Access to all articles, new health classes, discounts in our store, and more!
The Black Cytotoxic Test
Read at the Twenty-fourth Annual Meeting of the American College of Nutrition, Pasadena, California, May 21, 1960. Published in The Journal of Applied Nutrition, Vol. 13, No. 2, pp. 84-90, December 1960.
* * *
Dr. Pottenger was President of the American Academy of Applied Nutrition from 1959-1960. His final illness prevented presentation in person and the paper was read before the American College of Nutrition by his trusted assistant, Mrs. Ruth Kolseth. Dr. Pottenger’s obituary appears elsewhere in this issue of the Journal.
The diagnosis of food allergy constitutes one of the most baffling problems confronting clinicians. This important paper confirms the original work of Arthur P. Black and provides allergists with another important approach to the recognition of food-allergens.
All clinicians will admit to seeing the occasional dramatic case where the ingestion of a food will result in urticaria, migraine or asthma. Yet, few clinicians are aware of the ubiquity of allergic reactions in the population and how frequently foods are the precipitating factors. This is not strange when you realize that diagnosis has depended, to a large degree, on the symptoms of the patient. Their symptoms frequently follow by several hours or days the ingestion of the food and more often are frequently associated with foods the patient eats every day. Likewise, the symptoms persist for four or five days after the food has been eaten. Finally, other allergens, including bacteria and inhalants, may cause identical symptoms.
One of the first great advances in the diagnosis of clinical allergy was the recognition that certain allergic individuals had a special antibody, called a reagin, in their blood, and its presence could be determined by skin tests. Positive tests were soon ascribed to food proteins; but the final analysis showed that many food-sensitive patients gave negative reactions, that many positive reactions occurred in patients who showed no symptoms following the ingestion of the foods which had given a positive test. Many will agree that skin testing for foods is not a satisfactory way of working out a diet in an average case of food allergy. But, at the same time, they will agree that a strongly positive reaction, for instance to egg, in a child, has considerable clinical significance.
Albert H. Rowe, 1931, published a monograph1 in which he summarized the then current knowledge of allergic reactions due to food and described his “elimination diets” for detection of allergenic foods. Since, in my experience, only 40-60% of perennial symptoms clinically indistinguishable from allergic reactions due to food turn out to be food allergies, the application of elimination diets may become very trying.2 Vaughan, 1934, proposed his Leucopenic Index. This test was not performed under what we would now consider adequately controlled conditions and was not technically satisfactory.3 Coca, 1942, described his pulse-diet record for the identification of food allergens. This has the great advantage of being an objective test. Though there is no question of the association of an increased pulse rate with food allergy reactions, I have not always beensure that the pulse accelerating factor is the particular food I am testing. Randolph, Rinkel and Zeller, 1951, summarized their many clinical studies. These studies have further clarified our understanding of food allergy.4
I have spent my professional life, somewhat frustratedly, trying to solve by means of elimination diets the chronic complaints of many patients with food allergies. Any physician treating chronic disease would welcome an accurate objective laboratory test that would identify the foods to which a patient is allergic. When I first heard of the possibility of such a test two years ago, I undertook to study it.5 Nantz and Blatt described a test for bacterial hypersensitivity which depended on the cytotoxic effect of specific bacterial filtrates on leucocytes from the sensitized patient. Later they6 determined that this bacterial sensitivity depended on an intracellular antibody of the leucocytes, and that in atopic allergy this intracellular antibody was not present. Black, 1956,7 in analyzing the work of Nantz and Blatt, hypothesized that in pollen and food allergy the antibody might be humeral. His subsequent observations confirmed this hypothesis. He suspended leukocytes, generally the patient’s, in the patient’s plasma and added a minute amount of a powdered extract of food protein. The leucocytes were then observed under the microscope for 4 hours. Food extracts, to which the donor of the suspending serum was sensitive, caused the cells to decrease in motility, round up and frequently disintegrate.
Black reported that his preparations lived about 12 hours. Blatt kept his alive 30 hours. At first, in our work, we were unable to keep our preparations alive over 90 minutes when we washed our slides. Later we discovered that by using Clay Adams Gold Seal cover slips and slides as they came from the packages, our preparations lived quite predictably 8-10 hours. These slides and cover slips are prewashed and treated at the factory. We altered Black’s technique in that we used dextran to separate the red cells from the leucocytes as described by Tullis.8 We did not siliconize the glassware. A minute amount of food protein was stirred into a drop of distilled water, spread evenly over the area of the slide to be covered by the coverslips and allowed to dry. A slight, but measured, excess of cell suspension was then put on the slide. Sealing of the cover slip with paraffin was delayed until sufficient evaporation had taken place so that the cover slip adhered to the slide. The preparations were studied by transmitted light using a Bausch and Lomb microscope with a 10X wide field eyepiece and a 60X achromatic objective. Various filters for the light source were tried. The biological function of the leucocyte measured was the amoeboid activity. If a leucocyte appeared round without any pseudopodic projections, it was counted as round. All others were counted motile.
Although some cells appeared round in healthy, newly mounted preparations, and although cells appearing round sometimes would put out pseudopods again, the percentage of round cells could be used as a rapid way of estimating the health of the preparation. The cover slip preparations were mounted six to a 2 x 3 inch slide, using five foods and one control. Counts of 25-40 cells were made at 1 hour, 2 hours and 4 hours. Figures I and II.
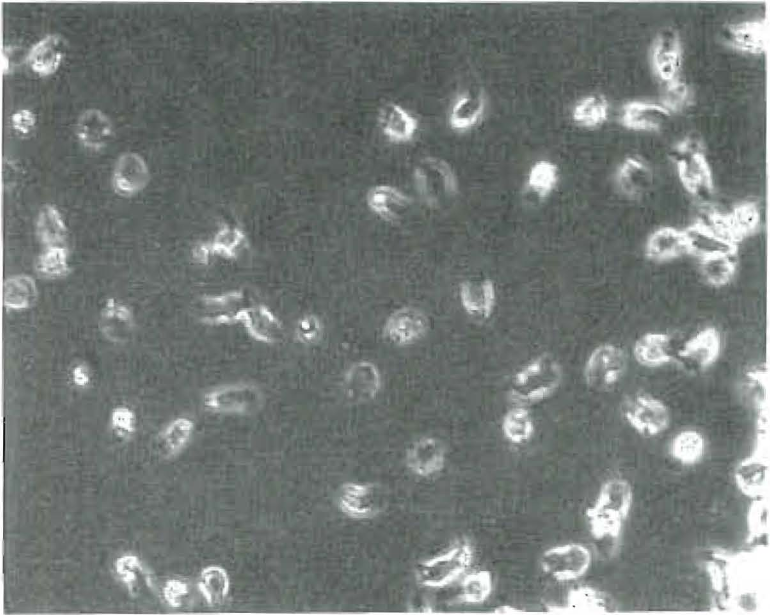
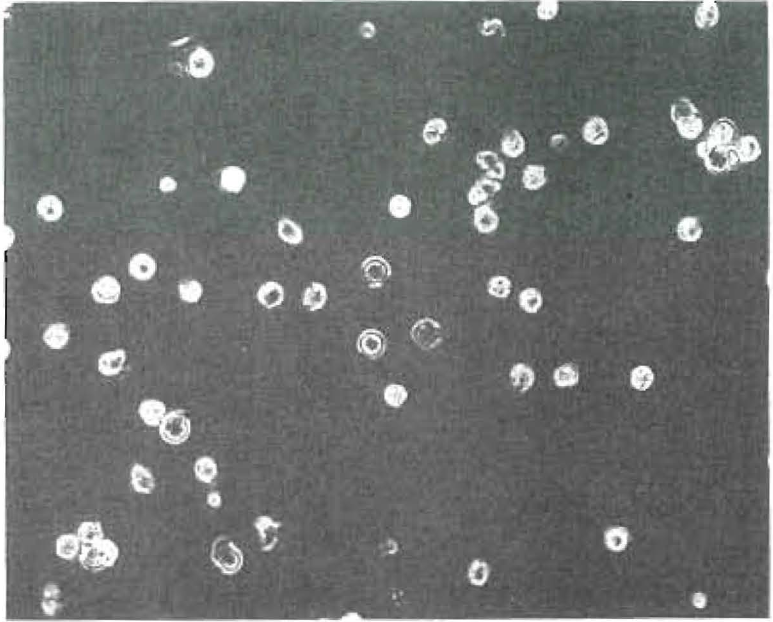
Figure 1. Phase contrast photograph of viable white cells 7 hours after withdrawal of blood from patient and 4 hours after introduction of cell suspension to the antigen. All cells demonstrate viability. No interaction between serum and antigen. This constitutes a negative Black Test. Objective at 25X achromatic. Eye piece 8X, camera 32 X; optivar 1.25. Exposure time 1/25 sec.
Figure 2. Phase contrast photograph showing effects of interaction between the serum and food antigen, 7 hours after withdrawal of blood from patient, and 4 hours after introduction of cell suspension to the antigen. All cells are round and demonstrate loss of viability. This constitutes a positive Black Test Objective 25X Neoflor Eyepiece 8X. Camera 32X. Optivar 1.25. Exposure time on photocell 6.
The clinical evaluation of the Black Cytotoxic Test was made on three groups of patients. Our first group was one of twenty old patients whose diets had previously been successfully worked out. Our second group consisted of a number of patients whose continuing minor symptoms after the completion of elimination diet trial led us to believe that some error had crept into our elimination technique. Finally, our third group was made up of new patients who were started on diets worked out by the Black Test. We ran 40 food tests and 8 controls on a single blood sample, repeating the tests a week later. The technician knew nothing of the clinical picture of the patient, had no access to the results after finishing her report each day. As we ran three blood samples a day, memory could not influence the second count.
Our first problem was to determine what constituted a positive cytotoxic effect. Our approach to this problem was to analyze 237 consecutive controls which were counted at 1 hour, 2 hours and 4 hours. We abandoned the one hour count as approximately 7% of the cells were round when first put on the slide. This percentage persisted through the first hour and there were too few cells for statistical analysis. These 237 controls at the end of 2 hours gave an average count of 13.9% round cells, and all but 2.1% of these counts were less than 50% above this average figure. In other words, 97.9% of the counts were less than 20.85% round. At the end of 4 hours, the average number of round cells in the 237 controls was 21.75%, and 98.3% of all the counts were less than 50% greater than this figure, or 32.62%. The sum of the two percentage figures, that would include 98% of the 2 hour and 98% of the 4 hour counts, would be 53.5. An analysis of 658 foods showed that the average percentage of round cells in the negative foods ran two percentage points higher than the controls. Of the clinically negative foods, 4% of the counts were between 35% and 45% round at 4 hours. It is statistically inconceivable that the difference between the food tests and the controls could represent chance distribution. We concluded the best index of cytotoxic effect was obtained by adding the figures for the percentages of round cells at 2 and 4 hours. We then assumed that any figure under 60 represented a negative test, and any figure 60 or over represented a positive one.
I noted above that our clinical material was composed first of old patients whose food allergies had been worked out by diet trial, and who had been free from symptoms by avoiding the offending foods from 1 to 10 years. These patients were tested to varying numbers of foods they were eating plus the foods we thought had produced symptoms. In this group the foods that agreed clinically generally showed no cytotoxic effect on either cover slip preparation and were called negative. About one out of seven showed a cytotoxic effect on one cover slip and none on the other. There were 27 foods in 141 tests in a group of four old patients that showed a cytotoxic effect in both cover slip preparations. Two of these foods were clinically negative. Four were clinically positive and the patient had had allergic reactions attributed to each of them in the last 6 months. In 21 positive tests, the foods had not been eaten for 6 months or more. Thus, there were two false positives in 27 tests. However, the important observation was that in 3 cases foods known to have caused allergic reactions in the past gave a negative test on both cover slips. In these 3 cases the patients again developed symptoms after ingesting the food for 3 days to 2 weeks. Subsequent tests showed a positive cytotoxic effect was then present. This is consistent with the observation that humeral antibodies disappear from the blood stream after sufficient time has elapsed since the challenging dose of antigen. It is also consistent with the known development of tolerance on withdrawal of a food. An illustrative case report of such a reaction follows.
On October 6, 1958, a 54-year-old housewife was given a diet of 19 foods which had shown no cytotoxic effect on either cover slip. Of these 19 foods, the patient suspected two–eggs and chicken. A clinical trial done in 1940 had given no evidence that they upset her. In the diet were cashew nuts and coconut, which had never been checked by clinical trial but which the patient rarely ate–3 or 4 times a month in small amounts. Eight days after starting the diet, the patient reported she had developed hives, had much gas, heartburn, a loose stool and “felt awful.” She then put herself on a diet she believed agreed with her. However, her bowel still was upset. On November 10 she ate cashew nuts for 3 days and again had hives and indigestion. After clearing, she ate peanuts 3 days and had indigestion.
The end of November we ran the food tests to the balance of the 70 foods we were testing, plus the doubtful results of the first test. We found her now positive to cashew and coconut and peanuts. Eggs were still negative. We showed her that squash disagreed with her, which she had in her “basic diet.” We instructed her to go back on all the foods in the first diet except cashew and coconut. She would not try eggs again.
On January 12, she was living on a diet as worked out by the Black Test. She felt very well and had no complaint. Later she reported she was eating eggs in desserts without difficulty.
Our second type of clinical material was composed of patients whose symptoms persisted after the completion of their elimination diets. These persistent symptoms raised the question of error in our diet technique or error in our diagnosis. This moment of doubt is very hard on the morale of a patient and Black’s Test was often of great help. One such patient was a young mother of 27 years of age on whom a diagnosis of ulcerative colitis had been made. She responded beautifully to an elimination diet. Shortly after her diet was worked out, she went to Italy to live for a year. She was unable to control her diet and finally returned, having lost a good deal of weight and being quite physically depleted. She had a return of one of her original complaints, diarrhea. I started her back on her diet and gave her cod liver oil as a tonic. She improved but went along for several months with continuing symptoms. Finally, we ran a Black Test on all the foods she was eating and found both cover slips showed a positive cytotoxic effect to codfish. Stopping cod liver oil resulted in the complete control of her diarrhea. Analyzing her record, I found I never had tested her to codfish. Though the test rescued a number of patients, it was not without its drawbacks, as there was the occasional false positive test and the all too frequent tests where one cover slip was positive and the other negative, which tended to confuse the patient.
The third type of clinical material was the new patient on whom we had no information except the initial examination. We had these patients divide the foods they were eating into three groups–first, foods they ate twice a week or more often; second, foods they had had 1-8 times a month in the last 6 months; and third, foods they had not eaten for 6 months or more. We then tested the patient to some 70 foods. We made up his initial diet from the first group of foods which had shown a double negative test. About 3 times out of 4 this diet resulted in the clearing of symptoms. An example of the effectiveness of the technique is a female child, age 17 months, who weighed 19 pounds, did not care to eat and was considered a fretty, cross child. She had frequent colds. We used her serum and her father’s white cells and tested the seventeen foods she was eating regularly. She gave a positive test only to codfish. After stopping her cod liver oil she became an extraverted, energetic child and in 5 months gained 7 pounds.
In the clinical evaluation of a test for food allergy, you are confronted with frequent dietary indiscretions on the part of the patient which invalidates any statistical conclusions in a consecutive series of cases. It is impractical to retest patients routinely to the foods which give a positive test. On the advice of Dr. Fred Moore, we used 200 consecutive food tests which fulfilled the following criteria. First, the patient had to be eating the food at least twice a week, as indicated in answering a questionnaire. Second, both cover slips had to show no evidence of a cytotoxic effect according to the criteria described above. Third, there had to be an adequate clinical trial of the food after the test. Of the 200 foods tested, 194 were clinically negative and 6 clinically positive. One of the negative tests came from a patient with a proven allergy to wheat of 3 years duration, who nibbled a little twice a week. When told she could eat wheat she then indulged freely-developed her gastrointestinal and arthritis symptoms after 5 days. Subsequently, the Black Test became positive.
Our next study was concerned with foods a patient had not eaten for a year or never had eaten. Again the foods were selected by using the results of a questionnaire as the first step. The second step was to perform a Black Test and select from the test only those foods which showed both cover slips negative. The third step was to run a clinical trial of those foods for a week or more, and then retest them by the Black Test. Our results showed that in 7 cases where the first Black Test was negative, the foods when ingested produced symptoms and the Black Test subsequently became positive. In 20 cases, the foods caused no symptoms after eating every day for a week and the Black Test remained negative.
When we tested 70 foods using 2 cover slip preparations per food, there would be on the average 10 foods that would have one cover slip show a cytotoxic effect and the other none. Subsequent clinical trial showed that about seven times out of 10 single positive cover slips represented a false positive test and three times out of 10 the single negative cover slip would represent a false negative. The test was technically quite difficult and tedious.
Dr. Moore pointed out that there was a large overlap between the area of normal death of cells and the area of cytotoxic activity. He advised us to work along two lines, prolonging the period of motility of the healthy cells and enhancing the cytotoxic effect.
It should be noted here that Black, in his paper, reports a much more complete cytotoxic effect than we observed. It is important to remember that the normal life of a leucocyte is four or five days, so that a toxic effect is manifest in all the coverslip preparations and it is probably variable. Nevertheless, the test, as described in this paper, is statistically significant and clinically very useful. Our present studies indicate the test will become more accurate.
Addendum*
In addition, what we were trying to do subsequently was to develop a technique that could be photographed so that we would have a permanent record. This led to the chance observation that, in one instance, 80% of the cells with which we were working were still alive and active at 24 hours. If we could develop a technique in which the cells would normally live 80% motile at 24 hours, this technique would mean that no toxic effect was being exerted on the cells by anything but the allergens, as the normal life of a leucocyte is approximately 5 days.
In trying to find out why the cells died generally in 6 to 8 hours, we observed several phenomena. Heavy metals, such as chromium, used in cleaning glass slides, were implicated. Detergents, even those suitable for tissue culture work, seemed to be toxic. The glass after exposure to snog became toxic. Further studies showed that the toxicity of glass itself varied from batch to batch, as well as the methods used in handling the glass.
Dr. Pottenger planned to discuss these latest developments of the Black Test in his Presidential Address, and they will be reported fully at another time. The Black Test, as we perform it, is now an excellent diagnostic tool and reproducible.
Note:
*Dictated by Dr. Pottenger May 20 while in the Huntington Memorial Hospital.
References Cited:
- Row, Albert H.: Food Allergy, Lea and Febiger, 1931.
- Vaughan, W.: Journal of Allergy, 5: 601, 1934.
- Coca, A. F.: Familial Nonreaginic Food-Allergy, Charles C. Thomas, Springfield, 1st Ed., Dec., 1942.
- Riokel, Herbert J., Randolph, Theron C., and Michael Zeller: Food Allergy, Charles C. Thomas, Springfield, 1951.
- Blatt, Hermaon and Frank A. Nantz: “The application of a tissue culture technique in the clinical evaluation of bacterial hypersensitivity,” Allergy, 5: 554-557, 1947.
- Ibid: “Further studies on the use of tissue culture of blood leukocytes in the clinical evaluation of bacterial hypersensitivity of the tuberculin type,” Allergy. 8: 662-625, 1950.
- Black, Arthur P.: “A new diagnostic method in allergic disease,” Pediatrics, 17: 716-724, 1956.
- Tullis, J. L.: “Prevalent nature and identification of leokocyte antibodies.” Eng. J. Med., 258: 578-82 Mar. 20, 1958.