Access to all articles, new health classes, discounts in our store, and more!
Blood Sugar Levels During the Dental Experience: II. Effect of Sedation on Blood Sugar in the Waiting Room
Published in Journal of Oral Surgery Anesthesia and Hospital Dental Service, Vol. 19, January 1961.
* * *
Since it is well known that anxiety and exposure to stress cause the adrenal glands to liberate endogenous epinephrine and this in turn will mobilize glycogen, the effect of sedation on diabetic and nondiabetic patients undergoing a dental experience has been studied. In sedated and nonsedated, diabetic and nondiabetic patients, the blood glucose level did not show a significant difference 45 minutes after administration of a sedative or placebo when compared with the initial blood glucose level in the waiting room. No significant difference was found when sedation was administered to patients with controlled diabetes and those with uncontrolled diabetes.
In an earlier report,1 it was shown that the blood sugar concentrations of diabetic and nondiabetic patients obtained in the waiting room of the dental office are not significantly different from those derived under basal conditions.
This report will attempt to answer the following questions with regard to blood sugar content:
- Does sedation alter the blood sugar concentration of the nondiabetic patient while he is seated in the waiting room of a dental office anticipating the extraction of a tooth?
- Does sedation alter the blood sugar concentration of the diabetic patient while he is seated in the waiting room of a dental office anticipating the extraction of a tooth?
A third report2 will analyze the effect of epinephrine in local anesthetic solutions on the blood sugar level. A fourth and last report3 will consider the effect of tooth extraction on blood sugar concentration.
Review of the Literature
In order to understand the blood sugar changes which can occur while the patient is seated in the waiting room anticipating the extraction of a tooth, it is necessary to review briefly: (1) the effect of sedative agents on stress, and (2) the action of sedation on the blood sugar level.
Effect of Sedative Agents on Stress–Limited reports are available on the effect of sedative agents on stress. They may be grouped into: (1) studies on lower animals, and (2) studies on human beings.
1. DeBias, Paschkis and Cantarow4 reported that chlorpromazine increases survival rates in adrenalectomized rats exposed to the stress of heat. They postulated that the protective mechanism against heat and cold differs from that which protects against other types of stress.
To investigate the suprarenal response to fatal body irradiation in the rat, Bacq and Fischer5 gave pentobarbital followed by morphine. This was done to inhibit the hypothalamic-pituitary-adrenal response to stress. The premedicant did not decrease the mortality rate since the results were the same in the experimental and control groups.
2. Promazine and meprobamate versus placebos have been studied by Szmyd and McCall6 in relation to postoperative sequelae in human patients after oral surgery. This study showed that pain, swelling and trismus were not affected significantly. This observation is emphasized by the unchanged postsurgical narcotic demand. The duration of postsurgical convalescence actually was increased significantly for patients receiving the drugs. McGuire and Leary7 confirmed the observation that stress tolerance is reduced rather than improved with the use of meprobamate.
Cleghorn and his group8 have observed, in patients with anxiety, that patients with the most severe form have greater relief with the placebo (isotonic saline) than with ACTH.
In a clinical report of 1,100 instances of major surgery, Lear, Chiron and Pallin9 have shown that, although chlorpromazine reduces the need for preoperative narcotics and protects against shock, it has an undesirable hypotensive side-effect.
Beecher,10 in his study using morphine versus a placebo as a pain reliever, has shown that the placebo is 77 per cent as effective as the morphine with the percentage effectiveness decreasing as the pain intensity diminishes. As in the study by Cleghorn’s group,8 the greatest relief with placebos was evidenced when pain symptoms were most intense.
Effect of Sedation on the Blood Sugar Level–The effects of sedation on blood sugar levels as reported in the literature have been well summarized by Goodman and Gilman in 194111 and in 1955.12 It is interesting to note that the 14 years between editions necessitated no change in wording in the following quotation:
“The response of the blood sugar to medication with barbiturates varies considerably, dependent upon the particular drug selected, dose, route of administration, species being studied, and previous diet. In man, soporific doses do not significantly or consistently alter the level of blood sugar and barbiturates are not contraindicated in patients with diabetes mellitus.”
The effect of sedation on the blood sugar level has been reported in: (1) studies on lower animals, and (2) studies on human beings.
1. Studies have been made on cats, dogs, rabbits, pigeons, rats, mice and hamsters. Three studies13-15 utilized adrenalectomized or decerebrated animals. Morphine in adrenalectomized animals does not seem to yield the same hyperglycemic effects observed in control animals.13,14 This seems to be due to the removal of the adrenal glands which ordinarily are stimulated by morphine to produce epinephrine with a resultant mobilization of glycogen.16 In the decerebrated cat, morphine produces a transient hypoglycemia, the mechanism of which is not known.15 Barbital lowers the blood sugar levels, at least for the first few hours, in pigeons and cats.17 Amobarbital and pentobarbital seem to have little effect in lowering blood sugar levels after administration of ether or epinephrine, but pentobarbital prevents hyperglycemia when its administration precedes that of ephedrine.18 Larson,19 in an experiment using rats, found that morphine has little effect on blood sugar levels in small doses (12 mg. per kilogram), and a varied action with larger doses (25 or 50 mg. per kilogram). He found that barbiturates, in contrast to morphine, exert no effect on blood sugar levels when administered in dosages from 25 to 200 mg. per kilogram. Meperidine hydrochloride in small doses (15 mg. per kilogram) has no effect; in medium doses (30 mg. per kilogram), it causes slight hypoglycemia; and with larger amounts (50 mg. per kilogram), hyperglycemia.
Watts,20 in a study of dogs, found sedative doses of pentobarbital to inhibit partially the hyperglycemic response to l-methadone and morphine. In another study on rabbits, Watts21 discovered that preanesthetic amounts of 5 to 30 mg. of pentobarbital per kilogram have no action on blood glucose levels but inhibit the hyperglycemic effects of ether to an increasing degree with increase in dose. Norman and Hiestand22 found that chlorpromazine produces hyperglycemia in the mouse and hamster and no change in the rat.
2. Rosenkrantz and Bruger23 compared the effect of placebos versus phenobarbital in diabetic and normal persons undergoing the dextrose tolerance test. They found an increase in dextrose tolerance in those diabetic patients given phenobarbital. There was also a general increased tolerance on repeated performance for all (diabetic and nondiabetic) subjects, which was attributed to the allaying of anxiety and nervousness. In other words, blood sugar proved to be lower when anxiety was decreased either by drug (for example, phenobarbital) or by adaptation during repeated testing.
Reitmann24 found that the administration of amobarbital sodium intravenously induces a slight hyperglycemia in psychotic and neurotic patients. Occasionally amobarbital also seems to depress transiently the glucose tolerance curve, apparently because of the hypothalamic action of the drug.
Hunter and Merivale25 reported the case history of an adult man who, after a partial pancreatectomy, showed a normal glucose tolerance pattern. Seven years later, after one week of barbiturate therapy, another test definitely indicated diabetes mellitus. After this test the patient was given increasing doses of amobarbital (amylobarbitone) sodium for ten days, and a prolonged true glucose tolerance test was performed. This test again indicated severe diabetes mellitus. The use of the barbiturate was then discontinued. Tests were made on the subsequent twelfth and nineteenth days. Although the sugar level was still high, the curve was more normal. No further tests could be made, but the authors estimated as long as four weeks for the blood level to return to within normal limits.
It would appear from this brief review of the literature that the statement by Goodman and Gilman11,12 still stands and applies to sedative agents in general, though their reference deals more directly with the barbiturates. The report most pertinent to the present investigation is that of Rosenkrantz and Bruger.23 From their study, it appears that persons with anxiety show slight hyperglycemia and that relief of apprehension (either by barbiturates or education) counters the elevated blood sugar.
Method of Investigation
The entire study, which is being described in four parts, was performed on 80 patients in the department of oral surgery of the University of Alabama School of Dentistry. The project included 63 Negroes and 17 white persons; 71 females and 9 males. The patients ranged in age from 16 to 80 years in the nondiabetic group and from 18 to 80 in the diabetic category. Included in the group were 60 nondiabetic, 14 uncontrolled diabetic and 6 controlled diabetic patients.
The procedure used in this study was designed to reproduce, as nearly as possible, the situation in the average office. This plan was previously described in connection with the first phase of this investigation and in a study of the effect of the dental experience on blood pressure and pulse rate.26-29
In the first portion of this study,1 attention was focused on the blood sugar concentration of the patient while seated in the waiting room anticipating the extraction of a tooth. Immediately after the patient arrived in the waiting room, he was seated in a chair, the finger was pricked and capillary blood drawn. All the microestimations of blood sugar were made with Folin filtrate.30 Immediately thereafter, each patient was given a red capsule. In some instances, the capsule contained 1½ grains of secobarbital; in others, the capsule served as a placebo. The investigator did not know the contents of the capsule.
Precisely 45 minutes later by stop watch, the patient was ushered into the operatory and seated in the dental chair. Again the finger was pricked and capillary blood drawn to determine the blood sugar concentration. In this manner the blood sugar level obtained in the waiting room was compared with that observed in the dental chair 45 minutes after the administration of a sedative agent or a placebo.
Actually, 60 experiences in nondiabetic and 20 experiences in diabetic patients were studied under these conditions. The findings of this group of 80 different experiences constitute the subject of this report.
Results
The findings will be considered in two categories:(1) the blood sugar concentration of the patient while seated in the waiting room anticipating the extraction of a tooth, and (2) the level of blood sugar 45 minutes after the administration of a sedative agent or a placebo.
On the basis of the history, the clinical record and a glucose tolerance test, the subjects were divided into four groups: (1) sedated nondiabetic, (2) nonsedated nondiabetic, (3) sedated diabetic and (4) nonsedated diabetic patients.
Blood Sugar Level in the Waiting Room–For sedated nondiabetic patients the blood sugar concentrations ranged from a high of 126 mg. per hundred cubic centimeters to a low of 80 mg., with a mean for the group of 108.5 mg.
In nonsedated nondiabetic patients the blood sugar level ranged from a high of 130 mg. per hundred cubic centimeters to a low of 68 mg. The mean for this group was 102.4 mg.
The sedated diabetic patients showed a greater spread of blood sugar concentrations in the waiting room than was obtained in the nondiabetic groups. Actually, the range for the sedated diabetic patients extended from a high of 282 mg. per hundred cubic centimeters to a low of 100 mg. The mean for the group is 176.4 mg.
For the nonsedated diabetic group the blood sugar concentration ranged from a high of 396 mg. per hundred cubic centimeters to a low of 92 mg. The mean blood sugar level was 182.9 mg.
Blood Sugar Level Forty-Five Minutes After Administration of Sedative or Placebo–In sedated nondiabetic patients the blood sugar concentrations ranged from a high of 130 mg. per hundred cubic centimeters to a low of 84 mg. The mean for the group was 104.3 mg.
In nonsedated nondiabetic patients the range of blood sugar concentrations was from a high of 130 mg. per hundred cubic centimeters to a low of 64 mg. The mean blood sugar level was 99.9 mg.
In sedated diabetic patients the blood sugar concentrations ranged from a high of 236 mg. per hundred cubic centimeters to a low of 100 mg. The mean blood sugar level for the group was 148.8 mg.
In nonsedated diabetic patients the blood sugar concentrations ranged from 388 mg. per hundred cubic centimeters to 92 mg., with a mean of 169.1 mg.
Discussion
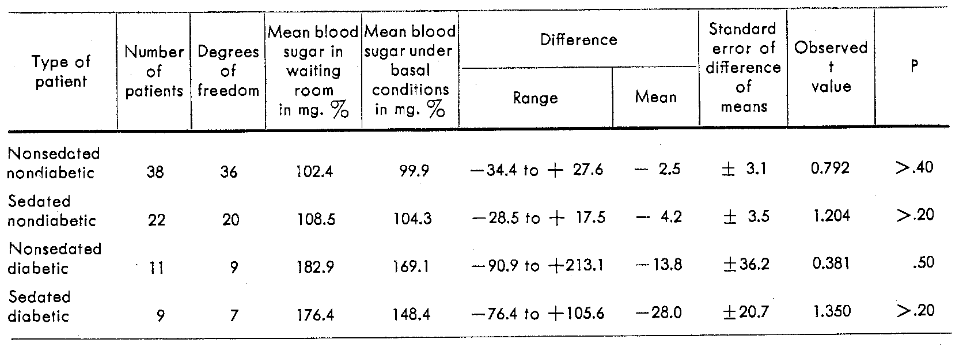
Table 1 indicates that the blood sugar concentration was higher in the waiting room than it was 45 minutes later after the administration of a placebo or sedative agent. This was generally true whether the group was diabetic or nondiabetic, sedated or nonsedated. The decrement is probably physiologic (increasing time since last carbohydrate ingestion) and, in every instance, the probability values indicate the lack of statistical significance.
Table 1–Comparison of the blood sugar initially (in the waiting room) with that obtained 45 minutes later in the dental chair
Table 1 indicates that the diabetic group experienced a greater mean decrease in blood sugar concentrations than the nondiabetic patients. This is true of both sedated and nonsedated groups. Again, the fall is probably physiologic because of the period of fasting. In no instance are the data statistically significant.
The results in these groups are not in agreement with those of Rosenkrantz and Bruger.23 However, no real comparison can be made because of the many different variables in the two experiments. For example, the subjects in their study, if apprehensive, might well have been so only minimally (the only possible cause for anxiety was the threat of a glucose tolerance test). In contrast, the subjects in this experiment were aware of the impending oral surgical procedure.
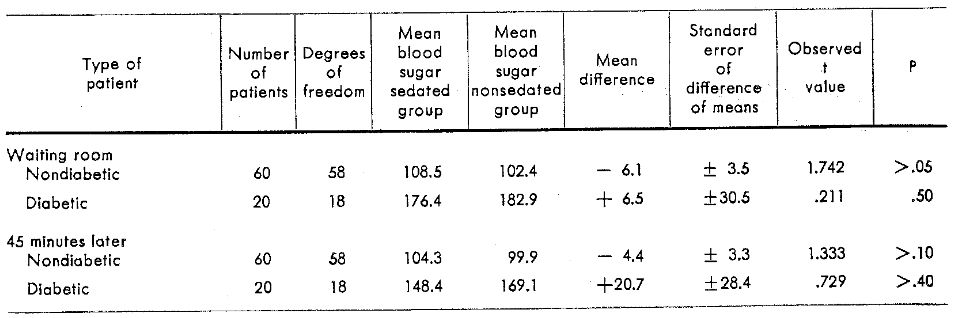
Attention should be directed to the column showing the mean difference in Table 1. The average decrease in blood sugar in the sedated nondiabetic patients is almost twice that observed in the nonsedated nondiabetic patients. In other words, the ratio is approximately two to one. Similarly, there is the same ratio among the diabetic patients with the sedated persons showing a blood glucose mean difference almost twice that noted in the nonsedated diabetic group. However, the lack of statistical significance is clearly shown in Table 2 where the sedated groups are compared directly with the nonsedated groups.
It can be observed in Table 1 that the nonsedated diabetic patient group shows a mean difference five times that for the nonsedated nondiabetic patient group. A seven to one ratio can be seen by comparing the mean differences of the sedated diabetic group and the sedated nondiabetic group. Again, Table 2 shows the lack of statistical significance for this comparison.
Table 2–Blood sugar concentrations of sedated and nonsedated patients in the waiting room and 45 minutes after administration of a sedative or placebo
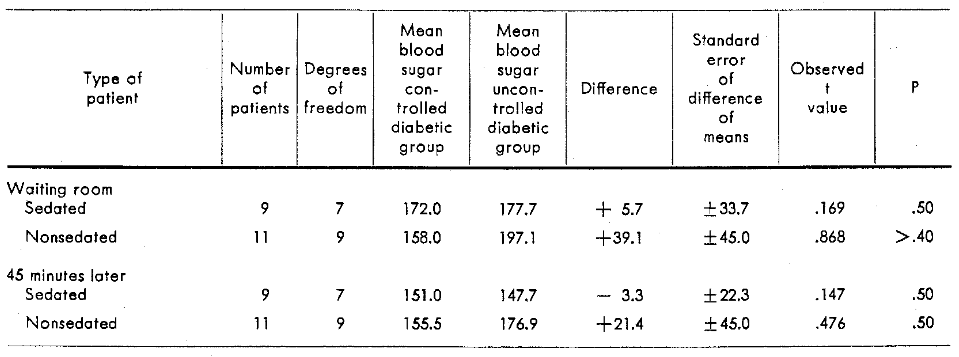
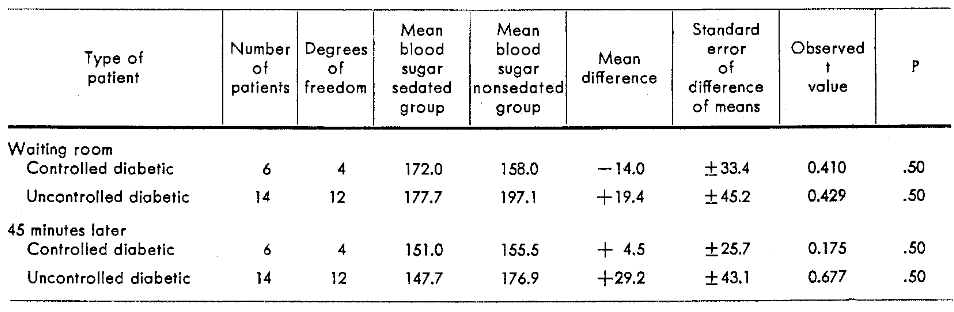
The question logically arises whether the effect on the blood sugar in patients with controlled diabetes mellitus is different from that in patients with uncontrolled diabetes mellitus. Tables 3 and 4 summarize, within the limits of this small sample, that there does not appear to be any significant difference in the glucose behavior of these groups.
Table 3–Blood sugar concentrations of controlled and uncontrolled diabetic patients in the waiting room and 45 minutes after administration of a sedative or placebo
Table 4–Blood sugar concentrations of sedated and nonsedated, controlled and uncontrolled diabetic patients in the waiting room and 45 minutes after administration of a sedative or placebo
Summary
There is a paucity of literature to evaluate the effect of sedation on blood sugar and the action of sedation on stress.
Within the limits of this experiment, the blood sugar concentration of the patient seated in the dental chair 45 minutes after the administration of a sedative or placebo agent appears to be essentially unchanged when compared with the initial blood sugar level while the patient is seated in the waiting room. This result was noted in sedated and nonsedated, diabetic and nondiabetic patients.
A report to follow2 will analyze the effect of epinephrine on the blood sugar during and after the injection of a local anesthetic solution.
Editor’s note: Since the era in which this article was written, society’s understanding of respectful terminology when referring to ethnic and cultural groups has evolved, and some readers may be offended by references to “Negroes” and other out-of-date terminology. However, this article has been archived as a historical document, and so we have chosen to use Cheraskin’s exact words in the interest of authenticity. No disrespect to any cultural or ethnic group is intended.
References Cited:
- Cheraskin, E.; Flynn, F. H., and Fess, L. R. “Blood sugar levels during the dental experience. I. Blood sugar in the waiting room.” J. Oral Surg., Anesth. & Hosp. D. Serv. 18:54, Jan. 1960.
- Cheraskin, E., and others. “Blood sugar levels during the dental experience. III. Effect of epinephrine in local anesthetic solutions on the blood sugar levels.” Unpublished.
- Cheraskin, E., and others. “Blood sugar levels during the dental experience. IV. Effect of oral surgery on the blood sugar concentrations.” Unpublished.
- DeBias, D. A.; Paschkis, K. E., and Cantarow, A. “Effects of chlorpromazine and autonomic nervous system blocking agents in combating heat stress.” Am. J. Physiol. 193:553, June 1958.
- Bacq, Z. M., and Fischer, P. “Action of various drugs on the suprarenal response of the rat to total-body x-irradiation.” Radiation Res. 7:365, Oct. 1957.
- Szmyd, Lucian; McCall, C. M., and Enright, E. T. “Tranquilizing drugs in oral surgery.” J. Oral Surg. 16:310, July 1958.
- McGuire, T. F., and Leary, F. J., III. “Tranquilizing drugs and stress tolerance.” Am. J. Pub. Health 48:578, May 1958.
- Cleghorn, R, A., and others. “Anxiety states: their response to ACTH and to isotonic saline.” In Proceedings of the First Clinical ACTH Conference, 1950. Philadelphia, The Blakiston Co., p. 561-565.
- Lear, E.; Chiron, A. E., and Pallin, I. M. “Chlorpromazine–an adjunct to premedication.” New York State J. Med. 55:1853, July 1, 1955.
- Beecher, H. K. “Evidence for increased effectiveness of placebos with increased stress.” Am. J. Physiol. 187:163, Oct. 1956.
- Goodman, Louis, and Gilman, Alfred. Pharmacological basis of therapeutics, ed. 1. New York, Macmillan Co., 1941, p. 135.
- Goodman, Louis, and Gilman, Alfred. Pharmacological basis of therapeutics, ed. 2. New York, Macmillan Co., 1955, p. 133.
- Stewart, G. N., and Rogoff, J. M. “Morphine hyperglycemia and the adrenals.” Am. J. Physiol. 62:93, Sept. 1922.
- Bodo, R. C.; CoTui, F. W., and Benaglia, A. E. “Mechanism of morphine hyperglycemia.” J. Pharmacol. & Exper. Therap. 57:115, June 1936.
- Bodo, R. C., and Brooks, C. M. “Effects of morphine on blood sugar and reflex activity in chronic spinal cat.” J. Pharmacol. & Exper. Therap. 61:82, Sept. 1937.
- Beckman, Harry. Drugs: their nature, action and use. Philadelphia, W. B. Saunders Co., 1958, p. 195.
- Ellis, M. M., and Barlow, O. W. “Barbital narcosis. II. Blood sugar and blood coagulation time during barbital hypothermia.” J. Pharmacol. & Exper. Therap. 24:259, Nov. 1924.
- Campbell, D., and Morgan, T. N. “On the hyperglycaemic action of certain drugs.” J. Pharmacol. & Exper. Therap. 49:456, Dec. 1933.
- Larson, E. “Effect of hypnotics on blood sugar and on action of insulin.” Endocrinology 44:301, April 1949.
- Watts, D. T. “Effect of methadone isomers, morphine, and pentobarbital on blood glucose of dogs.” J. Pharmacol. & Exper. Therap. 102:269, Aug. 1951.
- Watts, D. T. “Effect of morphine and pentobarbital on ether hyperglycemia.” Anesthesiology 13:33, Jan. 1952.
- Norman, D., and Hiestand, W. A. “Glycemic effects of chlorpromazine in the mouse, hamster and rat.” Proc. Soc. Exper. Biol. & Med. 90:89, Oct. 1955.
- Rosenkrantz, J. A., and Bruger, M. “Effect of phenobarbital on normal and impaired dextrose tolerance.” Am. J. M. Sc. 201:815, June 1941.
- Reitmann, F. “Further observations on sodium amytal experiments.” J. Ment. Sc. 88:122, Jan. 1942.
- Hunter, R. A., and Merivale, W. H. H. “Case illustrating some effects of barbiturates on glucose tolerance test.” Guy’s Hosp. Rep. 103:375, April 1954.
- Cheraskin, E., and Prasertsuntarasai, T. “Use of epinephrine with local anesthesia in hypertensive patients. I. Blood pressure and pulse rate observations in the waiting room.” J.A.D.A. 55:761, Dec. 1957.
- Cheraskin, E., and Prasertsuntarasai, T. “Use of epinephrine with local anesthesia in hypertensive patients. II. Effect of sedation on blood pressure and pulse rate.” J.A.D.A. 56:210, Feb. 1958.
- Cheraskin, E., and Prasertsuntarasai, T. “Use of epinephrine with local anesthesia in hypertensive patients. III. Effect of epinephrine on blood pressure and pulse rate.” J.A.D.A. 57:507, Oct. 1958.
- Cheraskin, E., and Prasertsuntarasai, T. “Use of epinephrine with local anesthesia in hypertensive patients. IV. Effect of tooth extraction on blood pressure and pulse rate.” J.A.D.A. 58:61, Jan. 1959.
- Bio-photocol handbook. New York, Hellige, Inc.