Access to all articles, new health classes, discounts in our store, and more!
A Study to Determine the Effectiveness of Aphanizomenon Flos-Aquae to Alter the Course of Leprosy in Humans
Preliminary proposal, undated.
* * *
Introduction
“Then Naaman, captain of the armies of the King of Syria, went down and dipped himself seven times in the River Jordan, according to the saying of the prophet, Elisha, a man of God: and his flesh, ravished by leprosy, came again like unto the flesh of a little child, and he was healed.” – II Kings 5:14
“And many lepers were in Israel in the time of Elisha, the prophet; and none of them was healed, saving Naaman, the Syrian.” – St. Luke 4:27
Since this dramatic episode was recorded in Biblical scriptures, man has sought to unravel the mysteries surrounding the most dreaded disease described in historical literature–leprosy. Feared by all humans alike, its contraction meant social isolation and a slow degenerative death. As Mother Teresa, Nobel Laureate, has so poignantly stated, “even today with modern medication, the disease of leprosy continues almost unabated because without LOVE, the leper can know no healing.” The relationship between the way a person thinks of himself and the physical condition of that individual are quite similar. To “wipe out” leprosy from the world’s human population in our lifetime would require, in our opinion, a mechanism by which we could physiologically “connect” the brain to the immunological systems (identified and unidentified) of the human body–direct the brain to regulate the body’s defense mechanisms. To reach the necessary populations, the brain-to-body connective treatment would need to be easily administered to the affected populations and would necessarily require a low technological level of input since, in most areas affected by leprosy, it is nonexistent. We propose to evaluate a type of treatment/preventive for leprosy that would be able to pass the above criteria. We believe that the proposed treatment will prove to be as effective in treating “modern leprosy” as was the treatment which Elisha, the prophet, prescribed for Naaman.
We propose to initiate a study to determine the effectiveness of utilizing the blue-green algae Aphanizomenon flos-aquae to treat the symptomology associated with various types of leprosy in humans. The basis for the proposed study exists in our work with patients having the corollary disease Herpes Simplex–Type II. This disease is a virus-induced malady which has brought about a group of “modern” social outcasts. It is reported by the National Institutes of Health that Herpes Simplex–Type II is the second most prevalent venereal disease in the United States and may soon overtake syphilis. There was no known reported cure in January 1980 for Herpes Simplex–Type II. It is characterized by outbreaks of blisters in the genital region; the blisters break, releasing billions of active vires, which are passed on by sexual contact. Most scientists describe the clinical manifestations of this disease as being 75 to 85% psychologically induced, and the retention of the disease as nearly 100% neurological- physiological dysfunction. In all patient cases (≅25) with which we have been associated, and who have undergone a 30- to 60-day treatment using specially prepared algal extracts, there has been complete remission of “outbreak” symptomology. The tentative conclusion regarding the active mechanisms of the extracts used is as follows: The orally ingested material raises the energy level of the body by stimulating brain-to-body recognition sequences that effect the individual’s “self image.” When this happens, the outbreaks no longer appear. Since there is no method for detecting the “nonpresence” of the virus in the central nervous system, we are not able to evaluate the exclusion of the virus from the body, only the lack of clinical manifestations. An attached article written by us is included as part of the proposal to more clearly describe our concept of disease as it is related to the body’s free energy and brain function.
Proposed Clinical Testing Program
Description
The clinical program of treatment is unusual in that it does not call for drug therapy. Rather, the algal-based substances which will be administered to leprotic patients are essentially nontoxic foods (not classical medicines) which have been shown to have efficacious side effects in the treatment of some bacterial and viral infections. To improve our understanding of the physiological and psychological benefits of these unique algae-based foods, we propose a limited group study of leprotic patients who would ingest them for a one-year period. This time period is similar to that used for our studies with Herpes Simplex–II and Sickle Cell Anemia. During this time, the test individuals would be monitored for their progress, insofar as conditions permitted.
We propose to call this limited research effort “THE NAAMAN PROJECT” and to coordinate it with the efforts of existing leprosy treatment centers. THE NAAMAN PROJECT calls for selection of from 25 to 100 patients who have been diagnosed as having one of the “typical” types of leprosy. They would first be given medical examinations and the relevant medical information would be compiled and entered into the individual’s project file.
Under our supervision, in cooperation with the local participating entity, the “NAAMAN” patients would be divided into several test groups and given adequate materials to last for a one-month period. Each month, the clinical progress of each patient would be evaluated at the appropriate center and at that time they would receive their next month’s supply of test materials. Consumption levels would be varied each month depending on the patient’s progress. Clinical data on the “NAAMAN” patients would be compiled and analyzed each month. After 12 consecutive months of test food consumption and clinical monitoring, the “NAAMAN” patients would again be given a complete medical examination and the final data compiled by participating groups. Results would be published within six months after completion of the study.
Follow-up monitoring and test food distribution would continue for an additional period of time for any patient who desired it. Judging from our experience with other patients we have worked with, we would need to be prepared for the continued use of these materials–initiation of a phase II project.
Objectives
The set of objectives for THE NAAMAN PROJECT could be infinite if everyone’s desires were included. However, the proposed project budget does not permit an inclusive compilation; thus, only those objectives considered to be primary target areas are described.
The objectives are as follows:
- To determine the effectiveness of Aphanizomenon flos-aquae (Klamath Lake strain) to alter the course of leprosy in humans.
- To identify differences in effect of the test food materials among different leprotic types. Isolate areas for further study and application.
- To launch large-scale, second-phase applications to test for regional/ environmental effects. This objective assumes that there will be a statistically significant success rate during the phase-I study. Success in this context means satisfactory control of the leprotic disease.
- To isolate, characterize, and possibly synthesize the active constituent(s) in Aphanizomenon flos-aquae (time and funds permitting, otherwise phase-II objective).
- To stimulate other independent research into the effects of nutrition on leprosy as a consequence of this effort and the resulting publications.
- To stimulate interest in eliminating leprosy during the next 10 to 20 years.
Proposed Budget
The costs of operating this project are low relative to the value of the expected research results.
Second-phase research will be considerably more expensive. By undertaking a more limited program at this time, which can be closely monitored at a moderate expense, we will be able to carefully target research areas for more expensive secondary work. And we will do so in the happy knowledge that we have correctly targeted phase-II work.
The first stage of THE NAAMAN PROJECT will cost about $25,000 by our best estimates. The real cost will perhaps be significantly higher, but these additional costs should be absorbed by existing programs funded from other sources. The test materials will be provided by KC Laboratories at their cost. The funds will be used to purchase test materials, provide administration of clinical data, provide for publications, pay for travel expenses, and pay for overhead costs incurred during the project.
Preparative Experimental Procedure
Cell Culture
The blue-green algae Aphanizomenon flos-aquae are obtained from Upper Klamath Lake which is located in Klamath County, Oregon. The Klamath strain of A.flos-aquae grows abundantly in the lake during the summer months and exists in the water at concentrations of 30 to 60 mgℓ-1. The algae are removed from the lake water (Fig. 1), washed with tap water, and concentrated to a thick paste (Fig. 2). The algae are flash frozen in plastic bags (30 cm x 30 cm x 2.5 cm) and stored at 0°C.
Fig. 1. Algae harvesting on Upper Klamath Lake. The continuous belt micro-sieving screens are shown mounted on a support barge. Each screen removes about 40 kg of algae per h from the lake.
Fig. 2. Secondary algae concentration equipment is located onshore about 45m from the primary dewatering equipment. Algae is pumped from the barge onto the screens where a vacuum is applied to remove additional water. The algae are removed from the micromesh screen (shown in figure), placed in plastic bags, and flash frozen.
Cell Analysis
Each harvest of algae is chemically analyzed to determine its quality and purity. Cellular protein, carbohydrate, lipid, and trace mineral content is determined by described methods and procedures. The algae are analyzed for the presence of heavy toxic metals (lead, cadmium, mercury, selenium, and arsenic). Further, the algae are analyzed for the presence of lindane, aldrin, other chlorinated hydrocarbons as p,p’-DOE and PCBs (as Arochlor 1260). The analysis for the presence of microorganisms includes aerobic plate counts, yeast, mold, coliform (MPN), coagulase positive staphylococcus, and salmonella.
Isolation and Preparation of Materials for Oral Ingestion
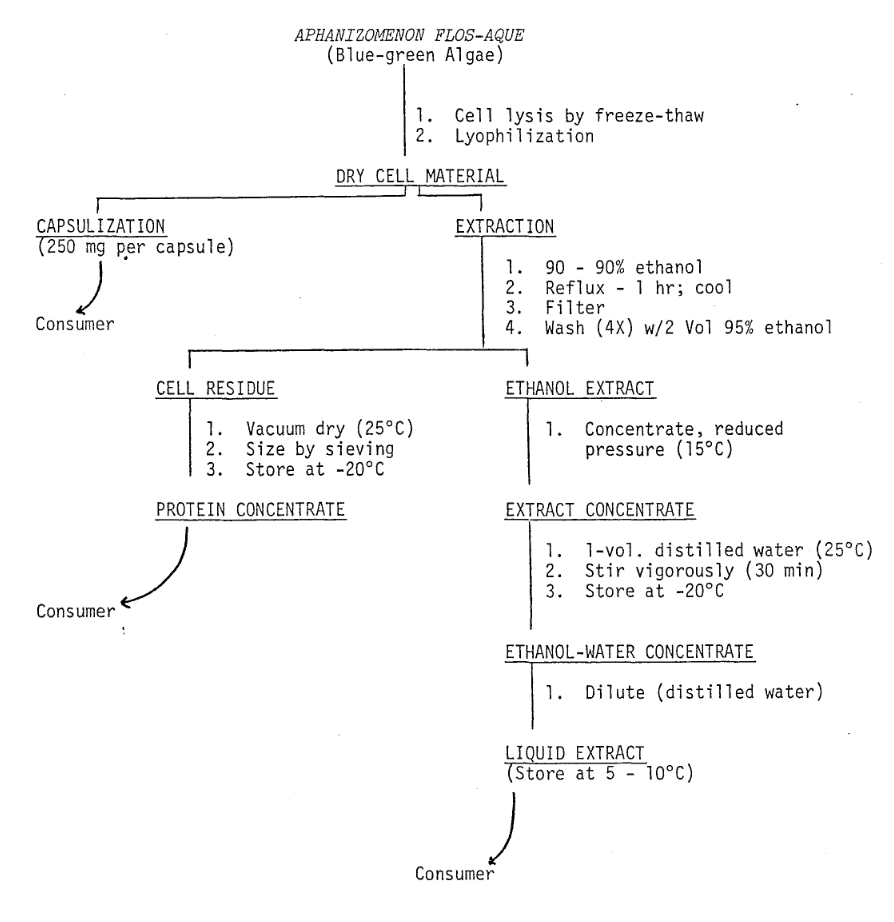
The fresh frozen algae are lyophilized using a virtus U.S.M.-15 freeze dryer equipped with stainless steel trays and automatic temperature controlling equipment which maintains the product at 15°C during the drying process. The freeze-dried cell material to be encapsulated (Fig. 3) is passed through a fine-mesh stainless steel screen to ensure uniform material size. The algae (250 mg per capsule) are subsequently packaged into size 0 gelatin capsules by Cosmo-Pharm, Inc. (North Hollywood, CA).
Fig. 3. Flow Diagram: Biochemical Preparation of Administered Material
Freeze-dried algae to be used for preparing protein concentrates and concentrated liquid extracts are suspended in ethanol (10:1 w/v) and refluxed. The suspension is filtered and the cell residue washed with ethanol (Fig. 3). The cell residue is dried in vacuo (25°C), sieved, and supplied to the consumer in powder form. The alcohol extract is concentrated using reduced pressure and steam. During volume reduction, the product temperature does not exceed 15°C. After the volume of the extract is sufficiently reduced (80 g solids ℓ-1), sterile distilled water is used to increase the volume (40 g solids ℓ-1). For consumer use, the concentrate is diluted with sterile distilled water to the desired concentration.
Preparation of Material for Nasal and Bronchial Application
Liquid concentrate containing 40 g solids per ℓ of solution is diluted with buffered normal saline (pH 7) to give a final concentration of 0.4% solids. The buffered solution is filtered and stored (4°C) in sterile glass containers. For dispensing to the consumer, the solution is placed in a nasal aspirator which is similar to those used to dispense common nasal decongestants.
Consumer Dosage
It is not possible to describe a single useful dosage level for everyone since individual differences exist. However, it is possible to outline a “typical” adult (40 yrs of age) level of consumption. Capsules (250 mg each) of algae (BLUE-GREEN MANNA)TM are provided at the average rate of 4 to 6 per day. Liquid algae concentrate (MANNACOL)TM is administered by volume (2 ml per day) using a calibrated dropper. The nasal spray/bronchial mist (MANNAMIST)TM is used two to four times daily, or as required. Protein concentrate is provided as a powder which is called MANNAPRO.TM The protein powder is a pure protein extract obtained from Aphanizomenon flos-aquae; it is consumed at the rate of 20 to 50 g daily depending on the patient’s consumption rate of other protein sources. For individuals who are unable to consume the material orally for certain clinical reasons, it is possible to provide various combinations of algal material to be used in intravenous applications.
Project Starting Date
Work on the project would begin as soon as funding is available. Administration of test materials would begin when selected patients have been identified and enrolled in the course of treatment.
GREATER THAN THE TREAD OF MIGHTY ARMIES IS A NEW IDEA WHOSE LIGHT HAS COME.