Access to all articles, new health classes, discounts in our store, and more!
Calcium, Its Activation, Utilization and Metabolism
Read before the Seventh International Dental Congress, Philadelphia, Pa., August 25, 1926. Published in The Journal of the American Dental Association, April 1928.
* * *
Few problems of biologic science have been more difficult of understanding than the processes by which the teeth and bones are formed. Some of the factors involved are coming to be understood. In this paper, I shall discuss some of the newer phases.
In previous communications, I have presented data which relate to some of the fundamental conditions. Evidence was presented which relates the type of reaction in bone, as it appears with different expressions in different individuals, in such a manner as to make possible the dividing of these individuals into groups on this basis, and further which indicates that these factors are related to defense and to susceptibility to disease. These data have been reviewed.1
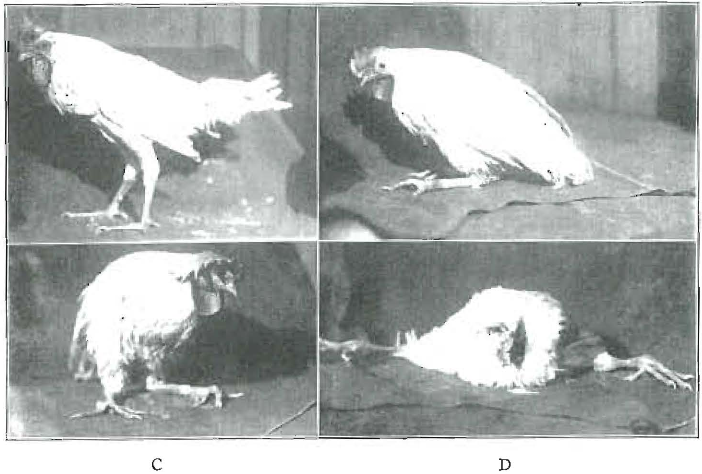
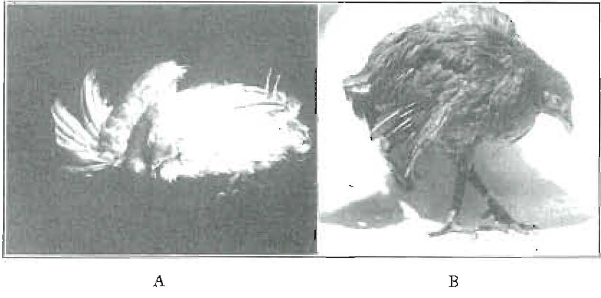
Many workers have presented data relating radiant and certain vitamins to calcium utilization. Outstanding among the workers in this field have been Mellanby, Hess and Steenbock. The bibliography is now too voluminous to recite. I have used more than 1,000 chicks in the last fifteen months in studies on this problem. These studies have brought out many important new data. The beneficial effects of radiant energy are readily demonstrated by many experiments. Figure 1 shows the progressive weakening of the legs of a chick deprived of radiant energy while on a diet low in phosphorus and the antirachitic factor, though ample in calcium. The control, at the upper left, A, received the same diet, with sunshine. B, C and D show progressive weakening of the legs, so that the chick came to complete prostration. A similar chick, so prostrated, is shown in Figure 2A. The same chick, a few weeks later, is shown in B, after having cod-liver oil added to the deficiency diet. There was the same absence of radiant energy. The legs are deformed, as will be noted, though they were progressing toward normal. A splendid illustration of the change in this chick is shown in three roentgen-ray views of it which appear in Figure 3. A shows the chick when down; B shows it thirty-three days later than at A, after it had been for that period on the original diet but with cod liver oil added, and C, taken thirty-one days later than B, shows the continued improvement in calcification. We are here directly concerned with factors which are involved in these processes, primarily because the same general factors obtain in the development of both tooth and bone structure. That the utilization of calcium proves not to be so simple a process as has been generally anticipated is abundantly demonstrated. Its presence in food does not necessarily provide that it become available for the body’s use. Certain chemical activators must be provided in the system. These same processes obtain with regard to the formation of teeth, and since teeth do not have a circulation comparable to that of bone, the reconstruction of imperfectly organized tissue is not provided by Nature. Enamel, once formed, undergoes but slight change. We are accordingly greatly concerned as a dental profession regarding these forces which have to do with tooth formation.
Fig. 1. Progressive weakening of the legs of a chick deprived of radiant energy and on a diet low in phosphorus and the antirachitic factor. The control (A) had the same diet but occasional sunshine.
Fig. 2. Two views of the same chick on a diet low in phosphorus and deprived of ultraviolet radiation. A, chick completely prostrated with typical weak legs. B, the same chick a few weeks later, after having cod-liver oil added to the food. The leg deformity may be noted.
Fig. 3. Roentgenographic views of the chick shown in Figure 2. A, appearance when the chick was prostrated; B, thirty-three days later; C, thirty-one days later than in B. Increasing calcification is evident.
The dentistry of the past has been largely concerned with the treatment of disease. We have come to the time in dental science when adequate programs should be worked out which will be competent to prevent in large measure the dental disease which has made dental repair necessary. These studies are, therefore, primarily concerned with preventive programs.
Parathyroid Glands
The animal body is marvelously provided with monitors to supervise great fundamental phases of vital function. These are spoken of as the glands of internal secretion. Without some of these, life cannot exist, and all are needed for efficient normal life. Two small glands in the neck, about as large as peas, the parathyroid glands, are, chiefly, the monitors for calcium metabolism. Several glands influence calcium metabolism, but with most animals the elimination of no other glands than the parathyroid produces death from resulting lowered calcium of the body fluids. After their removal, the animal loses weight, serum calcium goes down from an average of 10 to 11 mg. per hundred cubic centimeters of serum to 5, 6 or 7 mg., at which time the animal becomes very nervous and usually goes into convulsive spasms, called tetany, which may last several hours or but a short time, usually terminating in death. If, prior to death, there is injected into the animal some extract of parathyroid glands, the animal is restored and will continue to live in practically normal health provided a little of this extract is injected every day or two, but continuously. When the parathyroid glands are removed from rats, a change can be seen under the microscope in the structure of the growing teeth in as short a period as five days, and, in fifteen days, is marked. These continually growing incisors readily break off when a little strain is put on them, when their extrusion in normal growth processes has brought the point of weak structure within the zone of stress. The functioning of the parathyroid glands is essential for the maintenance of the quality of the blood which we may speak of as the solubility factor for calcium. The maintenance of the functional capacity of these glands seems to be directly related to certain activating substances which reach it through the circulating mechanisms of the body. When cholesterol, a lipoid in which the skin and many of the body tissues, particularly the brain, are rich, is exposed to ultraviolet light or sunlight, it has the property of aiding the body in metabolizing calcium. Certain fats, particularly butter and egg yolk, contain substances capable of activating the metabolism of calcium. Cod-liver oil has this property in a concentration approximately 400 times that of butter.
It is an interesting fact that the Eskimos of the far north, where the light is so restricted, live largely on sea food, particularly the fats of fishes, during the long winter. It is remarkable how the developing civilization has created what we may term an instinct. For example, the people living on the Isles of Lewis, north of Scotland, which are shrouded in fog and cloud, have a practice of taking as part of the diet for each day at least one stuffed cod’s liver. Children are started on this ration in very early life. When our explorers have gone into the north seas for the winters, and have tried to subsist through the long period of darkness on canned foods, flavored to tickle the palate, they have developed disease to which the Eskimos about them, whose diet they would scorn, are immune.
Since the ingestion of cholesterol unactivated by irradiation has no apparent effect on calcium metabolism, while irradiated cholesterol has a marked effect, an extract from it being capable of preventing rickets, in the amount of one one-hundred-thousandth gm. daily in a rat, it seems, in the light of the accumulating evidence from several workers, to be one of the important factors in the activation of the body for the metabolism of calcium. Its activation takes place normally through the skin, through the agency of the ultraviolet light from the sun. It has been assumed that, since artificial sources of ultraviolet produce greater concentrations of ultraviolet radiation than does the sun, these means can be used to displace sunlight or at least to compensate for it. Last year, I presented data1 indicating that overexposure of fatty substances to ultraviolet light produces distinctly poisonous effects. Increasing information strongly indicates that ultraviolet radiation, from artificial sources such as the mercury vapor lamp, is not a substitute for sunlight though it may be an important adjutant. It is probable, from researches at present, in that means for modification of progress, the artificial sources to limit them to the proper ranges will make them competent substitutes.
Since the best sources of stored-up activator known at present is cod-liver oil, it has become important to consider carefully its advantages and disadvantages. In the first place, it is disagreeable for many people to take. There is some evidence that long continued taking of large doses has distinctly undesirable, if not harmful, effects There is considerable difference in the relative and actual value or efficiency of different samples of oil. This probably is not the fault of the source of supply, but probably relates, in part, to the difference in the time of the season during which the fish are caught.
The normal source of activator for most mammalian forms is the maternal milk.
Calcium Activation
Since calcium may be present in ample quantity and not be utilized by the body, it becomes very important to determine what means may be available for enhancing the body’s capacity to utilize the food. While it is known, in a general way, that certain procedures, such as exposure to ultraviolet, the administration of cod-liver oil or the eating of fish fats and oils, will greatly increase the body’s capacity to utilize calcium, there are as yet few data regarding the mechanisms by which these substances act. In order to throw light on this phase of the problem, I have been carrying on extended studies regarding the effects on animals and humans produced by the administration of various substances. In order to determine these effects, it is, of course, necessary to take samples of blood. The taking of one or two or a small number of specimens may be relatively unimportant as far as changes produced by the hemorrhages are concerned. When these are carried over an extended period of time, the loss of blood becomes an important factor in the change in the blood picture. It not infrequently occurs that individuals who have had extractions have either continued seepage or recurring hemorrhage with considerable loss of blood. There have been no data as to the effect of such loss on the calcium of the blood. As a preliminary, therefore, to determinations of the effects of medication on the calcium levels, it has been important to determine the effect of frequent hemorrhages on these same calcium levels. Rabbits have been used, chiefly, and the blood has been taken by perforating the marginal ear vein and, after a little massaging with a towel, which relaxes the capillaries, the blood drops quite freely so long as the vein is compressed centrally to the lesion. The bleedings have been in general carried out as follows:
Once on the late afternoon of one day; hourly, beginning at 8 a. m. the next day, and continuing until 8 or 9 p. m., and once early the following day. When medication was given, it followed the second taking of blood, after the first morning sample, so that the control period of two readings on different days was had with at least fifteen hours intervening, and then a series of thirteen successive hourly observations with one or two on the following day (sometimes still later) to determine the projection of the influence.
To determine the three factors–total calcium, diffusible calcium, and non-diffusible calcium–by the older methods, and allow for duplicate readings, about 5 c.c. of blood is required. As this clearly means too great a strain on even a large rabbit, most of the determinations have been made on half this quantity by using specially designed micropipets. In a rabbit weighing approximately 3 kg. (nearly 7 pounds), the hemorrhages were produced hourly for thirteen successive bleedings of approximately 5 c.c. each, which makes a total of approximately 65 c.c. of blood. The total weight of the blood of this animal would be approximately one thirteenth of the body weight, or 230+ gm. We would, therefore, remove within the thirteen hours approximately 25 per cent of the total blood. A much larger percentage of blood than this can be removed without serious permanent injury to the animal, although it takes a considerable period of time for complete recovery. Chickens can have approximately 75 per cent of the total blood removed and show little effect from the operation, with a relatively early recovery.
There are two purposes in this part of the determination: (1) to know the effect of hemorrhage on calcium levels, and (2) to ascertain the correction that should, therefore, be made for determining the change in calcium level which exists from medication when no hemorrhage is produced.
Since successive hemorrhages produce a regular and continuous decline in total calcium, it is particularly significant that our observations of the calcium levels of medicated rabbits are too low by an amount approximately equal to the depression which would exist as a result of the number of hemorrhages preceding that determination. We would, therefore, show in the charts both the observed calcium levels and the calculated calcium levels by inserting the progressive correction needed to compensate for the hemorrhages.
In the rabbit referred to above, the total calcium decreased progressively with the increased number of hemorrhages and toward normal overnight after the last hemorrhage on the day before. In a chart referring to this case, diffusible and non-diffusible curves cross each other five times in that length of time. This throws important light on the mechanism for readjustment and the factors disturbed by hemorrhage. This represents a rather extreme condition, since the quantity of blood removed at each reading is relatively large, and the total correspondingly considerable in proportion to the total blood of the body. When smaller quantities of blood are taken, the change in calcium levels is approximately proportionate.
There has been relatively little basis for judgment as to the size and frequency of dosage for either calcium carrying foods or medicaments, so-called activators. A general procedure in administering the medicine has been on the presumption that the effect is extended until that supply is exhausted, and, therefore, relatively larger doses taken less frequently would be quite comparable to smaller doses taken more frequently. Important new light is thrown on this phase of the problem by these studies. We read in the literature of calcium compounds, such as calcium chloride and calcium lactate, being administered to patients in doses of several grams. The fact that the administration of calcium has frequently been tried but has not been deemed of considerable helpfulness, and further, that it is not in general use, is probably ample evidence as to its lack of efficacy, though it does not throw light on whether the lack of efficacy is due to lack of knowledge regarding the method of administration or the presence of negligibly helpful if not distinctly harmful effects.
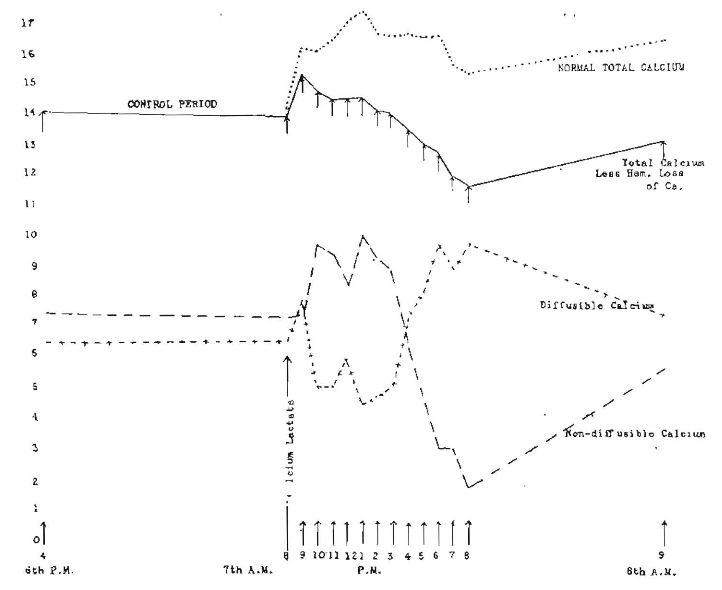
We will, therefore, consider first the giving of one relatively large dose of calcium lactate to a rabbit. There have been two reasons for selecting the rabbit for these observations: (1) it is desirable because of the ease of securing blood without an anesthetic or need for heart puncture, and (2) the rabbit has a recognized lack of sensitiveness to change of calcium levels (of all the milk of mammals whose blood has been tested, the rabbit’s carries the highest concentration of calcium.) In Figure 4 will be seen the effect of the administration to a rabbit of 2 gm. of calcium lactate in a single dose, and the blood determinations made hourly. It will be noted that the blood total calcium increased the first hour from 13.98 to 15.35 mg. per hundred cubic centimeters of blood and, by the second hour, had fallen to 14.78 and maintained a level of approximately from 14.48 to 14.58 for the next three hours. There was then a progressive decline to 11.65, the last reading that evening being 11.65. On the following morning, the observed calcium was 13.10. Since this rabbit lost 5 c.c. of blood at each bleeding, we shall presume that there was a progressive loss from the first corresponding with that change in the last figure from hemorrhage alone. When, therefore, we add this amount to the observed total calcium, we find a very differently shaped curve as shown in the dotted line, above the total, the maximum increase reaching ing. at the end of five hours, and decreasing to 1.13 at the end of twelve hours, with an increase of 2.19 the following morning. Were we considering only total calcium, and were it the only important consideration, we might consider that the evidence justified this type of treatment. When we study the actual change in diffusible and non-diffusible, and their proportions, we observe that the non-diffusible has been depressed at the end of twelve hours to the tremendously low point of 1.93 mg. In other words, nearly all the available calcium has become diffusible. Since the diffusible and non-diffusible calcium should presumably balance, the diffusible in normal usually exceeding the non-diffusible by about 10 per cent, it will be observed that we have created a serious imbalance between these two calcium factors. If we consider this as a negative instance, we shall probably see somewhat of the reason for the serious depression which individuals realize after taking calcium lactate alone, without an activator.
Fig. 4. Hourly changes in three calcium levels following one administration of 2 gm. of calcium lactate at 8 a. m. (Rabbit 1403).
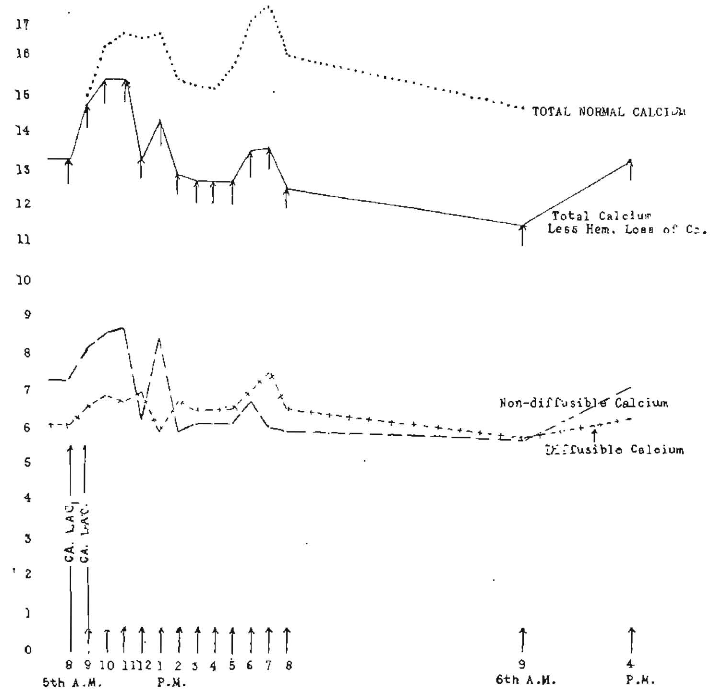
In order to further study this particular phase, we have repeated these same doses in one hour as shown in Figure 5. The observed total calcium line is found to be flattened in its first rise and experiences a second rise followed by a third rise. The correction for total calcium is also shown in the dotted line by adding the amount hour by hour that had been observed in the control rabbit as being due to succeeding hemorrhages. Since hemorrhage itself produces an oscillation between diffusible and non-diffusible calcium, and since two successive doses brought succeeding positive and negative phases in such a relation as to, in part, neutralize each other, we do not have the extreme oscillations shown in the single dose.
Fig. 5. Hourly changes in three calcium levels following administration of 2 gm. of calcium lactate at 8 and 9 a. m. (Rabbit 1356).
About two weeks later, an effort was made to repeat the observation on this rabbit (1356) by adding to the calcium lactate activated cholesterol. The rabbit was not in good condition for either of the tests, probably owing partly to its age. A few hours after the administration of the calcium and activated cholesterol, it was observed to be depressed, and the hemorrhages were terminated. It died sometime during the following night, probably owing to an increase in the total calcium level and the disturbance of the calcium balance by a too extensive and too rapid procedure.
Collip has shown that, by the administration of parathyroid extract, the total calcium can be increased markedly and that a too great increase caused death. I showed in my report of last year that, by the administration of freshly made parathyroid extract, we were able to double the total calcium level of a rabbit in six hours, at which time the rabbit died. It seems probable that this dose was sufficient to increase so greatly the total calcium and so disturb the distribution of the calcium as to cause death. This would be equivalent to a dose of 40 gm. for an individual weighing 160 pounds, which is clearly out of all proportion. However, it accomplishes our purpose of determining the need for knowledge regarding dosage and the method for its administration.
In order to study further the relation of quantity and distribution of dosage, we have administered calcium lactate alone, and with various activators in distributed small doses in order to determine the effect on calcium levels.
In the following five charts will be seen the results of the administration of raw cod-liver oil alone, activated cod-liver oil alone, raw cod-liver oil and calcium lactate, activated cod-liver oil and calcium lactate, and activated cholesterol and calcium lactate.
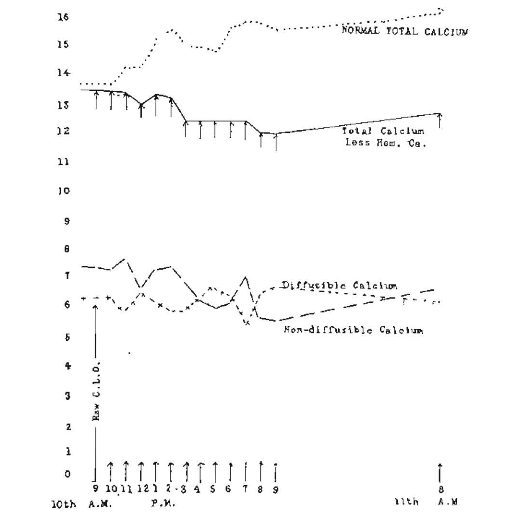
Figure 6 (rabbit 1404) shows the effect on the calcium levels from the administration of 2 c.c. of raw cod-liver oil to this rabbit. The total calcium level at the beginning of the test was 13.36 mg. per hundred cubic centimeters of serum. The line marked “Total calcium less hemorrhage calcium” shows a slight depression below the starting level. The dotted line marked “Total calcium level” is the same calculated to correct for the depression due to successive hemorrhages as previously discussed. There was a sustained increase of from 1 to 2 mg. throughout the period of the experiment. A conspicuous difference in this chart from some of the others is the narrowness of the oscillations between the diffusible and non-diffusible calcium.
Fig. 6. Hourly changes in three calcium levels following administration of 2 c.c. of raw cod-liver oil at 9 a. m. (Rabbit 1404).
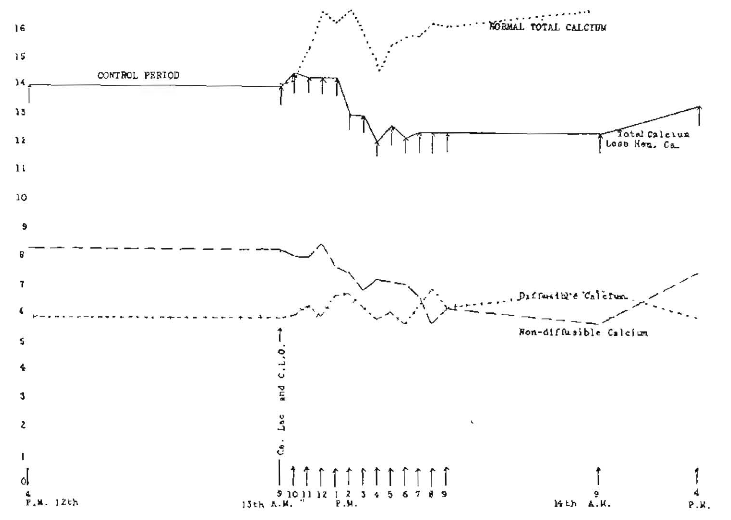
This is strikingly unlike the results produced by the administration of calcium lactate alone, which are particularly conspicuous in Figure 7 (rabbit 1419), showing the hourly changes following the administration of 5 grains of calcium lactate and 0.5 c.c. of raw cod-liver oil hourly for six consecutive hours. The solid line shows total calcium less the hemorrhage loss of calcium, and notwithstanding the progressive loss from hemorrhage, the calcium increased over 2 mg., approximately 20 per cent, in the first three hours. The hourly administration started at 9 a. m. and ended at 2 p. m. Immediately after the cessation of the administration, there was a tremendous increase in diffusible calcium with a corresponding decrease in non-diffusible, the latter reducing to 1.16. Having tried a somewhat similar experiment on myself, I know somewhat of the sensation that is produced by throwing the ratio of diffusible and non-diffusible out of adjustment. The body makes an heroic effort at readjustment. There is an interesting phase of this which I am discussing in an extended text on this subject, one tremendously significant, since it is so often a part of the terminal picture in the bacterial overwhelming from toxic material from infection sources. The upper dotted line marked “total normal calcium” is the calculated total by making the correction for the progressive depression of calcium due to loss from hemorrhage in taking the specimens of blood.
Fig. 7. Hourly changes in calcium levels following administration of 0.5 c.c. of raw cod-liver oil and 5 grains of calcium lactate hourly for six consecutive doses (Rabbit 1419).
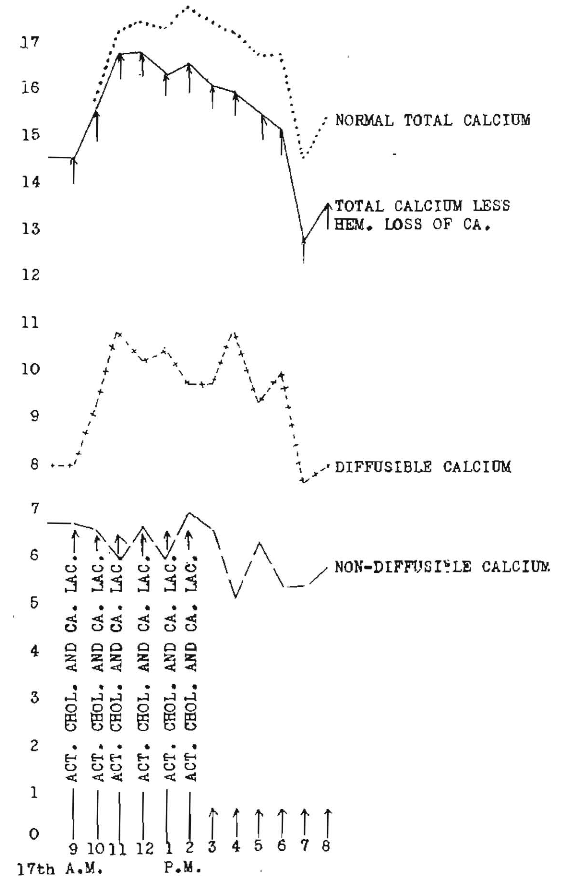
Figure 8 (rabbit 1376) shows the hourly changes in three calcium levels following the hourly administration of 2 grains of calcium lactate and 2.5 gm. of activated cod-liver oil. There is a control period of seventeen hours preceding the test, and one following, checking the recovery. The striking characteristics are: the stabilizing effect of the activated cod-liver oil on diffusible and non-diffusible calcium which I have shown to oscillate under certain treatments very strikingly. There is a sustained increase in total calcium as corrected for hemorrhage loss. It must be understood that the extreme results produced here emphasize the necessity for great care not to overdose and also emphasize the great importance of the progressive increasing or pyramiding effect produced by the continued administration of large doses.
Fig. 8. Hourly changes in three calcium levels following one administration of 2 gm. of calcium lactate and 2.5 gm. of activated cod-liver oil at 9 a. m. (Rabbit 1376).
Figure 4 (rabbit 1403) shows the hourly changes in the three calcium levels following one administration of 2 gm. of calcium lactate at 8 a. m. The control period over night shows the three calcium levels under consideration approximately stationary. The striking effect is that the diffusible calcium rises during the first hour almost precisely in accordance with the total calcium. This is followed by a sharp reversal which is apparently Nature’s method of reestablishing an equilibrium between these factors. At the eighth hour, there is a reversal of the positions of the diffusible and non-diffusible, and the pendulum swings far in the opposite direction to that which obtained during the preceding seven hours. This is a striking illustration of the inadequacy or deficiency of calcium lactate alone. This large dose increases total calcium to a point shown by the correction for the loss due to hemorrhage over 2 mg. above the normal level.
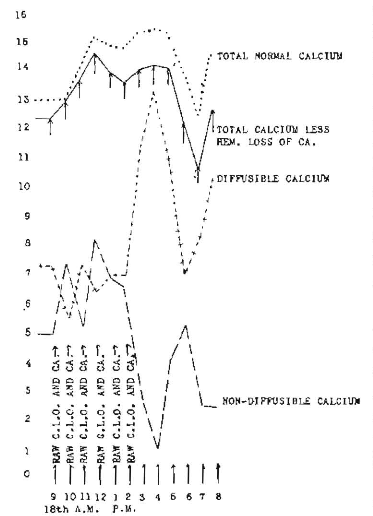
In striking contrast to this is the result obtained by the administration of calcium lactate hourly, and cholesterol activated and dissolved in olive oil, the latter not activated, which is shown in Figure 9 (rabbit 1418). It will be seen that there is much less oscillation between the diffusible and non-diffusible. They have retained their general relationship, without reversal, the diffusible at all times being greater than the non-diffusible. When we correct for the loss due to hemorrhage, we find that we have an increase in total calcium from 14.50 at the start to 17.65 at the maximum, which is 3.15 mg. increase, or approximately 22 per cent.
Fig. 9. Calcium changes following six hourly administrations of each 33 grains of cholesterol activated for ten minutes under a quartz mercury vapor arc lamp and given in olive oil, and 5 grains of calcium lactate (Rabbit 1418).
There is a distinct relationship between the size of the dose and the length of time before its effect appears in the blood. As shown previously, large single doses given to rabbits show their maximum effect in from one to two hours.
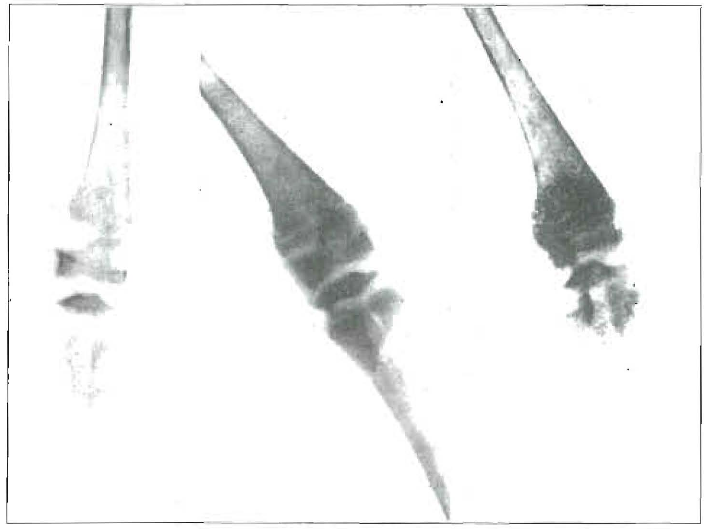
A striking illustration of the practical application of this general principle is shown in Figure 10, which presents three views of the second joint of a chicken. A shows the condition after, the chicken had been on a deficiency diet (Steenbock No. 2966), plus such sunshine as was available in the winter time through the smoke of Cleveland. I have recently reported at length on this phase.2 The rubbing of cod-liver oil on to this chicken’s leg made a marked difference in as short a time as twenty days in its ability to use the leg. The change is also marked as shown in the roentgenogram in B. C shows a marked change in the following six days, when the cod-liver oil was added to the chicken’s diet, plus its being applied to the second joint. This strongly emphasizes the marked influence of the activators on animals which are being fed a deficiency diet at a time and under conditions when normal sunshine is not available.
Fig. 10. Left: Effect of deficiency diet plus sunshine through city smoke. Effect of rubbing cod-liver oil on leg, as shown twenty days later. Right: Effect of adding cod-liver oil to diet, as shown six days later.
Milk as a Source of Activator
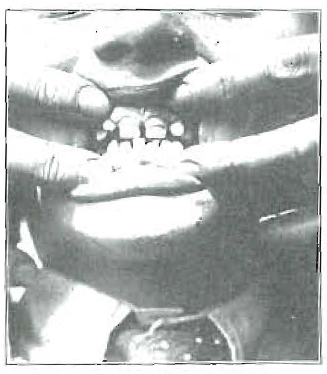
We have looked on milk as being adequate for providing the needs of young life, whether furnished by the mother or by another species. In using milk from other origins as a source of activator as well as of calcium, it has generally been assumed that milk is milk, and is always competent. In the accumulation of my clinical data, I found striking evidence of an inadequacy in the supply of milk. Figure 11 shows the teeth of a boy, 8 years of age, who has the following history. He suffered from severe convulsions in babyhood, which extended with decreasing frequency to between the third and fourth years. At this time in the spring of the year, the parents moved to a country town, where the child played outdoors and had a different milk supply. His convulsions promptly ceased, and he started growing rapidly. It is probable that the change in calcium metabolism indicated by the change in shape of these teeth corresponds with this change of environment and milk source. Several years later, he developed mild symptoms of convulsions, which were completely relieved by the administration of cod-liver oil. The accumulation of a quantity of this type of data led me to make a study of the comparison of the relative values of different milk, and also of means for enhancing and reinforcing those qualities in milk which are primarily responsible for the improvement of calcium metabolism. A review of the available data revealed that there was relatively little information at hand indicating normal levels of various factors in both blood and milk from the same individual. Before relative values could be observed, it has been necessary, therefore, to establish a group of approximately normal factors and to observe those factors which are influenced chiefly by such changes of diet and environment as will make the milk of such an animal more beneficial for such factors as growth, calcification and defense against disease. Since this is a preliminary report, and since data involved are too voluminous to be included in detail in a single communication, it will be necessary to summarize here some of the factors determined. For these studies, an arrangement was made with a skilled dairyman, whereby a group of sixteen cows were placed under conditions such as would make possible the development of comparable data. We are greatly indebted to Mr. M. J. Barker of the Beardsley Farms, of Garrettsville, Ohio, and his staff for their splendid cooperation, and painstaking assistance in this work.
Fig. 11. Hypoplasia of the enamel, associated with a history of convulsions ending abruptly at about the age of 4 years with a change of the source of the milk supply and with outdoor life.
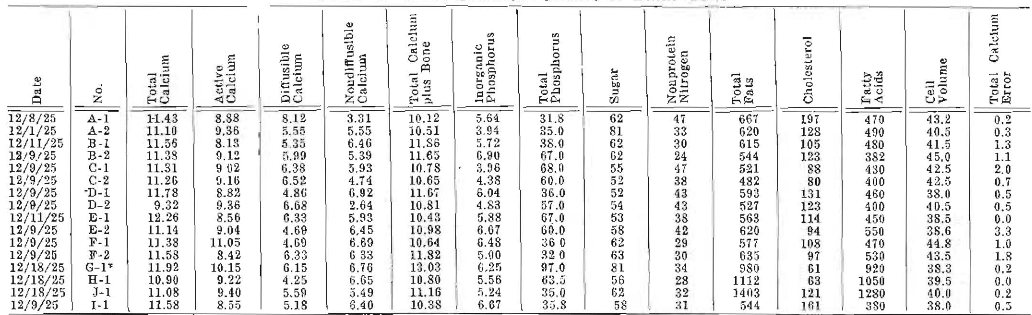
Specimens of blood were taken frequently from the jugular veins for extended blood chemical analyses, and samples of milk were similarly analyzed at frequent periods. Table 1 presents one blood chemical analysis for each of sixteen cows, and Table 2, one set of milk analyses. A control period of about sixteen weeks was utilized in order that levels might be established and individual characteristics noted. Since we have been primarily concerned with calcium metabolism, our blood studies have included total serum calcium, active calcium, diffusible calcium, non-diffusible calcium, serum solubility factor for calcium, inorganic phosphorus, total phosphorus, sugar, nonprotein nitrogen, total fats, cholesterol, fatty acids and cell volume.
Table 1–Blood Chemical Studies of Dairy Cows
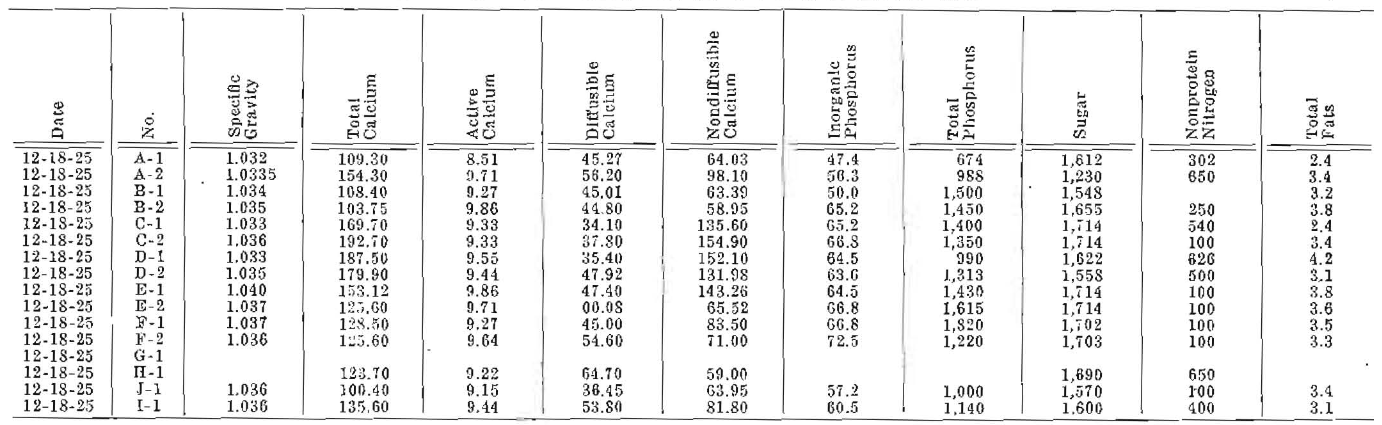
Table 2–Chemical Analysis of Milk of Dairy Cows
For those who are concerned regarding experimental error in making calcium determinations, a column is added, called “total error present.” Determinations were practically always made in duplicate, and often in quadruplicate. The maximum difference in the readings obtained are given in this column, and are shown to be in the order of a plus and minus error averaging 0.85 per cent. It will be observed that in only one determination was the experimental error more than 2 per cent.
The milk determinations were made for the purpose of furnishing data in the following factors: specific gravity, total calcium, active calcium, diffusible calcium, non-diffusible calcium, inorganic phosphorus, total phosphorus, sugar, nonprotein nitrogen, total fats and curd tension. The daily milk output was recorded, and, from this, the total calcium provided in milk was calculated from the concentration of the calcium in the milk. Unfortunately, space does not permit a detailed critical analysis of the data obtained. This will appear later. This is primarily a progress report. The experiment is still in progress and will be for an indefinite period.
Certain factors are of such importance that they must be noted before passing to the summary of the results. The levels for the various calcium factors of the blood of these cows are strikingly comparable to those of humans. The nonprotein nitrogen is from a half greater to double that of human beings. The cholesterol is much lower; in many cases, about half. The total blood fats are comparable. The cell volume is approximately the same.
The milk of the cow provides calcium in a concentration approximately ten times that of the blood. The ratios between diffusible and non-diffusible calcium are very dissimilar from those of blood. Inorganic phosphorus of the milk is approximately ten times that of cow’s blood, which is approximately twice that of the adult human. The sugar of the milk is approximately thirty times as high as that of the sugar of the blood.
After the cows were kept for about four months on their standard ration, they were divided into groups which received special treatment with regard to activators placed in the foods, and the degree of radiant energy to which the cows were exposed. The normal curve of the total milk production runs along at the maximum for about four months, following freshening, then drops progressively during the following five or six months to a level which is approximately one-third or one fourth of the maximum for a period of time which is dependent on subsequent pregnancy and intensity of milking. The rate of fall in production is greatly increased by pregnancy. Certain of the conspicuous results of these researches have been changes in the curve of production. In several instances, we have been able not only to check the normal falling of the total output of milk calcium, but actually to reverse the direction of the curve by producing an increase in total calcium, and this notwithstanding the fact that both the lactation period and the new gestation period were operative in depressing these factors. In several instances, the total calcium of the milk was increased 25 per cent or more. The hardness of the curd has been found to be, in general, quite in proportion to the total milk calcium.
In addition to the blood and milk chemical analyses, biologic determinations were made of the efficiency of the milk for producing growth in animals on deficiency diets, and in preventing and healing rickets-like disturbances in animals on deficiency diets. An evidence of this difference is shown in the following observations on chicks. Ten chicks were placed in each of eight groups, fifty-nine in the ninth group. All received the same deficiency diet, all were the same age and were kept under the same conditions. The group of fifty nine were protected from all ultraviolet light, and were required to eat by ruby lamp. All but two died in two weeks, or 96.5 per cent, whereas, only 10 per cent of those died which were on this same diet, and received sunshine. The percentage of increase in weight in twenty-three days of chicks receiving the sunshine was 98 per cent. For those receiving the same general diet plus milk from the cow kept in the dark barn without either radiant energy or activator added to her diet, the increase was 89 per cent. The chicks receiving the milk from the cow receiving the ultraviolet light gained in this period 122 per cent. The chicks receiving the milk of the cow which was treated with ultraviolet light plus 2 ounces of activated cod-liver oil daily gained 137 per cent, and the chicks receiving milk from the cow which received 2 ounces of activated cod-liver oil daily gained 119 per cent. The chicks receiving the ultraviolet light alone in addition to the ascribed diet gained 91 per cent. The chicks receiving raw cod-liver oil with this diet gained 128 per cent. The roentgenograms show differences in calcification in the different groups. These studies are still in progress and will be published in greater detail later, together with histologic bone changes of the different groups. I have given some detail here from one only of several special biologic studies that are being made to determine the relative efficiency of these various factors.
The data already available indicate that it is possible by means of the addition of activators to the diet of cows and by the exposure of the cows to ultraviolet radiation to enhance certain of the growth promoting and antirachitic factors in the animals placed on diets and in environments which have conduced to the development or disturbances in these regards.
An illustration of the changes in the blood calcium in pregnancy, before and after treatment, is shown in Table 3. There was marked physical improvement as would be expected.
Table 3–Calcium Stress of Pregnancy (Case 1683)*
*Second pregnancy, eighth month. The first resulted in a miscarriage at the seventh month.
Treatment
The accumulated data indicate the need for small doses, three or four times daily, rather than larger single doses daily. Two grains of calcium lactate, with 20 drops of raw cod-liver oil in a capsule, three times a day with meals is better than the same total in one dose. Activated cod-liver oil, from 10 to 20 drops, may be used instead of raw, if care is used not to overactivate. One to five minutes in direct noon-day sun in summer or 10 to 15 minutes in winter is sufficient. Do not activate with the mercury vapor lamp without a filter. If activated cholesterol is used, expose the crystals to direct sun for thirty minutes, then place in a good grade of olive oil to make 3 per cent. It is very easy to give an overdose. Give from 5 to 10 drops of this with each meal, with from 2 to 5 grains of calcium lactate. Owing to the length of this paper, extended details cannot be included here. They are accordingly being put in text form.
References to Technical Methods
Tisdall, F. F.: “Kramer-Tisdall Method for the Determination of Calcium in Small Amounts of Serum,” J. Biol. Chem., 56: 439 (June) 1923.
Clark, E. P., and Collip, J. B.: “Tisdall Method for the Determination of Blood Serum Calcium with a Suggested Modification,” J. Biol. Chem., 63: 461 (March) 1925.
Cruikshank, E. W. H.: “Studies in Experimental Tetany I. Distribution of Calcium. II. Colloidal and Ionic Calcium,” Brit. J. Exper. Path., 4: 213 (August) 1923.
Rona, P., and Takahashi, D.: “Ueber das Verhalten des Calciums in Serum and über den Gehalt der Blutkörperchen an Calcium,” Biochem. Ztschr., 31: 336, 1911.
Vines, H. N. O.: J. Physiol., 55: 86 (May) 1921.
West, Fred, Bauer, J., and Barnickel, K.: “A New Method for the Determination of Calcium and Thrombin in Serum,” J.A.M.A., 78 1042 (April) 1923.
Clark, G. W. Updegraff and Greenberg: J. Biol. Chem., 71: 87-117 (December) 1926.
Clark, Janet: Amer. J. of Hygiene (August) 1923.
Roe, Joseph M. and Kahn, Bernard S.: “A Colorimetric Method for the Estimation of Blood Calcium,” J. Biol. Chem., (March) 1926.
Bloor, J. R.: “A Method for the Determination of Fat in Small Amounts of Blood,” J. Biol. Chem., 17: 377, 1914; “A Method for the Determination of ‘Lecithin’ in Small. Amounts of Blood,” ibid., 20: 133, 1915.
Roe, Joseph H.; Irish, Oliver J., and Boyd, James, I.: “A Study of the Molybdic Oxide Colorimetric Method for the Estimation of the Phosphorus Compounds of the Blood,” J. Biol. Chem. (March) 1926.
“Blood Ph.,” J. Biol. Chem., Vol. 64 (May) 1925.
Moritz, Alan R.: J. Biol. Chem., Vol. 64 (May) 1925.
Michaeliz, L.: Jour. of General Physiology, Vol. X, No. 5.
References Cited:
- Price, W. A.: “New Knowledge of Calcium Metabolism in Health and Disease, with Special Consideration of Calcification and Decalcification Processes, Including Focal Infection Phenomena,” J.A.D.A., 13: 1765 (Dec.) 1926.
- Price, W. A.: J. Indust. & Eng. Chem., 48 (July) 1926.