between the third and fourth year. This fragmentation is early noticeable in the epiphyses and heads of the metatarsals, which may be delayed in appearance. Fragmentation of the bases of the metatarsals is often present, and the navicular, cuboid, and three cuneiform bones may be laid down with many fragmented centers of ossification. These tend to heal so that, by 6 years of age, only the scars resulting from fragmentation are present. These scars consist of a thickening or internal condensation of the trabeculae in the tarsals and the bases of the metatarsal bones. Later on, however, at about 10 to 12 years, as the epiphysis at the base of the fifth metatarsal appears, a new evidence of fragmentation in the foot may be seen (Fig. 38).
Fig. 12.—No. 4164(3). Foot of male, aged 2 years, 1 months, Note fragmentation or bases of metatarsals. Increased fragmentation of proximal head of metatarsal 1. Evidence of disturbance in mesial distal head of metatarsal 1; beginning fragmentation at head of metatarsal 1. Failure of heads to develop on metatarsals 3, 4, and 5. Cuneiform fragmented. Cuneiform 2 fragmented. Cuneiform 3 rough and fragmented. Some internal condensation.
During this entire period, with the exception of certain scarring in the carpal bones that is represented by thickening or condensation of the internal trabeculation, no appreciable evidence of bone damage is found in the hand. In spite of profound disturbances which may be seen in the tarsal and metatarsal bones, bone age delay in the hand does not necessarily follow.
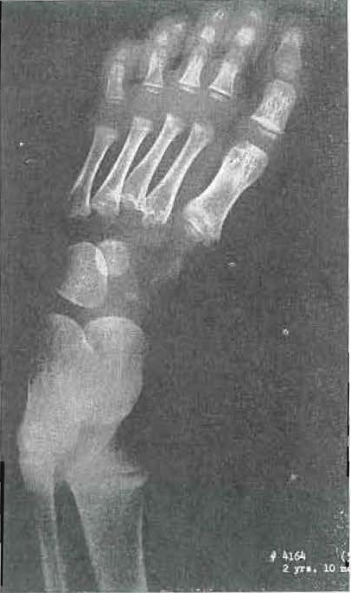
Fig. 13.—No. 4164(5). Hand of male, aged 2 years, 10 months. Essentially normal hand.
In the three cases which I am presenting, the carpal development of the patients is very close to the standards of Todd. When one finds evidence of fragmentation in the tarsal and metatarsal bones, it is only necessary to look at the face and into the mouth to see that other growth damage is also taking place. In some, it will be disturbance in growth of the middle third of the face alone. In others, it may be primarily a disturbance in expansion of the mandible. Some may have a combination of the two. Likewise, when evidences of severe metatarsal fragmentation are seen, one usually finds additional evidence of deficiency in the form of poor osseous structure and coarse trabeculation of the jawbone. ‘This same disturbance in the trabecular pattern is apparent in the osseous structure of the entire body as well as in the foot and the hand. If the orthodontist wishes to obtain x-ray evidence from other portions of the body to correlate with his studies of the jaw, he should examine a bone or group of bones that is under stress at the time the dental structures are developing.
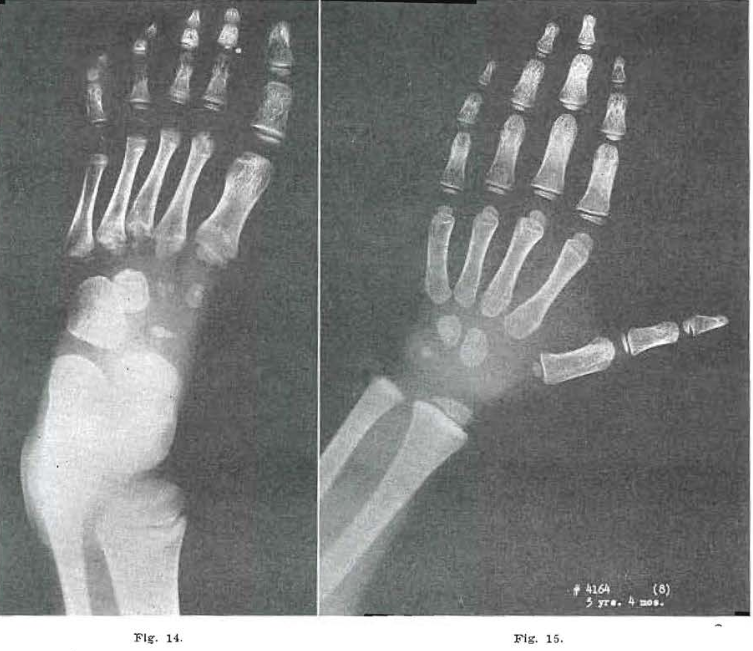
Fig.14.—No. 4164(7). Foot of male, aged 3 years, 4 months. Note extensive fragmentation of metatarsal bases continuing, with slight amount of healing of metatarsal. 5. Continued fragmentation of 2, 3, and 4. Appearance of metatarsal hands still delayed. Proximal epiphysis of metatarsal still fragmented. Metatarsal head of 1 shows increased condensation. Internal condensation noted in cuboid and third cuneiform. Fragmentation of cuneiforms 1 and 2, and navicular, still prominent.
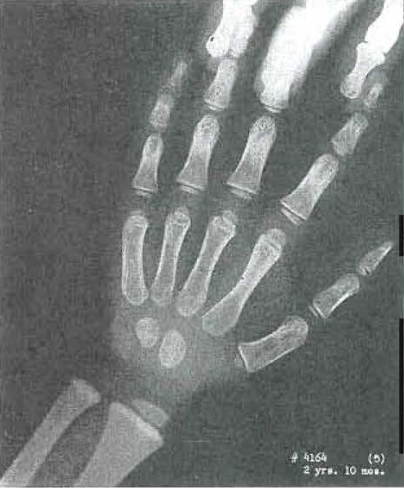
Fig. 15.—No. 4164(8). Hand of male, 3 years, 4 months of age. Capitate and hamate still show internal condensation and coarse Internal trabeculation of the bones. Otherwise essentially normal.
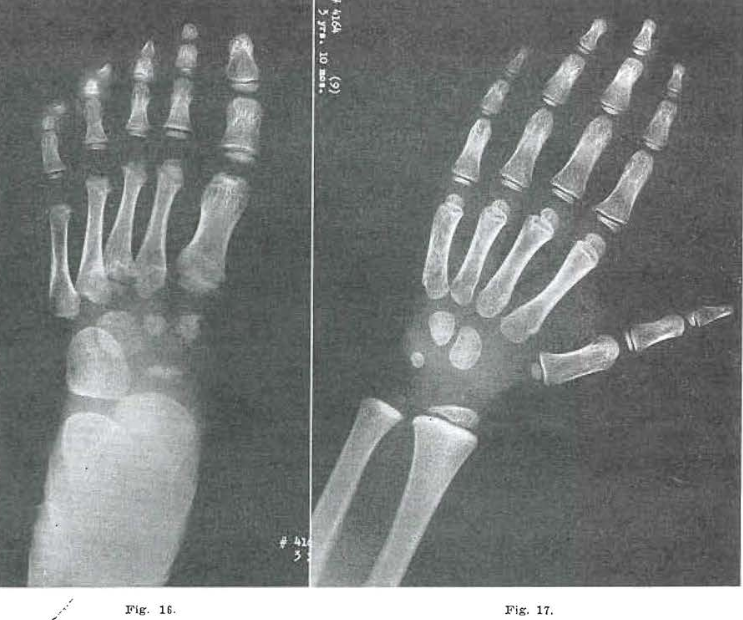
Fig 16.—No. 4164(11). Foot of male, aged 3 years, 10 months. Metatarsal 5 shows continued healing of the base. Healing just begun in metatarsal 4. Base of metatarsals 2 and 3 still clearly fragmented. Metatarsal head of 2 beginning to develop. Metatarsal 3 bifid; 4 and 5 still delayed. Frank osteochondrosis noted at the head of metatarsal 1, and proximal epiphysis still fragmented. Navicular and cuneiforms 1 and 2 still fragmented. Cuneiform 3 largely healed.
Fig. 17.—No. 4164(9) Hand of male, aged years, 1 months. Essentially normal.
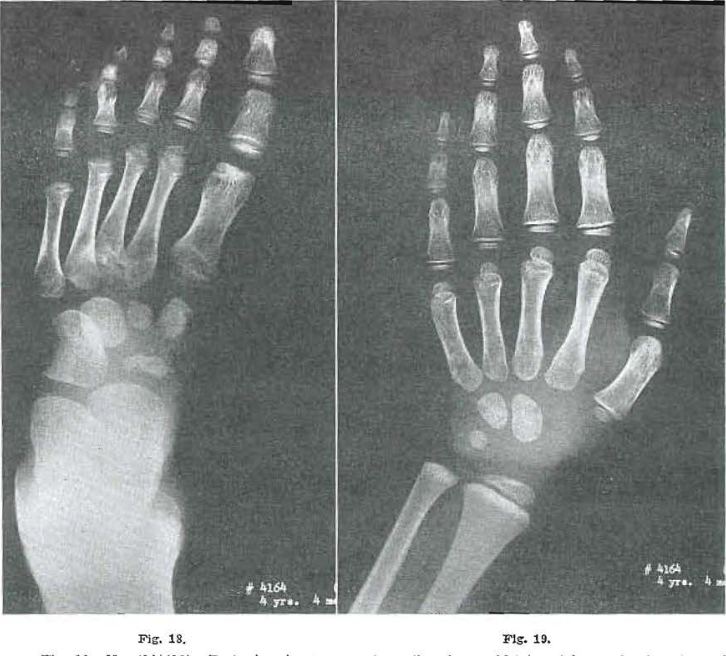
Fig. 18.—No. 4164(12). Foot of male, 4 years, 4 months of age. Metatarsal bases showing signs of healing. Greatest fragmentation noted in 3. Proximal epiphysis of 1 largely healed. Osteochondritis in the head of 1 continuing active. Epiphysis of 2, developing fairly normally. Epiphysis of 3 still bifid. Epiphysis of 4 appearing fragmented; epiphysis of 5 not yet present. Navicular and first cuneiform still fragmented; second cuneiform and third cuboid showing signs of healing.
Fig. 19.—No. 4164(14). Hand of male, aged 4 years, 4 months. Progress essentially normal.
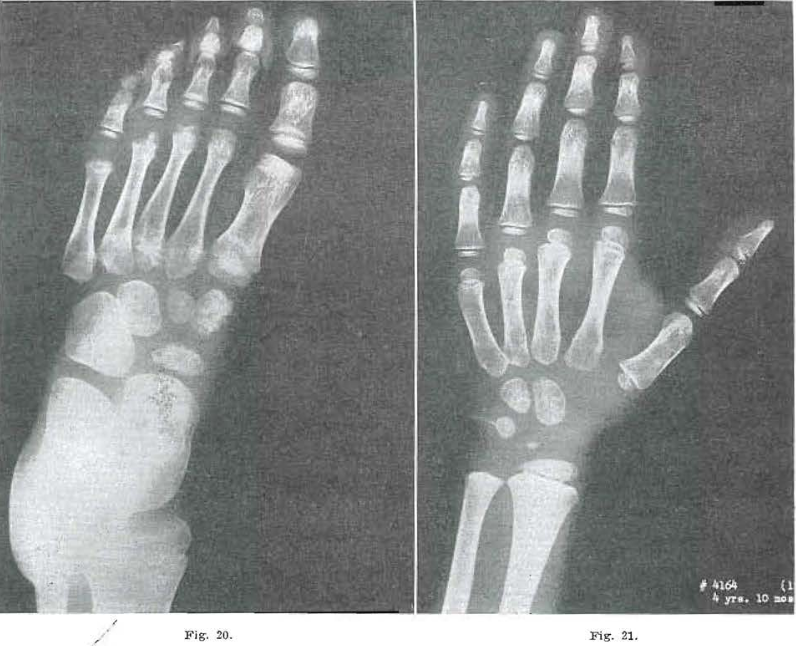
Fig. 20.—No. 4164(17). Foot of male aged 4 years, 10 months. Bases of 4 and 5 essentially healed. Note internal condensation in bases. Two and 3 still healing with internal condensation evident. Proximal epiphysis of 1 largely healed. Distal portion of 1 shows osteochondritis still present. Epiphysis of 2 fairly well developed; 3 still bifid. Four came in as hemiepiphysis. Five not yet present. Second cuneiform almost healed. Navicular and first cuneiform healing. Internal condensation in all distal bones.
Fig. 21.—No. 4164(15). Hand of male, 4 years, 10 months of age. Condensation of capitate and hamate, Todd Standard 12: 4 years, 9 months.
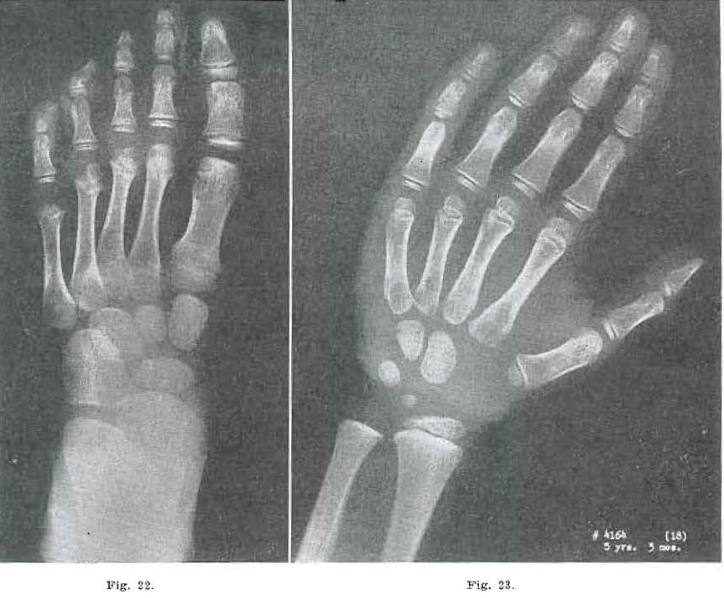
Fig. 22.—No. 4164(20). Foot of male, 5 years, 3 months of age. Bases of all metatarsals healing. Showed considerable condensation. Osteochondritis of first metatarsal healing. Epiphysis of second essentially normal. Epiphysis of third slightly bifid; head of 4 still a hemiepiphyseal; 5 just beginning. Navicular and first cuneiform still rough. Second and third cuneiform and cuboid show internal condensation.
Fig. 23.—No. 4164(18). Hand of male, aged 5 years, 3 months. Essentially normal. Todd Standard, 5 years, 3 months.
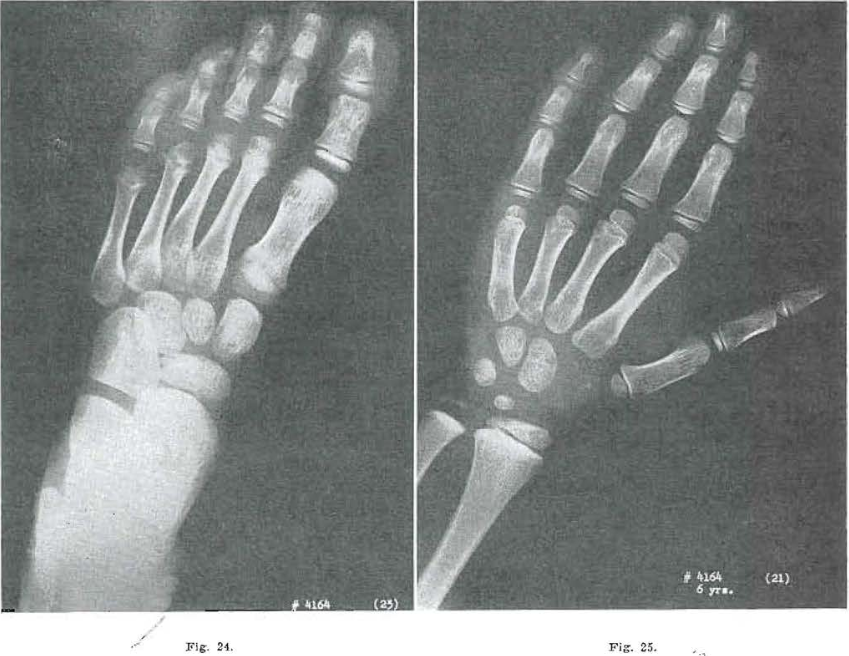
Fig. 24.—No. 4164(23). Foot of male, aged 6 years. Navicular and first cuneiform still slightly rough. Marked in thermal condensation in all anterior tarsal bones. Marked internal condensation in bases of metatarsals. Head of metatarsal 1 shows osteochondritis nearly healed. Epiphysis on second metatarsal well developed. Third still partially developed; fourth less well developed than third, and fifth is fragmented.
Fig. 25.—No. 4164(21). Hand of mate aged 6 years. Internal condensation of capitate, hamate and triquetrum in evidence. Essentially normal development.
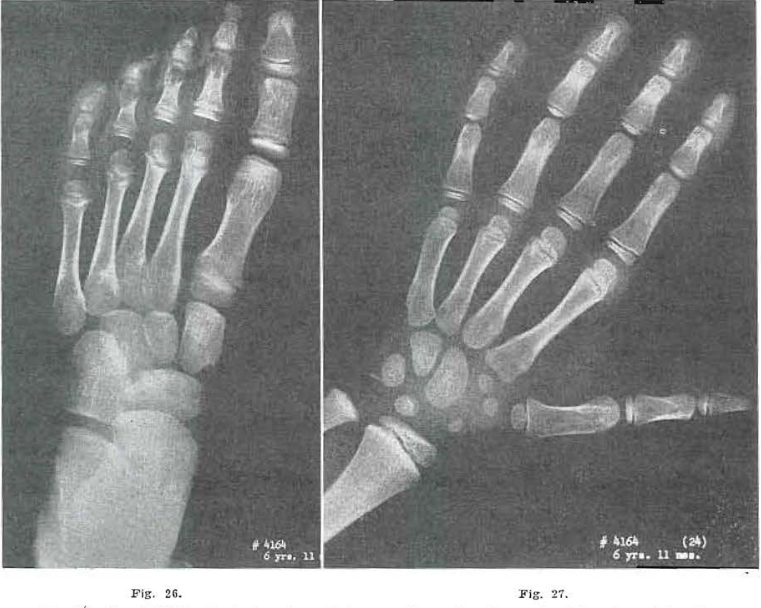
Fig. 26.—No. 4164(26). Foot of male, aged 6 years, 11 months. Bases of metatarsals and tarsals essentially the same as in Fig. 25. Head of 1 shows improvement and ossification. Two and 3 fairly well developed. Four is improving and is beginning to develop.
Fig. 27.—No. 4164(24). Hand of male, aged 6 years, 11 months. Essentially normal.
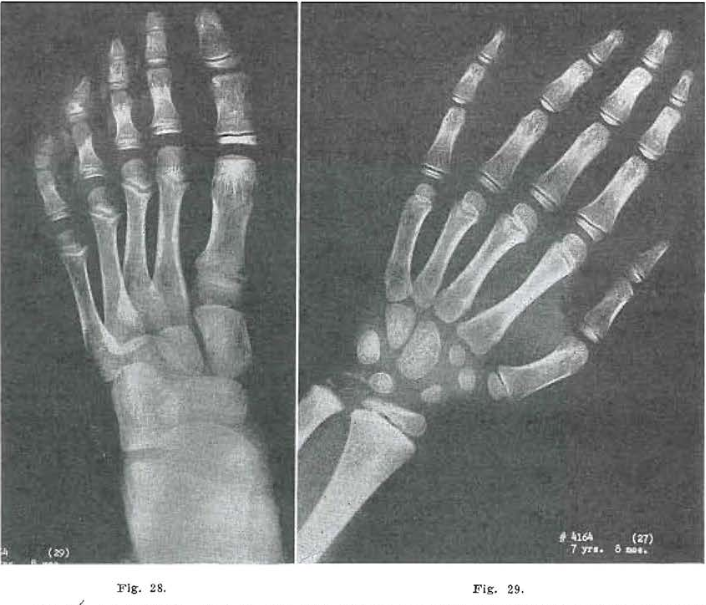
Fig.28.—No. 4164(29), Foot of male, age 7 years, 8 months, now essentially normal except for internal condensation in the cuneiform, cuboid, and navicular bones, and the bases or the metatarsals. Slight evidence of healed osteochondritis in the first metatarsal head end flattening of the fifth metatarsal epiphysis.
Fig. 29.—No. 4164(27). Hand of male, aged 7 years, 8 months. Compares favorably with Todd Standard for 7 years, 9 months. With the exception of internal condensation shown in the capitate, hamate, triquetrum, and lunate, the hand is essentially normal.
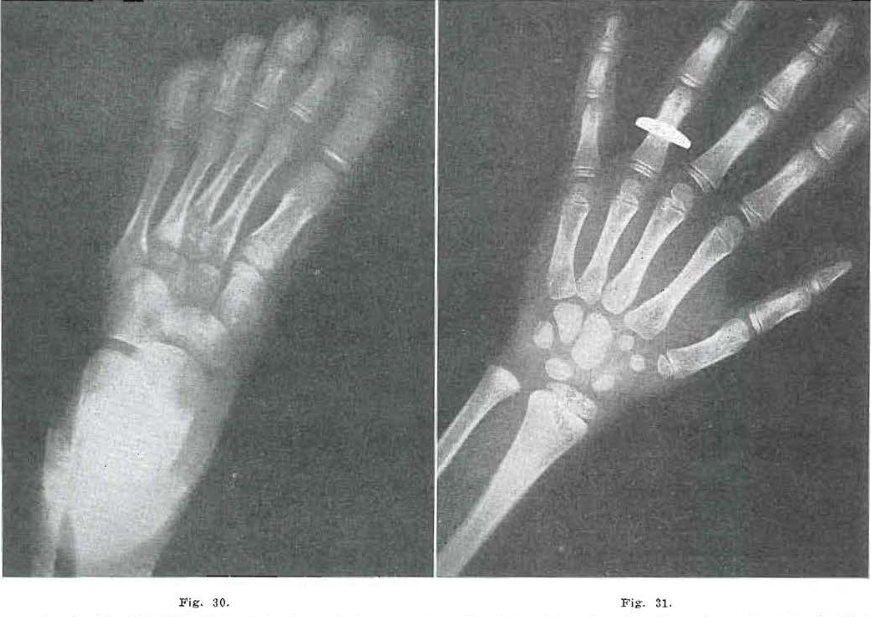
Fig. 30.—No. 4209(62). Foot of female, aged 4 years, 11 months. Notice internal condensation in base of metatarsals, third cuneiform, navicular, and cuboid. Third cuneiform is apparently not quite healed.
Fig. 31.—No. 4209(60). Hand of female, aged 4 years, 11 months. Interior of capitate hamate and triquetrum shows condensation of trabeculation suggesting evidence of old injury. Bone study suggests Todd Standard 14: 5 years, 9 months.
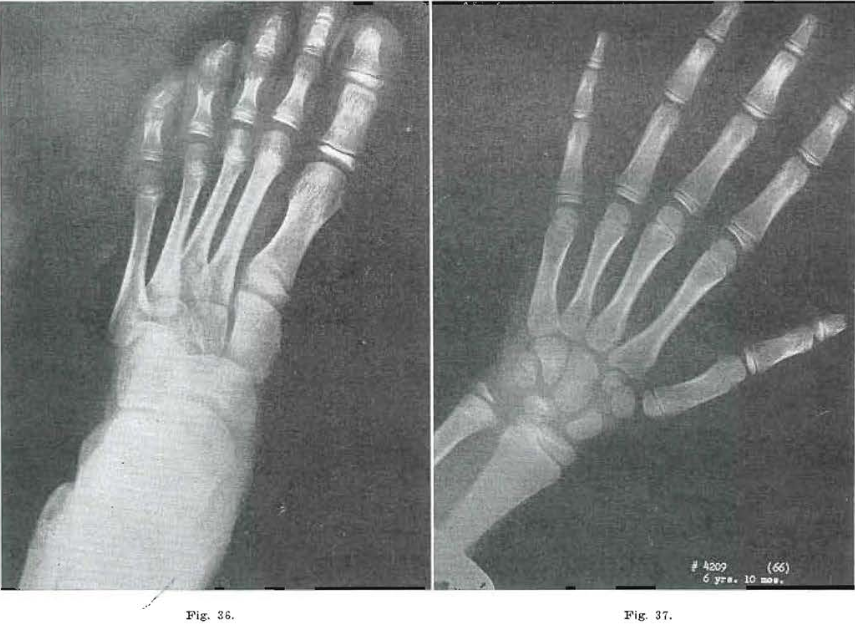
Fig 32.—No. 4209(65). Foot of female, aged 5 years, 9 months. Same scars noted as in Fig. 30.
Fig. 33.—No. 4209(63). Hand of female, aged 5 years, 9 months. Same bones show condensation as in Fig. 31. Coarse trabeculation. Todd Standard 16: 6 years, 9 months.
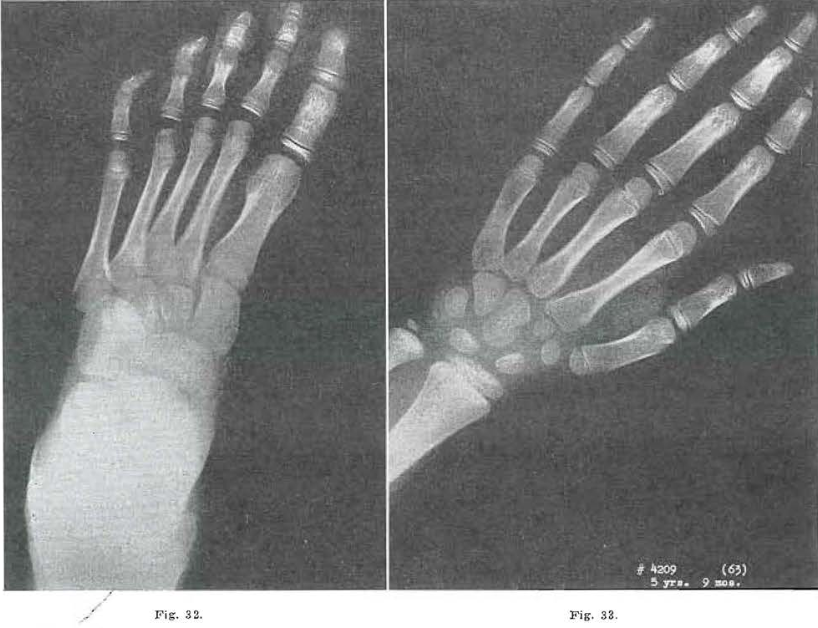
Fig. 34.—No.4209(68). Foot of female, aged 6 years, 10 months. Same as Fig. 32, except for growth.
Fig. 35.—No.4209(66). Hand of female, aged 6 years, 10 months. Sane scars noted as in Fig. 32. Todd Standard 18: 7 years, 9 months.
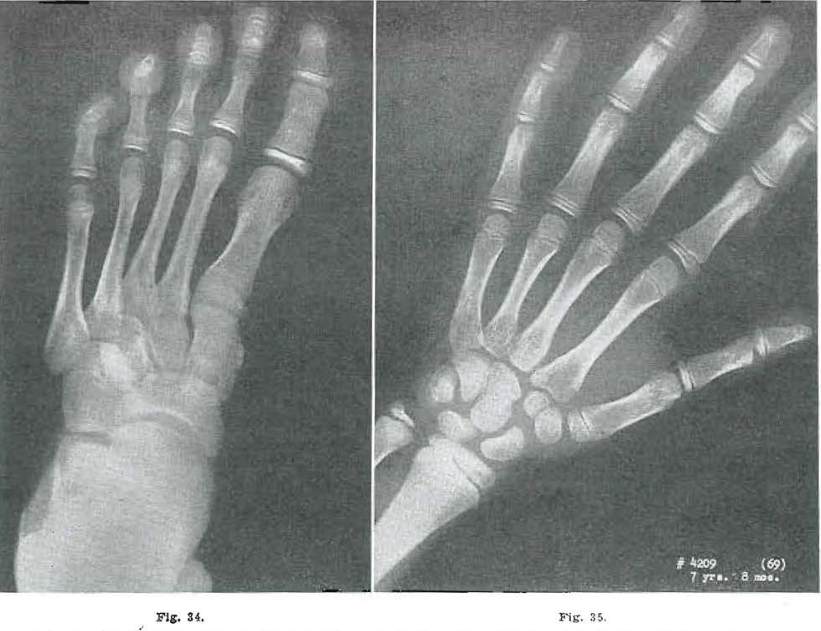
Fig. 36.—No. 4209(71). Foot of female, aged years, 8 months. Further increase in size. Pattern course.
Fig. 37.—No. 4209(69). Hand of female, aged 7 years, 8 months. Same. Very coarse trabeculation. Todd Standard 20: 8 years, 9 months.
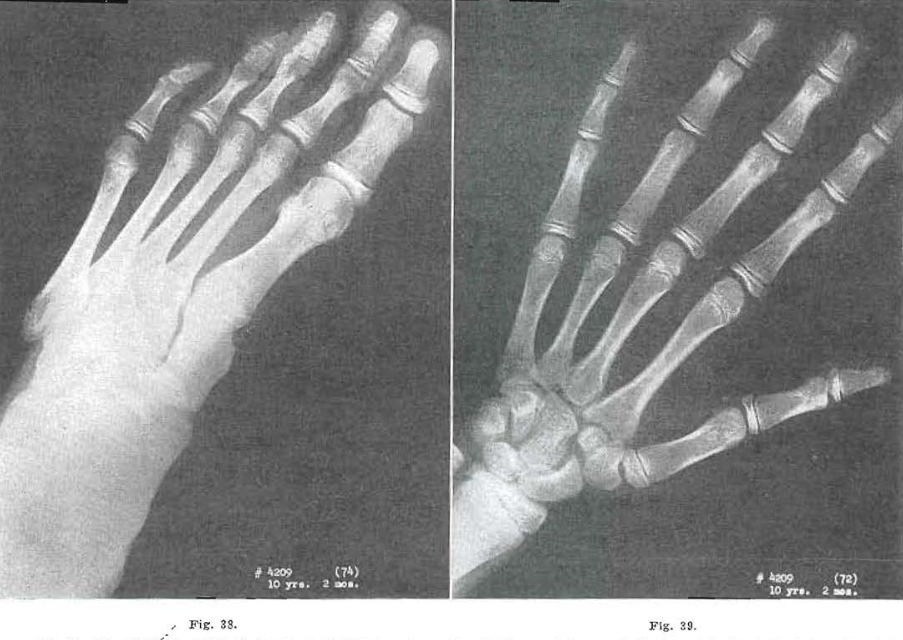
Fig. 38.—No. 42094(74). Foot of female, aged 10 years, 2 months. Evidence of fragmentation in epiphysis of the tuberosity of the fifth metatarsal. All other sears carried on.
Fig. 39.—No. 4209(72). Hand of female, aged 10 years, 2 months, Evidence of internal condensation in metacarpal bases and all carpal bones. Todd Standard 25: 11 years, 3 months.
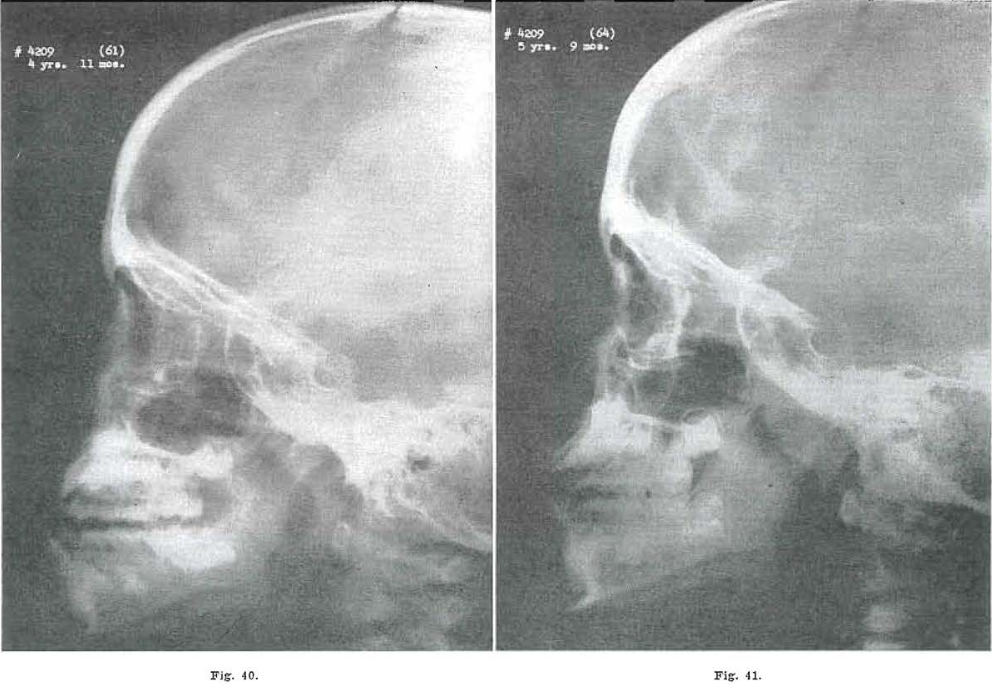
Fig. 40.—No. 4209(61). Skull of female, aged 4 years, 11 months. Failure in forward movement of middle third of the face and base of skull, giving an apparent mandibular protrusion
Fig. 41.—No. 42094(64). Skull of female, aged 6 years, 9 months. Middle third and base of skull showing forward movement.
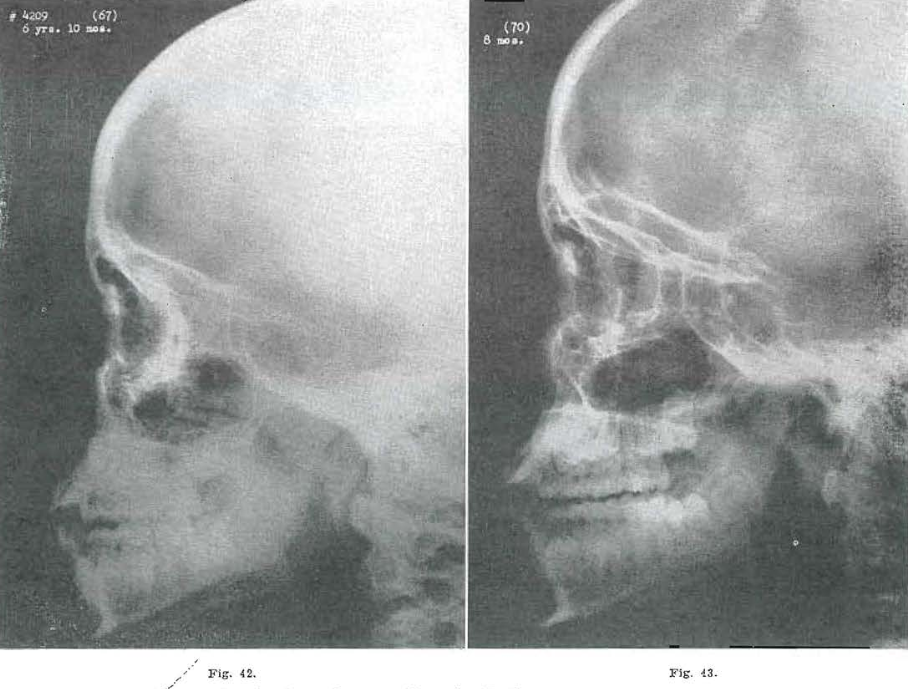
Fig. 42.—No. 4209(67). Skull of female, aged 6 years, 10 months. Further progress.
Fig. 43.—No. 4209(70). Skull of female, aged 7 years, 8 months. Malar prominence moving forward. Glabellar prominence beginning to develop.
The scars that are laid down in these bones during periods of fragmentation are apparently carried on into adult life. We have been able to follow evidences of this scarring as long as seven years after the process has largely healed (Figs. 30 through 46). In this one instance, we are showing you the process five years after the healing has taken place. We have not been studying the patients long enough to carry any single individual from infancy to adult life, so as to be able to say conclusively that a particular scar on a particular bone is evidence of a specific fragmentation that occurred in infancy. We have observed, however, that those of our adult patients who show dental and facial failures commonly acknowledged to be developmental in nature exhibit scars in their bones which are similar to those we have followed in our children.
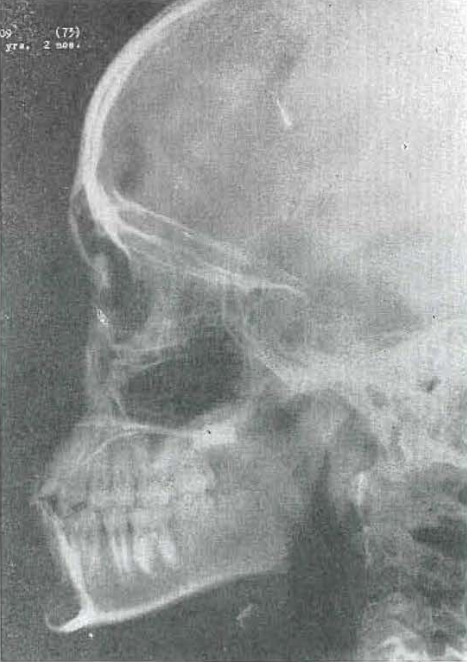
Fig. 44.—No. 4209(73). Skull of female, aged 10 years, 2 months. Notice changing angle of upper teeth. Good alignment of teeth. Good forward projection of face and base of brain case. Series shows increase in growth of middle portion of face in respect to mandible.
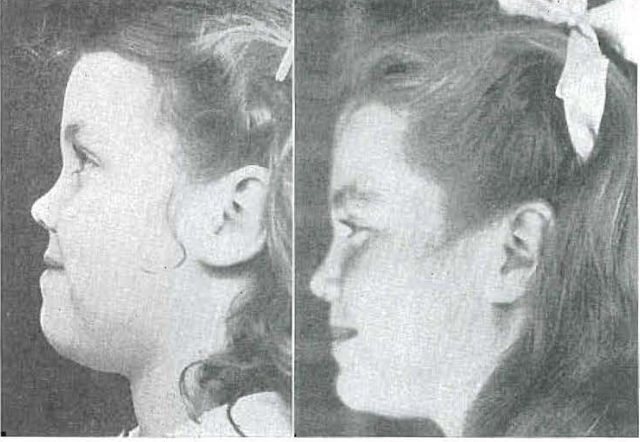
Fig. 45.—No. 4209. Lateral views of patient at 4 years, 11 months of age, and 10 years, 2 months of age, showing changes in facial configuration and tissue stability.
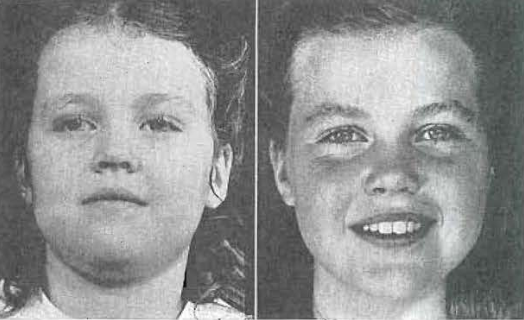
Fig.46.—No. 4209. Frontal view of patient at 4 years, 11 months of age, and 10 years, 2 months of age.
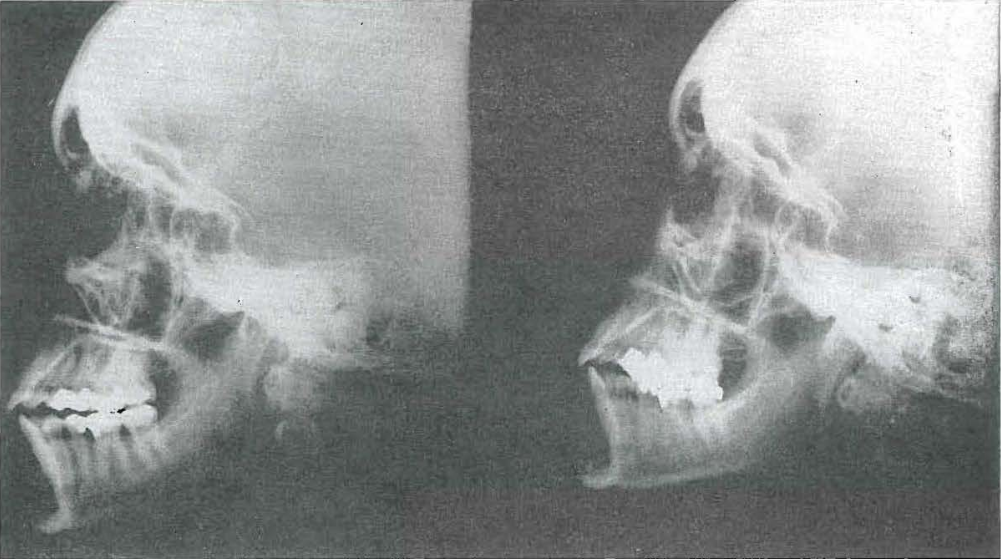
Fig. 47.—No. 4491. Skull of female aged 32 years, before improvement in dietary (left), and three years later (right), after giving birth to two children. Note changes in skull configuration and laying down of scar bone.
When one states that these processes leave their scars even into adult life, he is apt to give the impression that he feels the individual is foredoomed to developmental failures that will follow him throughout his life. It is my impression at this time that it is impossible to remove the scars themselves. It is impossible to change the developmental patterns of bones laid down. However, if the proper forces of growth and development are applied in sufficient amount, material change in trend of growth can be obtained.
In the accompanying x-rays and photographs (Figs. 30 through 46) I am showing you a child who, at the age of 5 years, had a disfiguring mandibular protrusion. No orthodontic treatment was administered to this child, but the results obtained can be seen in photographs of her at the age of 10 years. She had been placed upon a vital diet rich in growth and developmental factors. She was encouraged to use her facial structures, by chewing tough objects, and by making faces. At the age of 4 years and 10 months, she had a profound mandibular protrusion; at the age of 10 years and 2 months, this had largely been corrected by the development of the middle third of the face.
I have selected this as representative of a group of children in which I have seen improvement in facial development due solely to improvement in intake of growth-promoting factors.
It is possible for the adult to rebuild calcium in his bones by consuming adequate quantities of growth-promoting factors. Relative efficiency of all the organs of the body can also be increased noticeably.
It seems that the new bone laid down in this process is not the original bone that was present before the damage occurred, but is a sear bone, as is shown clearly by x-ray (Fig. 47).
Regarding the development of the orbital and maxillary arches, we find the same factors at work in the human being as we found experimentally in the cat. I our allergic children we have found a narrowing of the transverse diameter between the malar prominences, a narrowing of the transverse diameter between the lateral orbital prominences, failure in the forward movement of the malar prominence; all of these, or a combination of any may occur in an individual.
Muscle tone is largely controlled by factors similar to those that control growth and development. The density and strength of bone is determined by its calcium content and the muscle pulls that are exerted on it. These, in turn, are controlled by the factors of growth and maturity. It is not just the chewing on rawhide that develops the face of the Eskimo and it is not just the consumption of large quantities of raw fish, blubber, and entrails that produces his well-developed features, hut it is the combination of them all, plus a firm inheritance that has not been interfered with in the ancestry of these individuals.
Every structure of the body, including the generative organs, is involved in the developmental process. However, from the standpoint of the dentist, if he is to correlate the findings in the jaw with other osseous structures, he will find a parallel in the x-ray of the foot, which is an organ under stress during the critical period of growth and development of the dental structures of the child.
aThe Effect of Heat-Processed Foods and Metabolized Vitamin D Milk on the Dentofacial Structures of Experimental Animals, AM. J. ORTHODONTICS AND ORAL SURG. (Oral Surg. Sect.) 32:467, 1946.
Editor’s note: Since the era in which this article was written, society’s understanding of respectful terminology when referring to societal and cultural groups has evolved, and some readers may be offended by references to “primitive” people and other out-of-date terminology. However, this article has been archived as a historical document, and so we have chosen to use Pottenger’s exact words in the interest of authenticity. No disrespect to any group is intended.
References Cited:
- Price, W. A.: Nutrition and Physical Degeneration, New York, 1939, Paul B. Hoeber, Inc.
- Wrench, G. T.: The Wheel of Health, 1938, The C. W. Daniel Co., Ltd.
- McCarrison, Robert: Studies in Deficiency Diseases, Oxford Medical Publications, 1921.
- Todd, T. Wingate: Atlas of Skeletal Maturation, St. Louis, 1937, The C. V. Mosby Co.
- Pottenger, F. M., Jr.: The Importance of a Vital, High Protein Diet in the Treatment of Tuberculosis and Allied Conditions, Bull. Am. Acad. Tuber. Physicians, 5: 55-60, 1941.